Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.108 n.3-4 Pretoria Jan. 2012
REVIEW ARTICLE
Factors influencing the crystallisation of highly concentrated water-in-oil emulsions: A DSC study
Karina Kovalchuk; Irina Masalova
Research Rheology Laboratory, Engineering Faculty, Cape Peninsula University of Technology, Cape Town, South Africa
ABSTRACT
Highly concentrated emulsions are used in a variety of applications, including the cosmetics, food and liquid explosives industries. The stability of these highly concentrated water-in-oil emulsions was studied by differential scanning calorimetry. Crystallisation of the emulsions was initiated by exposing the emulsions to a low temperature. The effects of surfactant type, electrolyte concentration and electrolyte composition in the aqueous phase on emulsion crystallisation temperature were studied. Surfactant type affected the emulsion crystallisation temperature in the following order: PIBSA-MEA=PIBSA-UREA<PIBSA-MEA/SMO<PIBSA-IMIDE<SMO. These results are in the same sequence as results obtained for the stability of these emulsions in aging studies, that is, PIBSA-MEA was the most stable with age and SMO was the least. The effect of the surfactant type on emulsion crystallisation can probably be attributed to the differing strengths of the surfactant-electrolyte interactions, which result in different molecular packing geometry and differing mobility of the surfactant lipophilic portion at the interface. These results enhance our understanding of the factors that affect the stability of explosive emulsions.
Introduction
Highly concentrated emulsions are used in a variety of applications, including the cosmetics, food and liquid explosives industries. The droplets of these emulsions comprise a super-cooled aqueous solution of ammonium nitrate salt dispersed in a paraffin-based dispersant, which results in a thermodynamically unstable system. Hence, the system 'ages' with time, resulting in changes to the rheological properties and phase composition of the system. The flow or rheological properties of liquid explosives (highly concentrated water-in-oil emulsions) are well described and are of practical importance in their application in open-cast mining.1,2 The instability of these emulsions is related to the crystallisation of the ammonium nitrate solution inside the emulsion drops. In most cases, the crystallisation of solutions of super-cooled phases is quick, where small nucleation sites within the system cause the entire system to change over short periods, such as minutes or seconds. However, the crystallisation processes of emulsions of super-cooled salts are kinetically slow and can take up to a few months. The instability of the emulsion results in droplets rupturing, which in some cases can initiate further nucleation and crystallisation of adjacent droplets. The emulsion droplets usually contain a range of additives and surfactants that stabilise these colloidal two-phase systems by preventing contact between the dispersed droplets. A number of studies have shown that an emulsion's crystallisation depends on the formulation and ratio of the various components1,3,4,5 and has a significant influence on a number of rheological properties, such as the ability to transport and pump the material to its application, and on its subsequent detonation ability.
The emulsion stability, generally speaking, depends on the properties of the interfacial layer that forms between two immiscible phases, and the interfacial properties are influenced by surfactant type and behaviour at the interface (i.e. surfactant interactions with the ammonium nitrate solution).3,6,7,8
Differential scanning calorimetry (DSC) is a suitable method for studying the behaviour of emulsions submitted to a temperature gradient. As reported in certain publications,3,4,5,9,10,11,12 the freezing temperatures of emulsions have been determined and successfully correlated to the emulsion stability. Moreover, the freezing temperatures have been found to be dependent on the emulsion composition. In the present work, the DSC method was used for the investigation of emulsion freezing (or crystallisation) temperatures as a function of surfactant type and concentration. The effects of electrolyte concentration and the admixture of sodium and calcium nitrates in the aqueous phase on the emulsion stability were also studied. The aim of the present work was to investigate the effects of surfactant and electrolyte concentrations on emulsion stability.
Materials and methods
Materials
Mosspar-H oil is manufactured by PetroSA (Cape Town, South Africa) and constituted the oil phase of emulsion. The polyisobutylene succinic anhydride (PIBSA)-based emulsifiers used for the emulsion preparation and stabilisation are produced and supplied by Lake International Technologies (Johannesburg, South Africa). This oil comprises mainly isoparaffins (80% - 90%), whilst n-paraffins and cycloparaffins constitute 10% - 15% and aromatics less than 0.1%. The oil density is 792 kg/m3 at 20 °C.
The surfactants used in preparing emulsions were based on organic derivatives of PIBSA, especially the alkanolamine derivatives. The following materials were used in concentrations from 8% to 20%: PIBSA-MEA, PIBSA-UREA and PIBSA-IMIDE. PIBSA-MEA is a PIBSA of molecular weight 1048, reacted approximately 1:1 with monoethanolamine to an uncondensed amide-acid head group. PIBSA-UREA is an adduct of PIBSA and urea. PIBSA-IMIDE is PIBSA-MEA condensed to an N-substituted pyrrolidinedione (succinimide) structure. The tail group is polyisobutylene, the repeat unit of which is -C(CH3)2-CH2-. A molecular weight of 1048 corresponds to 19 repeat units in the chain. The hydrophilic-lipophilic balance of all surfactants is low (between 2 and 4).
A low molecular weight surfactant, sorbitan monooleat (SMO), and its mixture with the polymeric surfactant were also used for the investigation. SMO is an ester formed between sorbitan and oleic acid (oleic acid is a C18 fatty acid with a single cis double bond). A mixture of PIBSA-MEA and SMO with the ratio PIBSA-MEA to SMO of 10:1 by weight in the oil phase was used.
The structures of the PIBSA-based surfactants' head groups are shown in Figure 1.
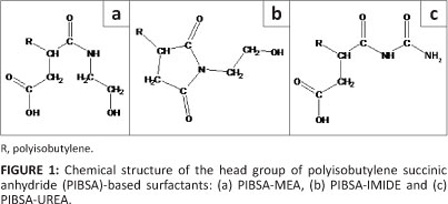
Emulsions with different concentrations of ammonium nitrate in the aqueous phase were used to investigate the effect of salt concentration on emulsion crystallisation. Compositions with varying additions of calcium and sodium nitrates in an aqueous phase were also studied.
Methods
The emulsions studied were water-in-oil type. Details of the composition and preparation of these emulsions can be found in earlier publications.2,13 In short, the concentration of the disperse phase was 90% - 96 % (w/w). This phase in the standard technological recipes is a super-cooled aqueous solution of ammonium nitrate. Water comprises less than 20% by mass of this phase. Thus, the salt concentration (<) exceeds 80%.
The equilibrium temperature for dissolution of such a concentration is approximately 65 °C. For the preparation of these super-cooled solutions, the granulated ammonium nitrate was added to the distillate water at a temperature of 80 °C. The main body of experiments was performed at room temperature, which means that the aqueous phase was a super-cooled (or oversaturated) solution. These solutions are thermodynamically unstable but kinetically they do not change over several weeks, so it is possible to perform different experiments with these materials and treat them as quasi-stable.
The measurement of interfacial tension
Interfacial tension between the oil phase with the surfactant and aqueous phases with ammonium nitrate was measured by means of a Krüss K100 tensiometer (Hamburg, Germany). The principle of operation of the K100 is straightforward. About 15 cm3 ammonium nitrate solution is placed in a clean 70-mm diameter glass dish. A hydrophilic platinum plate is suspended vertically from a sensitive force transducer with its lower edge penetrating the surface of the ammonium nitrate solution. About 50 cm3 of the oil phase is added, so that the plate is completely submerged. The interfacial tension is calculated by the following equation:

where F is the vertical force acting on the plate after a correction has been made for the plate buoyancy (mN), L is the wetted plate length (m) and ө is the contact angle. Because the plate is hydrophilic, the contact angle is assumed to be zero.
Minimum surfactant interfacial concentration, Γ (mol/m2), is given by the following equation, which is derived from the Gibbs adsorption isotherm:

where y is the interfacial tension (mN/m), a is the activity of the surfactant in the bulk, R is the gas constant (8.314 J/mol K) and T is the absolute temperature (K). In the present case, the surfactant activity is not known, and the assumption is made that it is approximately equal to the molar concentration c (mol/L). Thus, the surfactant interfacial concentration can be obtained from a plot of interfacial tension versus the natural logarithm of the surfactant bulk concentration.
The measurement of droplet size
The size of dispersed particles was measured using a Mastersizer-2000 device (Malvern Instruments, Malvern, United Kingdom). The procedure of measuring is based on sample dispersion (which is controlled by software) and the measurement of angle dependence of the intensity of scattering of a collimated helium-neon laser beam. Particle size in the range of 0.26 μm to 1500 μm can be measured; this range is much wider than the sizes of the real samples used in this work. The size distribution calculations are based on the rigorous Mie theory and using the standard software applied to the instrument. Each emulsion sample (a small number of samples was taken) was dispersed in a large volume of oil to reach a very dilute concentration of water droplets in oil and to avoid the formation of agglomerates.
Thermal analysis
DSC studies were carried out using a DSC Q 2000 instrument (Perkin Elmer, Waltham, MA, USA) coupled to a computer for data analysis. Liquid nitrogen was used as a coolant. About 5 mg of sample was taken for each run; the temperature range studied was 30 °C to -70 °C with the cooling rate being 2 K/min.
DSC analysis was used to determine the freezing point as a function of electrolyte concentration and surfactant type concentration. Samples with the admixtures of Ca(NO3)2 and NaNO3 were also investigated. All emulsions included in the study were characterised by the same droplet size, that is, 13 μm.
Results and discussion
Differential scanning calorimetry
Effect of surfactant type and concentration
The following surfactants were used for emulsion stabilisation experiments: PIBSA-MEA, PIBSA-UREA, PIBSA-IMIDE, a mixture of PIBSA-MEA and SMO, and SMO. Crystallisation of emulsions was accelerated by decreasing the temperature. From previous studies3,4,5,14 it is known that a lower crystallisation temperature of emulsion indicates higher emulsion stability. For the surfactant type, the following trend in emulsion crystallisation temperature was observed: PIBSA-MEA=PIBSA-UREA < PIBSA-MEA/SMO > PIBSA-IMIDE < SMO. In other words, PIBSA-MEA crystallised at a higher temperature than did PIBSA-IMIDE. These results are in the same sequence as the results obtained for the stability of these emulsions in aging studies, that is, PIBSA-MEA was the most stable with age and SMO was the least. Therefore we can correlate the crystallisation temperature of the emulsions with the stability of the emulsions before the onset of crystallisation.
The effect of the surfactant type on emulsion crystallisation can probably be attributed to the differing strengths of the surfactant-electrolyte interactions,15 which result in different molecular packing geometry and differing mobility of the surfactant lipophilic portion at the interface.5 It is reasonable to suggest that the difference in chemical structure, type and strength of the interaction between the surfactant and the ammonium nitrate melt plays an important role in crystal initiation.3,16
Indeed, the initiation of crystallisation depends mostly on the ability of additives (ions, molecules or surfactant head groups) to increase the separation distance between NH4+ and NO3- ions.3,16,17 The separation of charges inside the droplet is expected to keep the emulsion in a super-cooled state by reducing the chemical kinetics of oversaturated ammonium nitrate (in the case of PIBSA-MEA and PIBSA-UREA). Moreover, this separation could generate repulsive forces in the thin film between droplets, which could reduce the Van der Waals attraction and consequently improve the stability. On a qualitative level, a strong surfactant-ammonium nitrate interaction is expected to give rise to a more stable emulsion.3
Yubai et al.14 reported that the nucleation of the ammonium nitrate crystals in droplets can be induced by changes in the functionalities of the surfactant head group14 and thereby the ability of the head group to interact with ammonium nitrate ions and water molecules. As a hypothesis, it can be assumed that a non-ionic surfactant (i.e. a surfactant with no charge present in the head group, namely SMO and PIBSA-IMIDE) interacts preferentially with water molecules whilst the charged head group of a surfactant (such as PIBSA-MEA and PIBSA-UREA) is more attractive to the salt ions. The dispersed aqueous droplets in emulsions consisted of a supersaturated aqueous solution of ammonium nitrate salt (at room temperature). The ammonium nitrate concentration was 80% by mass whilst water comprised less than 20% by mass. It is worth noting that this concentration of ammonium nitrate corresponded approximately to a 1:1 molecular ratio of H2O to NH4NO3.14 This ratio implies that such a concentrated solution is very sensitive to the water content and in this case crystallisation could be induced by a reduction of water molecules in the ammonium nitrate melt network. Such a scenario is more likely with the surfactants SMO and PIBSA-IMIDE.
Another important point is that the molecular packing geometry and the mobility of the surfactant lipophilic portion at the interface can affect the crystallisation temperature of an emulsion.5 Whilst comparing the mobilities of different portions of a surfactant molecule present in an emulsion, it is evident that the mobility increases along the hydrocarbon chain - being zero at the head group region and a maximum at the chain end. The polymer chain is generally random18,19 and thus it can be assumed that tail groups of PIBSA-based polymeric surfactants have a higher degree of freedom than that of SMO (a low molecular weight surfactant). This increased mobility could result in a more efficient packing of SMO molecules at the interface, leading to the rapid crystallisation of the liquid interfacial film5 and to emulsion breaking.
The above suggestions imply that the low stability of an emulsion stabilised by a mixture of PIBSA-MEA and SMO, compared to an emulsion stabilised with only PIBSA-MEA, could be as a result of a reduced interaction between PIBSA-MEA and the electrolyte and possibly the dehydration of the ammonium nitrate melt as a result of a greater attraction of SMO to the water molecules. Moreover, the closer packing of surfactant molecules in the monolayer, as a result of the presence of SMO at the interface, may also induce crystallisation.
Emulsion stability to crystallisation improved with an increase in concentration of the surfactant in the oil phase. Surfactant concentration effect was investigated for two surfactants (PIBSA-MEA and PIBSA-IMIDE). PIBSA-MEA showed no improvement in emulsion stability after 14%, which can be regarded as the optimal surfactant concentration for emulsion stabilisation, whilst improvements in emulsion stability were noted for PIBSA-IMIDE for all concentrations studied (8% - 20%).
Effect of calcium and sodium admixture on emulsion stability
In the case of emulsions with both sodium and calcium nitrate salts, the salts lowered the crystallisation point of an ammonium nitrate solution (Table 1). Therefore, including an admixture of sodium and calcium ions, which resulted in a structural change, stabilised the aqueous phase (a solution which contains ammonium nitrate). Indeed, an overall relaxation of the structure of the ammonium nitrate solution was found with the presence of Ca2+ ions. In particular, the nearest-neighbour (N1 - N2) distance (the distance between nitrogen atoms in the nearest NH4+ and NO3- ions) was found to be 5.5 Å in the mixed molten salts compared to 4.5 Å in pure ammonium nitrate.18 This difference in distance provides clear evidence that the presence of doubly charged Ca2+ causes a significant difference in coordination and separation of the ammonium nitrate ions. The same effect can probably be ascribed to the addition of Na+ ions.
It is interesting to note that emulsions stabilised by PIBSA-MEA and PIBSA-UREA surfactants did not show improved stability with the addition of sodium nitrate to the ammonium nitrate. In contrast, a considerable reduction in crystallisation temperature was observed for the emulsions stabilised by PIBSA-IMIDE, PIBSA-MEA/SMO and SMO. It is reasonable to assume that enhanced stability for these emulsions can be explained in terms of extra adsorption of surfactant at the interface.4 To support this assumption, the concentration of PIBSA-IMIDE surfactant at the interface for two concentrations of ammonium nitrate (10% and 60%) was obtained from the slope of the critical micelle concentration measurements (Figure 2).
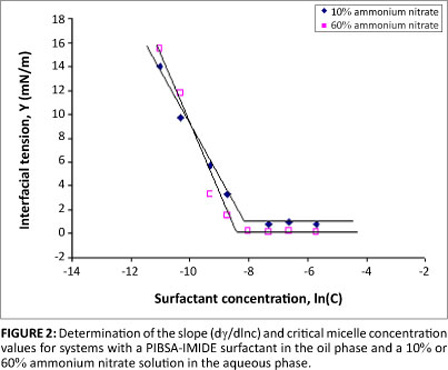
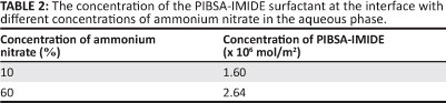
From Table 2 it can be seen that the minimum surfactant interfacial concentration (Γ) for the 10% ammonium nitrate solution is lower than that for the 60% ammonium nitrate in the aqueous phase. This difference implies that more PIBSA-IMIDE surfactant is absorbed at the interface with the 60% ammonium nitrate solution than with the 10% ammonium nitrate solution present in the aqueous phase. Obviously, the PIBSA-IMIDE surfactant is more densely packed at the interface with the 60% ammonium nitrate content in the aqueous phase (as evidenced by the experimental results). Dense packing at the interface can lead to a 'brush' formation (the chains in a brush are stretched out) and therefore an increase in the steric repulsive forces between the surfactant layers that result in a more stable emulsion. The interaction of the head group with the droplets can probably affect the distribution of ions and enhance the emulsion stability.
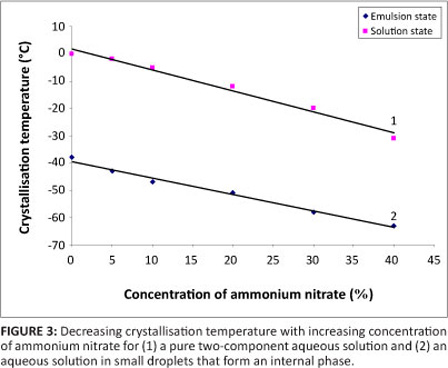
The addition of salts results in a decrease in the freezing temperature of a solution and, thereby, an emulsion - this effect is significant enough that salt solutions in micron-size droplets comprising an internal phase remain stable to a much lower temperature than pure two-component solutions (Figure 3). This physical peculiarity of a two-phase system in micron-size droplets makes it possible to create highly concentrated solutions in the aqueous phase in highly concentrated emulsions.
Conclusion
The surfactant type, concentration and salt composition in the aqueous phase influence the crystallisation temperature of highly concentrated water-in-oil emulsions, in the same order in which aging influences the emulsion stability to crystallisation. The results obtained suggest a link between emulsion stability and the strength of surfactant-electrolyte interactions as well as the packing efficiency of surfactant molecules at the interface. These factors can probably influence the ion distribution in the aqueous phase, which also has an effect on emulsion stability.
Acknowledgements
We thank Lake International Technologies and African Explosives Limited for their financial support, for providing the materials and for permission to publish the results.
Competing interests
We declare that we have no financial or personal relationships which may have inappropriately influenced us in writing this article.
Authors' contributions
K.K. performed the experiments and wrote the manuscript. I.M. was the project leader and was responsible for the project design.
References
1. Masalova I, Malkin AY, Ferg E, Taylor M, Kharatiyan E, Haldenwang R. Evolution of rheological properties of highly concentrated emulsions with aging - Emulsion-to-suspension transition. J Rheol. 2006;50:435-451. http://dx.doi.org/10.1122/L2206712 [ Links ]
2. Masalova I, Malkin AY, Slatter P, Wilson K. The rheological characterization and pipeline flow of high concentration water-in-oil emulsions. J Non-Newton Fluid Mech. 2003;112:101-114. [ Links ]
3. Ganguly S, Mohan VK, Bhasi VCJ, Mathews E, Adiseshaiah KS, Kumar AS. Surfactant-electrolyte interactions in concentrated water-in-oil emulsions: FT-IR spectroscopic and low-temperature differential scanning calorimetric studies. Colloid Surface. 1992;65:243-256. http://dx.doi.org/10.1016/0166-6622(92)80180-A [ Links ]
4. Aronson MP, Petko MF. Highly concentrated water-in-oil emulsions: Influence of electrolyte on their properties and stability. J Colloid Interface Sci. 1993;159:134-149. [ Links ]
5. Villamagna F, Whitehead MA, Chattopadhyay AK. Mobility of surfactants at the water-in-oil emulsion interface. J Dispersion Sci Technol. 1995;16:105-114. http://dx.doi.org/10.1080/01932699508943663 [ Links ]
6. Chattopadhyay AK, Ghaicha L, Oh SG, Shah DO. Salt effects on monolayers and their contribution to surface viscosity. J Phys Chem. 1992;96:6509-6513. http://dx.doi.org/10.1021/j100194a074 [ Links ]
7. Ghaicha L, Leblank RM, Chattopadhyay AK. Influence of concentrated ammonium nitrate solution on monolayers of some dicarboxylic acid derivatives at the air/water interface. Langmuir. 1993;9:288-293. http://dx.doi.org/10.1021/la00025a055 [ Links ]
8. Maheshwari R, Dhathathreyan A. Influence of ammonium nitrate in phase transition of Langmuir and Langmuir-Blodget films at air/solution and solid/solution interfaces. J Colloid Interface Sci. 2004;275:270-276. [ Links ]
9. Clausse D, Gomez F, Pezron I, Komunjer L, Dalmazzone C. Morphology characterization of emulsions by differential scanning calorimetry. Adv Colloid Interface Sci. 2005;117:59-74. http://dx.doi.org/10.1016/j.cis.2005.06.003, PMid:16253203 [ Links ]
10. Coupland JN. Crystallization in emulsions. Curr Opin Colloid Interface Sci. 2002;7:445-450. http://dx.doi.org/10.1016/S1359-0294(02)00080-8 [ Links ]
11. Gosch S, Rosseau D. Freeze-thaw stability of water-in-oil emulsions. J Colloid Interface Sci. 2009;339:91-102. http://dx.doi.org/10.1016/j. jcis.2009.07.047, PMid:19683718 [ Links ]
12. Zhu L, Chen J-Q, Jiu Y, Pan Y, Chang J-Y. Differential scanning calorimetry analysis for water-in-oil emulsions. Abstract. Fresenius Environ Bull. 2011;20:1117-1123. [ Links ]
13. Malkin AY, Masalova I, Slatter P, Wilson K. Effect of droplet size on the rheological properties of highly concentrated w/o emulsions. Rheol Acta. 2004;43:584-591. [ Links ]
14. Yubai B, Munger G, Leblanc RM, Ghaicha L, Chattopadhyay AK. Crystallization of ammonium nitrate under organized monolayers of various amphiphiles. J Dispersion Sci Technol. 1996;17:391-405. http://dx.doi.org/10.1080/01932699608943511 [ Links ]
15. Masalova I, Kovalchuk K, Malkin AY. IR studies of interfacial interaction of the succinic surfactants with different head groups in highly concentrated W/O emulsions. J Dispersion Sci Technol. In press 2011. [ Links ]
16. Adya AK, Neilson GW. Structure of a 50 mol kg-1 aqueous solution of ammonium nitrate at 373 K by the isotopic difference method of neutron diffraction. J Chem Soc Faraday Trans. 1991;87:279-286. http://dx.doi.org/10.1039/ft9918700279 [ Links ]
17. Oxley JC, Kaushik SM, Gilson NS. Thermal stability and compatibility of ammonium nitrate explosives on a small and large scale. Thermochim Acta. 1992;212:77-85. http://dx.doi.org/10.1016/0040-6031(92)80222-I [ Links ]
18. Myers D. Surfaces, interfaces and colloids: Principles and applications. 2nd ed. New York: Wiley VCH; 1999. http://dx.doi.org/10.1002/0471234990 [ Links ]
19. Tadros TF. Applied surfactants: Principles and applications. Weinheim: Wiley VCH; 2005. http://dx.doi.org/10.1002/3527604812 [ Links ]
 Correspondence to:
Correspondence to:
Karina Kovalchuk
Postal address: PO Box 8000
Cape Town 8000, South Africa
Email: karinask@chemeng.ntnu.no
Received: 25 Jan. 2010
Accepted: 22 Aug. 2011
Published: 01 Mar. 2012














