Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.108 n.1-2 Pretoria Jan. 2012
RESEARCH LETTERS
Five Ochna species have high antibacterial activity and more than ten antibacterial compounds
Tshepiso J. Makhafola; Jacobus N. Eloff
Phytomedicine Programme, Department of Paraclinical Sciences, Faculty of Veterinary Sciences, University of Pretoria, Onderstepoort, Pretoria, South Africa
ABSTRACT
New measures to control infections in humans and other animals are continuously being sought because of the increasing resistance of bacteria to antibiotics. In a wide tree screening survey of the antimicrobial activity of extracts of tree leaves (www.up.ac.za/phyto), Ochna pulchra, a small tree found widely in southern Africa, had good antibacterial activity. We therefore investigated the antibacterial activity of acetone leaf extracts of some other available Ochna spp. Antibacterial activity and the number of antibacterial compounds in acetone leaf extracts of Ochna natalitia, Ochna pretoriensis, O. pulchra, Ochna gamostigmata and Ochna serullata were determined with a tetrazolium violet serial microplate dilution assay and bioautography against Staphylococcus aureus, Escherichia coli, Enterococcus faecalis and Pseudomonas aeruginosa, bacteria commonly associated with nosocomial infections. The percentage yields of the extracts varied from 2.5% to 8%. The minimum inhibitory concentrations of the five species ranged from 40 µg/mL to 1250 µg/mL. E. coli was sensitive to all the extracts. The O. pretoriensis extract was the most active with minimum inhibitory concentrations of 0.065 mg/mL and 0.039 mg/mL against E. coli and E. faecalis, respectively. The O. pretoriensis extract also had the highest total activities of 923 mL/g and 1538 mL/g, indicating that the acetone extract from 1 g of dried plant material could be diluted to 923 mL or 1538 mL and would still kill these bacteria. Based on the bioautography results, the two most active species, O. pretoriensis and O. pulchra, contained at least 10 antibacterial compounds with similar Rf values. Some of these antibacterial compounds were polar and others were non-polar. Variation in the chemical composition of the species may have some taxonomic value. The order of activity of the species to the bacteria were O. pretoriensis > O. pulchra > O. gamostigmata > O. serullata > O. natalitia. Even before toxicity and bioavailability issues are considered, some Ochna spp. leaf extracts have the potential to be used in treating skin infections.
Introduction
Medicinal plants which form the backbone of traditional medicines have in the last few decades been subjected to many pharmacological studies. The search for novel antibacterial compounds in plants is relevant because of the increasing resistance of bacteria to synthetic antibiotics and the occurrence of fatal opportunistic infections.1,2
The genus Ochna comprises about 86 species of evergreen shrubs belonging to the family Ochnaceae and occurs widely in Africa, Asia and Madagascar. This family of plants has been used as food and for medicinal purposes for many centuries. Members of this family are characterised by the presence of flavonoids, biflavonoids and terpenoids as main secondary metabolites. Several members of this genus are also used for the extraction of edible oils.3,4
Previous phytochemical studies on some Ochna species found monomeric and dimeric flavonoids in the leaves of Ochna integerrima.5,6 Sibanda et al.7 isolated one anthrone and two vismiones from the root bark of Ochna pulchra. An isobiflavonoid, glycosides and saponins were also isolated from the stem bark of Ochna afzeli.8,9
In a study by Reutrakul et al.9, Ochna integerrima extracts had good antiviral and antiparasitic activities. The study involved the isolation of biflavonoids with antimalarial activity; a flavonoid glycoside with anti HIV-1 activity was also isolated from the same species.9 An infusion of the roots and leaves of O. integerrima is used locally as an antidysenteric and antipyretic preparation.10 There appears to be no previous report on the antibacterial activity of any of the 86 species of Ochna to date.
We have found that plants used traditionally to treat infections have very low antibacterial activity in in vitro assays. The low activity exhibited may be related to the extractant available to traditional healers because aqueous extracts of plants have very low activity.11,12 Consequently, we started investigating the antimicrobial activity of acetone leaf extracts of more than 600 South African trees. We used only acetone for extraction because it is the best extractant for antimicrobial compounds from plants, based on parameters such as efficacy of extraction, diversity of compounds extracted, ease of removal of solvent after extraction, human safety and safety to microorganism in bioassays.11 In these studies, Ochna pretoriensis had good antibacterial activity (Pauw and Eloff, unpublished results). The good activity of O. pretoriensis motivated the present investigation of the antibacterial activity and the number of antibacterial compounds present in other, easily available Ochna species: Ochna natalitia, O. pretoriensis, O. pulchra, Ochna gamostigmata and Ochna serullata.
Materials and methods
Plant collection, drying and storage
Leaves of the five plant species investigated were collected in summer at the National Botanical Gardens in Pretoria, the Lowveld National Botanical Gardens in Nelspruit and the University of Pretoria Botanical Gardens. The origin of each tree is documented in the database of the botanical garden and voucher specimens were deposited in different herbaria (Table 1). Only clean dry leaves without any lesions or dirt were selected. Leaves were not washed with water to limit the possibility of extracting some water-soluble compounds and especially to limit the possibility of fungal growth on moist leaves. The leaves were dried in the dark at room temperature and pulverised into a fine powder (with particles less than 1 mm in diameter) and stored in closed glass bottles in the dark until use.
Extraction
The dry leaf powder (2.0 g) was extracted with 20 mL acetone (technical quality; Merck Pharmaceuticals, Pretoria, South Africa), which is very effective in extracting antimicrobial compounds from plants.11 The resulting suspension was shaken vigorously in 50-mL polyester centrifuge tubes and centrifuged for 15 min at 4000 rpm (Hettich Centrifuge, Rotofix 32A, Labotec, Johannesburg, South Africa). The extracts were decanted into preweighed glass vials through Whatman No. 1 filter papers and concentrated to dryness under a stream of cold air. The dried extracts were made up to a concentration of 10 mg/mL (stock solution) in acetone to be used in subsequent assays and stored at 5 ºC in tightly stoppered glass tubes.
Phytochemical analysis
Samples of 10 µL from the different stock solutions (10 mg/mL) were loaded onto thin layer chromatography (TLC) plates (Merck, Kieselgel 60 F254) and developed in three mobile systems used in the phytomedicine laboratory of the University of Pretoria. The mobile systems used to develop the plates were EMW (ethyl acetate:methanol:water) 40:5.4:4 (v/v/v), CEF (chloroform:ethyl acetate:formic acid) 5:4:1 (v/v/v) and BEA (benzene:ethanol:ammonia hydroxide) 90:10:1 (v/v/v).12
After development, the plates were visualised under UV light and sprayed with vanillin-sulphuric acid reagent spray (0.1 g vanillin in 28 mL methanol and 1 mL sulphuric acid) and heated at 110 ºC until optimum colour development was achieved.
Test organisms
Microbial cultures used in the determination of minimum inhibitory concentrations (MIC) and bioautography were obtained from the Department of Medical Microbiology at the University of Pretoria. The bacterial cultures were maintained on Müller-Hinton agar at 4 ºC and in Müller-Hinton broth at 37 ºC prior to use for the determination of MICs and bioautography. The bacterial strains used in this study were: Enterococcus faecalis (ATCC 29212), Staphylococcus aureus (ATCC 29213), Escherichia coli (ATCC 27853) and Pseudomonas aeruginosa (ATCC 25922). These are the most important nosocomial pathogens in South Africa13 and the specific strains used are recommended for comparing antibiotics.14
Bioautographic assay
Chromatograms were prepared as described above. The plates were then sprayed with actively growing bacterial cultures until wet and incubated at 37 ºC under 100% relative humidity.15 Following overnight incubation, the plates were sprayed with 2 mg/mL tetrazolium violet solution (Sigma, Pretoria, South Africa) and incubated for between 30 min and 60 min; the colour change was monitored to detect growth and inhibition of the test organisms after scanning in the bioautograms. Clear zones against a purple background on the plates indicate inhibition of bacterial growth by antibacterial compounds present in the extracts.16
Minimum inhibitory concentrations
To determine the MIC values of the different plant extracts on the test organisms, the microplate serial dilution assay developed by Eloff17 was used. This experiment was performed in triplicate and repeated twice for verification. The MIC was recorded as the lowest concentration of the plant extract at which the bacterial growth was inhibited. The MIC values were read after both a 12-h incubation and a 36-h incubation with the plant extracts to determine if the activity was bacteriostatic only or also bactericidal. Gentamycin was used as a positive control to confirm the sensitivity of the system. To compare the activity of different plants the total activity in mL/g was calculated by dividing the total mass in mg extracted from 1 g of plant material by the MIC value in mg/mL.18
Results and discussion
Quantity extracted
The percentage yields in acetone from the five species were: O. gamostigmata (8%) followed by O. pulchra (7.5%), O. serullata (7%), O. pretoriensis (6%) and lastly O. natalitia (2.5%). The yields were in the same order as acetone extracts from leaves of different members of the Combretaceae.1
Phytochemical analysis
The intermediate polarity solvent system CEF separated the compounds in all these plant extracts more efficiently than did EMW and BEA (Figure 1). The efficient extraction and separation of phytocompounds is largely dependent on the type of solvent(s) used during extraction, particularly its polarity.15
There were similarities between the TLC chromatograms of O. pretoriensis and O. pulchra extracts; each had 10 similar bands, which is indicative of similar chemical compositions. The chromatograms may be useful in the taxonomy of the plants examined.19 Some of the spots were yellow when viewed in daylight and light blue when viewed under UV light (365 nm). This difference may be as a result of the presence of a strong chromophoric group which is often observed with plant phenolics (especially flavonoids and their glycosides).
Whilst there were some major differences in the TLC fingerprints of the five species, there were also identical bands in similar mobile phases with the same Rf values across a number of species, because of similar biogenetic pathways and metabolite pool sizes in the genus.
Bioautography
TLC-based bioautography is a useful technique that allows the localisation of antimicrobial compounds present in the crude plant extracts.15 The clear spots on the bioautograms indicate the location of the active compounds.16 Bioautography showed that O. pretoriensis and O. pulchra had four major active bands (Figure 1). Because EMW separated compounds that could not be separated by the other solvent systems, there were at least 10 different antibacterial compounds present in the extracts of O. pretoriensis and O. pulchra. CEF was the best overall solvent for the separation of bioactive compounds in this study. There were five prominent active bands present in the O. gamostigmata extract. Three of the active bands had the same Rf value as that of bands characteristic of flavonoids - yellow under daylight and light blue under 365-nm wavelength UV light - which may mean that the same compounds occur in different species. There were also major differences between the chromatograms from different plant species sprayed with vanillin-sulphuric acid. These differences may have some taxonomic significance.19
Minimum inhibitory concentrations
The MIC values of the plant extracts ranged from 0.039 mg/mL to 1.25 mg/mL after a 12-h incubation and from 0.078 mg/mL to 1.25 mg/mL after the 36-h incubation (Table 2A). O. pretoriensis was the most active against E. coli and E. faecalis with MIC values of 0.065 mg/mL and 0.039 mg/mL, respectively. O. pulchra was the most active against P. aeruginosa and S. aureus with MIC values of 0.065 mg/mL and 0.078 mg/mL, respectively. E. coli was sensitive to all the extracts. The order of activity of the species to the tested organisms was O. pretoriensis > O. pulchra > O. gamostigmata > O. serullata > O. natalitia.
Different patterns of sensitivity of the test organisms to the plant extracts were noted after 12 h and 36 h (Table 2), which indicates that some of the bioactive compounds in the extracts are bacteriostatic and some are bactericidal. The MIC of O. gamostigmata was 0.31 mg/mL at 12 h against both E. coli and E. faecalis; the MIC remained the same after 36 h, indicating that the bioactive compounds had bactericidal activity. In contrast, the MIC of O. pulchra against P. aeruginosa was 0.063 mg/mL after 12 h, but rose to 0.1 mg/mL at 36 h, which indicates that the activity may be partly bacteriostatic.
Total activity and reciprocals of the MICs were calculated for all extracts (Table 2B and Figure 2). The total activity is important because it shows the quantity and quality of the active compounds present in the plant extracts.20 Total activity is also dependent on the quantity of material extracted from the dried plant leaves; the higher the total activity, the greater is the potential for application of the specific plant extract.18
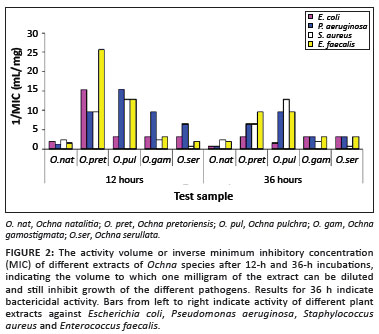
O. pretoriensis, O. pulchra and O. gamostigmata had high activity with the total activity of O. pretoriensis being 1538 mL/g (against E. faecalis), O. pulchra 1153 mL/g (against P. aeruginosa) and O. gamostigmata 769 mL/g (against P. aeruginosa).
Conclusions
Three of the five Ochna species investigated - O. pretoriensis, O. gamostigmata and O. pulchra - had significant antibacterial activity based on their MIC values and bioautography. The bioactive compounds in two of the most active species, O. pretoriensis and O. pulchra, were similar. O. pretoriensis had the highest total activity of 1538 mL/g against E. faecalis and 923 mL/g against E. coli. Publications on other species in the Ochna genus have reported the isolation of flavonoids and biflavonoids with antimalarial and anti-HIV activities. We report for the first time, the antibacterial activities, the number of antibacterial compounds present and the Rf values of antibacterial compounds of some Ochna species. Because of their high total activity it appears that acetone leaf extracts of several Ochna species could be valuable in treating infections caused by these nosocomial pathogens. O. pulchra is notoriously difficult to cultivate and would not be a good candidate for the development of a herbal medicine. O. pretoriensis is easier to cultivate and therefore is a better candidate for an in-depth investigation. We also have isolated and characterised several of the compounds responsible for the antibacterial activity of O. pretoriensis; these results will be published elsewhere.
Acknowledgements
The National Research Foundation of South Africa and the University of Pretoria provided financial support. The curators of the Pretoria National Botanical Gardens, the Lowveld National Botanical Gardens in Nelspruit and the Kirstenbosch National Botanical Garden allowed us to collect plant material.
Competing interests
We declare that we have no financial or personal relationships or competing interests which may have inappropriately influenced us in writing this article.
Authors' contributions
T.J.M. performed the research and wrote the first draft of the manuscript. J.N.E. identified the project, guided the research, and revised and submitted the manuscript.
References
1. Eloff JN. The antibacterial activity of 27 southern African members of the Combretaceae. S Afr J Sci. 1999;95:148-152. [ Links ]
2. Newman DJ, Cragg GM, Snader KM. Natural products as sources of new drugs over the period 1981-2002. J Nat Prod. 2003;66:1022-1037. http://dx.doi.org/10.1021/np030096l, PMid:12880330 [ Links ]
3. Oliveira MCC, Carvalho MG, Silva CJ, Werle AA. New biflavonoid and other constituents from Luxemburgia nobilis EICHL. J Brazil Chem Soc. 2002;13:119-123. http://dx.doi.org/10.1590/S0103-50532002000100020 [ Links ]
4. Estevam CS, Oliveira FM, Conserva LM, et al. [Chemical constituents and antimalarial activity of Ouratea nitida Aubl. (Ochnaceae)]. Rev Bras Farmacogn. 2005;95-198. Portuguese. [ Links ]
5. Kaewamatawong R, Likhitwitayawuid K, Ruangrungsi N, Takayama H, Kitajima M, Aimi N. Novel biflavonoids from the stem bark of Ochna integerrima. J Nat Prod. 2002;65:1027-1029. http://dx.doi.org/10.1021/np010630u, PMid:12141866 [ Links ]
6. Likhitwitayawuid K, Kaewamatawong R, Ruangrungsi N. Mono- and biflavonoids of Ochna integerrima. Biochem Syst Ecol. 2005;33:527-536. http://dx.doi.org/10.1016/j.bse.2004.10.014 [ Links ]
7. Sibanda S, Nyayira M, Nicolletti M, Galleffi C. Vismiones L and M from Ochna pulchra. Phytochemistry. 1993;34(6):1650-1652. http://dx.doi.org/10.1016/S0031-9422(00)90866-5 [ Links ]
8. Messanga BB, Tih RG, Kimbu SF, Sondengam BL, Martin MT, Bodo B. Calodenone, a new isobiflavonoid from Ochna calodendron. J Nat Prod. 1992;55:245-248. [ Links ]
9. Reutrakul V, Ningnuek N, Pohmakotr M, et al. Anti HIV-1 flavonoid glycosides from Ochna integerrima. Planta Medica. 2007;73(7):683-688. http://dx.doi.org/10.1055/s-2007-981538, PMid:17562490 [ Links ]
10. Perry LM. Medicinal plants of East and Southern Asia. Cambridge, MA: Massachusetts Institute of Technology; 1980. [ Links ]
11. Eloff JN. Which extractant should be used for the screening and isolation of antimicrobial components from plants? J Ethnopharmacol. 1998;60:1-8. [ Links ]
12. Kotze M, Eloff JN. Extraction of antibacterial compounds from Combretum microphyllum (Combretaceae). S Afr J Bot. 2002;68:62-67. [ Links ]
13. Sacho H, Schoub DB. Current perspectives on nosocomial infections. Pietermaritzburg: Natal Witness Printing and Publishing; 1993. [ Links ]
14. National Committee for Clinical Laboratory Standards (NCCLS). Performance standards for antimicrobial disk susceptibility tests. 4th ed. Approved Standard. NCCLS Document M2-A4. Villanova, PA: NCCLS; 1990. [ Links ]
15. Masoko P, Eloff JN. The diversity of antifungal compounds in six South African Terminalia species (Combretaceae) determined by bioautography. Afr J Biotechnol. 2005;14(12):1425-1431. [ Links ]
16. Begue WJ, Kline RM. The use of tetrazolium salts in bioautographic procedures. J Chromatogr. 1972;64:182-184. [ Links ]
17. Eloff JN. A sensitive and quick method to determine the minimum inhibitory concentration of plant extracts for bacteria. Planta Medica. 1998;60:1-8. [ Links ]
18. Eloff JN. A proposal on expressing the antibacterial activity of plant extracts - A small first step in applying scientific knowledge to rural primary health care in South Africa. S Afr J Sci. 2000;96:116-118. [ Links ]
19. Eloff JN. [The large difference in chemical composition and antibacterial activity between two related Leonotis species (Lamiaceaea) may have taxonomic value]. S Afr Tydskr Natuurwet Tegnol. 2010;29:30-38. Afrikaans. [ Links ]
20. Masoko P, Picard J, Eloff JN. Antifungal activities of six South African Terminalia species (Combretaceae). J Ethnopharmacol. 2005;99:301-306. http://dx.doi.org/10.1016/j.jep.2005.01.061, PMid:15894142 [ Links ]
 Correspondence to:
Correspondence to:
Jacobus Eloff
Postal address: Private Bag X04
Onderstepoort 0110, South Africa
Email: kobus.eloff@up.ac.za
Received: 04 Apr. 2011
Accepted: 11 Aug. 2011
Published: 23 Jan. 2012
© 2012. The Authors. Licensee: AOSIS OpenJournals. This work is licensed under the Creative Commons Attribution License.














