Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.107 n.3-4 Pretoria Mar./Apr. 2011
http://dx.doi.org/10.4102/sajs.v107i3/4.366
RESEARCH ARTICLE
Antibacterial and anticandidal activity of Tylosema esculentum (marama) extracts
Walter ChingwaruI; Gyebi DuoduII; Yolandi van ZylII; C.J. SchoemanII; Runner T. MajindaIII; Sam O. YeboahIII; Jose JacksonIV; Petrina T. KapewangoloV; Martha Kandawa-SchulzV; Amanda MinnaarII; Avrelija CencicI,VI
IDepartment of Microbiology, Biochemistry, Molecular Biology and Biotechnology, University of Maribor, Maribor, Slovenia
IIDepartment of Food Science, University of Pretoria, Pretoria, South Africa
IIIDepartment of Chemistry, University of Botswana, Gaborone, Botswana
IVResearch and Development Office, University of Botswana, Gaborone, Botswana
VDepartment of Chemistry and Biochemistry, University of Namibia, Windhoek, Namibia
VIFaculty of Medicine, University of Maribor, Maribor, Slovenia.
ABSTRACT
Bean and tuber extracts of Tylosema esculentum (marama) - an African creeping plant - were obtained using ethanol, methanol and water. Based on information that T. esculentum is used traditionally for the treatment of various diseases, the antibacterial and anticandidal effects of tuber and bean extracts were investigated. The antimicrobial activity of the extracts was tested on methicillin-resistant Staphylococcus aureus (MRSA, ATCC 6538), Mycobacterium terrae (ATCC 15755), Corynebacterium diphtheriae (clinical) and Candida albicans (ATCC 2091). We performed the broth microdilution test for the determination of the minimum inhibitory concentration (MIC) and a method to determine survival of microorganisms after in vitro co-incubation with the highest concentrations of T. esculentum extracts, followed by assessment of colony counts. Ethanol and methanol (phenolic) bean extracts exhibited higher potency against bacteria and yeast than aqueous extracts. Marama bean seed coat crude ethanolic extract (MSCE) and seed coat polyphenolic fractions, especially soluble-bound fraction (MSCIB), were highly antimicrobial against M. terrae, C. diphtheriae and C. albicans. All marama bean polyphenolic fractions, namely cotyledon acidified methanol fraction (MCAM), seed coat acidified methanol fraction (MSCAM), cotyledon insoluble-bound fraction (MCIB), seed coat insoluble-bound fraction (MSCIB), cotyledon-free polyphenolic fraction (MCFP) and seed coat free polyphenolic fraction (MSCFP) had high antimicrobial effects as shown by low respective MIC values between 0.1 mg/mL and 1 mg/mL. These MIC values were comparable to those of control antimicrobials used: amphotericin B (0.5 mg/mL) and cesfulodin (0.1 mg/mL) against C. diphtheriae, streptomycin (1.0 mg/mL) and gentamicin (0.4 mg/mL) against M. terrae, and amphotericin B (0.05 mg/mL) against C. albicans. Marama seed coat soluble-esterified fraction (MSCS) had closer activity to that of cefsulodin against M. terrae. High amounts of phenolic substances, such as gallic acid, especially in the seed coats, as well as high amounts of phytosterols, lignans, certain fatty acids and peptides (specifically protease inhibitors) in the cotyledons contributed to the observed antibacterial and anticandidal activities. Marama extracts, especially phenolic and crude seed coat extracts, had high multi-species antibacterial and anticandidal activities at concentrations comparable to that of some conventional drugs; these extracts have potential use as microbicides.
Introduction
Tylosema esculentum (TE) (Burch) (marama) A. Schreib (Family: Caesalpiniaceae or Leguminosae)1is a creeping plant found in the southern parts of Africa, namely South Africa, Namibia and Botswana. T. esculentum is a perennial plant bearing a large tuber underground and beans on the creeping part of the plant. The chemical composition of the marama plant has recently been explored, mostly within the EU FP6 INCO Marama II research project. Our work is however the first to report on the health benefits of the marama plant.
Plants are widely used in traditional herbal medicine against various diseases.2,3,4,5,6 T. esculentum plant parts, mainly the beans and tubers, are widely used as a food source and as a traditional medicine against various infections and for health maintenance by the San people of southern Africa.7
Marama beans are known to be rich in proteins (29% - 39%)8,9,10,11 and oil (24% - 42%).8,9,10,11,12,13,14,15 The protein content of marama beans is comparable to, or higher than, that of most other legume seeds, namely dry peas, chick peas, lentils, kidney beans, cowpea and lupine - each containing between 20% and 40% dry matter (dm),16,17 and equal to that of soybeans (33% - 46% dm).16,18,19 The high content of protein in marama beans allows great potential for use both as a nutritive food and as a source of food supplements. Marama protein composition is dominated by tyrosine (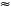 11%), aspartic acid (
11%), aspartic acid (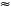 10%) and glutamic acid (
10%) and glutamic acid (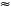 15%).9,20 The oil content of dry marama seeds ranges from 36% to 43%, which approximates that of peanuts.15 The oils of marama beans are reported to contain 31% unsaturated fatty acids, in particular oleic acid and linoleic acid (Ripperger-Suhler and Longenecker21 cited in Biesele and Murray22).
15%).9,20 The oil content of dry marama seeds ranges from 36% to 43%, which approximates that of peanuts.15 The oils of marama beans are reported to contain 31% unsaturated fatty acids, in particular oleic acid and linoleic acid (Ripperger-Suhler and Longenecker21 cited in Biesele and Murray22).
Based on empirical data on other legumes, mostly soybeans, it was expected that the marama plant would contain large amounts of phenolic substances with antibacterial and anticandidal effects. High amounts of phytosterols have also been detected in marama oil (about 75% of all phytosterols being 4-desmethylsterols and about 16% of the total being 4,4-dimethylsterols).14 In addition, marama beans are known to be an excellent source of lignans.11
Marama beans are also rich in minerals such as potassium (7.57 mg/g - 13.16 mg/g), phosphorus (3.34 mg/g - 5.54 mg/g), magnesium (2.48 mg/g - 3.74 mg/g), sulphur (1.91 mg/g - 2.36 mg/g) and calcium (1.19 mg/g - 1.33 mg/g).8,11 While some minerals, especially trace elements, contribute to toxicity, their presence in certain plant extracts has been shown to contribute to antimicrobial effects. Parlatan et al.23 have shown that minerals present in the extracts of Laser trilobum fruits, particularly sodium (9.26 mg/g) but also potassium, calcium, phosphorus, magnesium, arsenic and aluminium, resulted in the observed high antimicrobial activity, and not the essential oils dominating the fruit extracts, mainly limonene (41.03% - 72.24%) and perillaldehyde (4.23% - 32.75%).
Marama beans contain higher amounts of secoisolariciresinol (305 µg/100 g - 406 µg/100 g) than soybeans (13 µg/100 g - 273 µg/100 g) and peanuts (333 µg/100 g).24 Marama beans also contain higher levels of lariciresinol (614 µg/100 g - 825 µg/100 g)14 than soybeans (287 µg/100 g).25 However the pinoresinol content of marama beans (21 µg/100 g - 23 µg/100 g) is lower than that of soybeans (446 µg/100 g).25 Complementing this high lignan content is the fact that the levels of secoisolariciresinol and lariciresinol are retained after roasting of marama beans, which is an important characteristic because the beans usually are roasted before they are eaten.11 In contrast to soybeans, which are rich in isoflavones,26 no isoflavones have been detected in marama bean samples.11
Based on the established chemical composition of T. esculentum beans and tuber, and information available on traditional uses of the plant, we sought to establish the antimicrobial effects of T. esculentum extracts. The microorganisms selected for determination of antimicrobial effects were: Candida albicans, methicillin-resistant Staphylococcus aureus (MRSA), Mycobacterium terrae and Corynebacterium diphtheria; all pose potential threats to human health.
We hypothesised that T. esculentum beans and tubers have antimicrobial effects, in this case both antibacterial and anticandidal effects. We expected that the observed positive health effects in traditional African medicine were as a result of the combined effects of the components in T. esculentum. No research on the antibacterial and anticandidal effects of T. esculentum extracts has been performed prior to this work. As an extensive chemical characterisation of T. esculentum has been done, we sought to determine only the biological effects of T. esculentum extracts.
Materials and methods
Plant collection
All research on the plant was done with full authorisation granted to the Marama II INCO (EU-FP6) research programme by the Botswanian, Namibian and South African governments. A specimen of T. esculentum is kept in the Namibian National Herbarium (collecting number GM 1063 and herbarium number 59520). Mature and naturally dried (brown) T. esculentum beans were collected during the winter seasons (April-June) of 2007 from Botswana and Namibia, and 20 kg of tuber was obtained from Jwaneng, Botswana. The air-dried beans were stored in plastic bags at room temperature in the dark. Tylosema esculentum tuber (approximately 20 kg wet mass) was ground and air-dried at the University of Botswana, then further ground to a fine powder before the extraction was done at the University of Maribor, Slovenia.
Preparation of crude T. esculentum bean and tuber extracts
Aqueous extraction of the T. esculentum bean and tuber was done using modified standard methods as previously reported.27,28 A cold-water extract of T. esculentum was prepared from marama bean seed coats (MSCW, marama seed coat water extract), bean cotyledons (MCW, marama cotyledon water extract) and tubers (MTW, marama tubers water extract). Briefly, dried beans were de-hulled, and then separated into seed coats and cotyledons. Dried tuber was ground and powdered. Powdered samples (20 g) were mixed individually in 200 mL distilled water and incubated at room temperature for 16 h. The mixture was then blended and centrifuged at 4000 rpm (2050 g) for 30 min at 4 ºC using a Sorval Evolution RC Superspeed centrifuge (Thermo, USA). The supernatant was filtered using a 0.45-µm pore filter (Millipore, Carrigtwohill, County Cork, Ireland) and stored at -20 ºC.
Crude bean ethanolic extracts were prepared as previously described.29 Briefly, aliquots (100 g) of dry powdered bean cotyledons and seed coats were soaked in 70% ethanol (plus 1% hexane chloroform to facilitate extraction of non-polar components) until complete exhaustion of the material. The solvents were evaporated using a rotary evaporator (Labortechnik Ag, Ch-9230, Flawil, Switzerland). Residual water after evaporation of ethanol was removed by freeze-drying. Dried extracts were stored in labelled sterile screw-capped bottles at -20 ºC. The resultant extracts, composed mainly of lipids and other ethanol soluble compounds such as phenolic substances,30 were labelled as MSCE (seed coat ethanolic extract) and MCE (cotyledon ethanolic extract), and then stored at -20 ºC until further use. The yields of each extract of T. esculentum used are shown in Table 1.

Preparation of T. esculentum bean fractions
Preparation of free, soluble-esterified and insoluble-bound polyphenolic fractions
Free, soluble-esterified and insoluble-bound polyphenolic fractions were prepared from T. esculentum bean seed coat and cotyledon samples using the methods previously described31,32 with slight modifications. Tylosema esculentum bean seed coat and cotyledon samples (4 g each) were extracted with 30 mL of 80% aqueous methanol for 2 h using a magnetic stirrer. These were filtered using Whatman No. 1 filter paper (Whatman GmbH, Dassel, Germany).
Free polyphenolic fractions
The methanol in the filtrate was evaporated under vacuum at 40 ºC. The remaining aqueous solution was acidified to pH = 2 using 6 M hydrochloric acid and extracted three times, each with 30 mL ethyl acetate:diethyl ether (1:1). The organic phase was dried with anhydrous sodium sulphate, filtered, evaporated to dryness under vacuum, re-dissolved in 25 mL 80% aqueous methanol and acidified with 1 mL 2% aqueous acetic acid. These were labelled the free polyphenolic fractions: MCFP (marama cotyledon free polyphenolic) and MSCFP (marama seed coat free polyphenolic).
Soluble-bound polyphenolic fractions
The aqueous phases from the free polyphenolic fractions above were hydrolysed with 2 M sodium hydroxide by stirring for 2 h, acidified to pH = 2 with 6 M hydrochloric acid and extracted with ethyl acetate-diethyl ether as described above. The organic phase was dried with anhydrous sodium sulphate, filtered, evaporated to dryness under vacuum, re-dissolved in 25 mL 80% aqueous methanol and acidified with 1 mL 2% aqueous acetic acid. These were labelled the soluble-esterified polyphenolic fractions: MCS (marama cotyledon soluble-esterified) and MSCS (marama seed coat soluble-esterified).
Insoluble-bound polyphenolic fractions
The residues from the initial extractions with aqueous methanol were treated with 2 M sodium hydroxide by stirring for 2 h, acidified to pH = 2 with 6 M hydrochloric acid and extracted with ethyl acetate-diethyl ether as described above. The organic phase was dried with anhydrous sodium sulphate, filtered, evaporated to dryness under vacuum, re-dissolved in 25 mL 80% aqueous methanol and acidified with 1 mL 2% aqueous acetic acid. These were labelled the insoluble-bound polyphenolic fractions: MCIB (marama cotyledon insoluble-bound) and MSCIB (marama seed coat insoluble-bound).
Acidification of the extracts was done to facilitate good solubility and separation of the phenolic acids and flavonoids during high-performance liquid chromatography (HPLC) analysis. Furthermore, the eluting solvents used for HPLC were also acidic (0.1% acetic acid in water for solution A and 0.1% acetic acid in acetonitrile for solution B).
Preparation of acidified methanol polyphenolic fractions
The T. esculentum bean seed coat and cotyledon samples were extracted using acidified methanol (1% hydrochloric acid in methanol) as follows: 0.6 g of the seed coat was extracted with 60 mL acidified methanol and 0.6 g of the cotyledon was extracted with 40 mL acidified methanol using a magnetic stirrer for 2 h. These were then filtered with Whatman No. 1 filter paper and the filtrates were recovered as the acidified methanol extracts.
The following fractions were thus obtained: MCAM (marama cotyledon acidified methanol) and MSCAM (marama seed coat acidified methanol). The yields of T. esculentum polyphenolic fractions obtained are shown in Table 1. The polyphenolic fractions were dissolved separately in 10 mL of 80% methanol and stored.
Determination of total phenolic content
The total phenolic content of the extracts was determined by the Folin-Ciocalteu assay. The methanolic extract (0.25 mL), was placed in a 25-mL volumetric flask and 5 mL distilled water was added. Folin-Ciocalteu's phenol reagent (1.25 mL) was added and mixed. After 2 min, 3.75 mL 20% (w/v) sodium carbonate solution was added and the contents were mixed. Distilled water was added to volume, the contents mixed again and incubated for 2 h. The absorption was then measured at 760 nm using a Lambda EZ150 spectrophotometer (PerkinElmer Corporation, USA). The standard used was catechin and the results are expressed as mg catechin equivalents per 100 g sample on a dry basis.
HPLC analysis of phenolic compounds
The types and concentration of phenolic compounds of the samples were determined using reverse-phased HPLC. All the solvents and chemicals used during this assay were of HPLC grade. The sample extracts were filtered through a 0.2-µm PTFE syringe filter prior to HPLC injection. The HPLC system consisted of a Waters 1525 binary HPLC pump and a Waters 2487 dual wavelength absorbance detector (Waters Corporation, Milford, MA, USA). The separation was accomplished by means of a YMC-Pack ODS AM-303 (250 mm x 4.6 mm in diameter, 5 µm particle size) column. BreezeTM software33 was used to monitor the separation process. All samples were injected in volumes of 20 µL, with the analysis conducted at a flow rate of 0.8 mL/min and monitored at 280 nm. The mobile phase consisted of 0.1% glacial acetic acid in distilled water (solvent A) and 0.1% glacial acetic acid in acetonitrile (solvent B). A linear gradient was applied as follows: solvent B was increased from 8% to 10% after 2 min, to 30% after 25 min, to 90% after 23 min and to 100% after 2 min, kept at 100% for 4 min, and returned to the initial condition. Running time was 60 min and the column temperature was held at 25 ºC during the run. Pure standards of 20 phenolic compounds were purchased from Sigma Aldrich (USA). The phenolic acids were ferulic acid, p-coumaric acid, syringic acid, vanillic acid, protocatechuic acid, 4-hydroxybenzoic acid, caffeic acid, sinapic acid and gallic acid. The flavonoids were rutin, naringin, naringenin, myricetin, quercetin, catechin, hesperidin, hesperitin and kaempferol. The phenolic standards were prepared in dimethyl sulphoxide at concentrations of 200 ppm, 150 ppm, 100 ppm, 50 ppm, 25 ppm, 20 ppm, 10 ppm and 5 ppm (mg/L). Standards of 20 µL aliquots were chromatographed singly and as mixtures by injection into the HPLC system. Standard calibration curves were obtained for each phenolic compound by plotting peak areas versus concentrations. Regression equations that showed a high degree of linearity (R2 > 0.995) were obtained for each phenolic compound from the calibration curves. Phenolic compounds in the samples were identified by comparing the retention time of the unknown with those of the standard phenolic compounds. The concentrations of the identified phenolic compounds were calculated using the regression equations obtained and expressed as mg/100 g of sample (dry basis).
Microorganisms
The microorganisms used in this study were as follows: methicillin-resistant S. aureus (MRSA, ATCC 6538), C. diphtheriae (clinical isolate provided by Dr B Filipič, University of Ljubljana, Slovenia), M. terrae (ATCC 15755) and C. albicans (ATCC 2091). The microorganisms were maintained as frozen cultures as follows: MRSA and C. diphtheriae in nutrient broth (Merck, Darmstadt, Germany), and C. albicans in yeast extract peptone glucose (YEPG) broth (reconstituted media), both supplemented with 20% glycerol. To prepare the inoculums for both broth tests, the bacterial strains were grown on nutrient agar and C. albicans on YEPG agar; C. diphtheriae, M. terrae and C. albicans were incubated for 24 h at 37 ºC until visible colonies appeared. These bacterial and yeast colonies were then inoculated in 10 mL of nutrient broth or YEPG broth, respectively. The inoculated broths were incubated at 37 ºC until turbidity achieved spectrophotometric readings of 0.1 and 0.3 at an optical density of 620 nm, as measured spectrophotometrically (Labsystems Multiskan MS, Helsinki, Finland). The bacterial and yeast cultures were then diluted to final concentrations of 1 x 105 CFU/mL and 1 x 103 CFU/mL, respectively, using pre-determined equations. Before use in the tests, the broth cultures were shaken on a Vortex mixer.
Screening of antibacterial and anticandidal activities of T. esculentum extracts
The antimicrobial activities of the extracts were determined by the plate count and broth dilution methods. Briefly, C. albicans was grown in YEPG while MRSA, M. terrae and C. diphtheriae were grown in nutrient broth at 37 ºC for 24 h. The solid ethanolic bean extracts were dissolved in nutrient broth or YEPG to 1 x 10-2; the aqueous and total polyphenolic fractions were also diluted to 1 x 10-2 and filtered through a 0.45-µm membrane filter. High concentrations of each extract were used in this experiment to simulate the effect of consuming marama beans (assuming minimal dilution in the gut), as occurs when used in traditional medicine. A traditional practitioner using marama extracts to treat various diseases in Botswana diluted extracts minimally (similarly to the concentrations tested). Individual microorganisms, at concentrations of 1 x 105 bacterial cells/mL or 1 x 103 yeast cells/mL, were inoculated into liquid media (nutrient broth or YEPG, respectively) containing a twofold dilution of each extract as indicated above. After co-incubation of microorganisms and extracts for 3 h, aliquots of extract-microorganism cultures were introduced onto the nutrient agar (bacteria) or YEPG agar (yeast), and incubated at 37 ºC for 24 h. At each sampling time, microbial culture samples were spot plated on solid agar as previously described.34 Briefly, 10 µL of 10-fold diluted bacterial or yeast suspension was spotted on agar as follows: MRSA, M. terrae and C. diphtheriae on nutrient agar and C. albicans on YEPG agar. The spots were allowed to dry, and then plates were incubated at 37 ºC for 24 h for bacteria and 48 h for C. albicans, as previously described.35 Each test was done in triplicate. Counts of viable microbial cells were taken as determined by the ability to grow into colonies. The results are expressed as the percentage of reduced counts as compared to the control (without plant extracts added).
Broth microdilution tests were performed according to the Clinical and Laboratory Standards Institute (formerly NCCLS)document M27-A.36 This method was included to determine the minimum inhibitory concentration (MIC) of the different extracts/fractions. A method modified from that of Swenson et al.37 was adopted. A working suspension of the inoculum was made by a 1:100 dilution of 0.5 McFarland standard bacterial/yeast suspension (1 x 106yeasts or 1 x 108 bacterial cells) (modified) in 0.85% saline followed by a 1:20 dilution in nutrient broth. Twofold serial broth dilutions of each extract were made, then 100 µl of liquid suspension of the bacteria or yeast were added to each well to achieve the final standard inocula of colony forming units (CFU) of bacteria (1 x 105CFU/mL) or yeast (1 x 103CFU/mL) in 96-well plates in triplicate. As a control, 10-fold dilutions of antibacterial or anticandidal agents were prepared as follows: 50 mg/mL to 5 x 10-7 mg/mL amphotericin B (Fungizone, Bristol-Myers-Squibb, Abbot, France); 10 mg/mL to 1 x 10-6 mg/mL streptomycin sulphate (Sigma, Missouri, USA); 10 mg/mL to 1 x 10-6 mg/mL cefsulodin sodium salt hydrate (Sigma, Missouri, USA); and 4 mg/mL to 4 x 10-7 mg/mL gentamicin (Lek, Ljubljana, Slovenia). The control agents were then mixed with the prepared working suspensions of the inocula. The antibiotic-microorganism mixtures were then introduced onto 96-well plates (TPP, Trasadingen, Switzerland), 100 µl per well, in triplicate. The plates were incubated at 37 ºC for 24 h. The MIC was taken as the lowest concentration of the extract that completely inhibited microbial growth. Complete inhibition was recorded if no more than one well exhibited turbidity. Turbidity was determined by visual observation.
Antibacterial/anticandidal data analysis and interpretation of results
Microbial counts are presented as mean colony forming units per millilitre (CFU/mL) and standard deviations of triplicate counts. MICs are presented in mg/mL.
Antimicrobial data from broth microdilution and colony count methods were considered together during interpretation. The MIC values of the extracts and test antibiotics were compared to those for cranberry and those given in the literature.
Paired t-tests, comparing mean MIC values of extracts or fractions were carried out using STATISTICA®,38 yielding t-values. The magnitude of the t-value was assessed to determine the level of significance between treatments.
It has been shown that the MIC values of amphotericin B for Candida species generally are clustered between 0.25 µg/mL and 1.0 µg/mL. It should however be emphasised that the M27 method does not consistently permit detection of resistant strains and therefore isolates with MIC values greater than 1.0 µg/mL should be considered likely to be resistant. The interpretive category of gentamicin resistance for MRSA was that recommended by the Clinical and Laboratory Standards Institute, and the cut-off MIC was 8 x 10-4 mg/mL.39 The Clinical and Laboratory Standards Institute, however, does not define breakpoints for Corynebacterium spp.39 Penicillin G and erythromycin are the first choice of treatment in patients with diphtheria and close contacts retain their activity against C. diphtheriae. Currently no antibiotic breakpoints for C. diphtheriae are available,40 and therefore no meaningful comparisons can be made.
Results
Composition of T. esculentum beans and tubers
Tylosema esculentum seed coats contained significantly higher levels (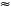 500 mg/100 g sample) of total phenolic substances than did the cotyledons (
500 mg/100 g sample) of total phenolic substances than did the cotyledons ( 100 mg/100 g sample) (Table 2, Table 3 and Table 4). The seed coat sample had 330% more combined total phenolic substances than the cotyledon one. For both seed coat and cotyledon, the total phenolic substances in the insoluble-bound fraction were higher than in either the free or soluble-esterified fractions. Overall, and also for the cotyledon sample, the insoluble-bound fraction had higher total phenolic content than the free and soluble-esterified fractions (Table 2). Tylosema esculentum seed coats also contained significantly higher amounts of free, soluble-esterified and insoluble-bound polyphenolic fractions than did the cotyledons, especially catechin, rutin, naringin, hesperidin and myricetin; while cotyledons contained significantly higher amounts of naringenin and kaempferol than did the seed coats (free, soluble-esterified and insoluble-bound polyphenolic fractions) (Table 3 and Table 4). Quercetin was shown to be equally distributed between cotyledons and seed coats (Table 3 and Table 4).
100 mg/100 g sample) (Table 2, Table 3 and Table 4). The seed coat sample had 330% more combined total phenolic substances than the cotyledon one. For both seed coat and cotyledon, the total phenolic substances in the insoluble-bound fraction were higher than in either the free or soluble-esterified fractions. Overall, and also for the cotyledon sample, the insoluble-bound fraction had higher total phenolic content than the free and soluble-esterified fractions (Table 2). Tylosema esculentum seed coats also contained significantly higher amounts of free, soluble-esterified and insoluble-bound polyphenolic fractions than did the cotyledons, especially catechin, rutin, naringin, hesperidin and myricetin; while cotyledons contained significantly higher amounts of naringenin and kaempferol than did the seed coats (free, soluble-esterified and insoluble-bound polyphenolic fractions) (Table 3 and Table 4). Quercetin was shown to be equally distributed between cotyledons and seed coats (Table 3 and Table 4).
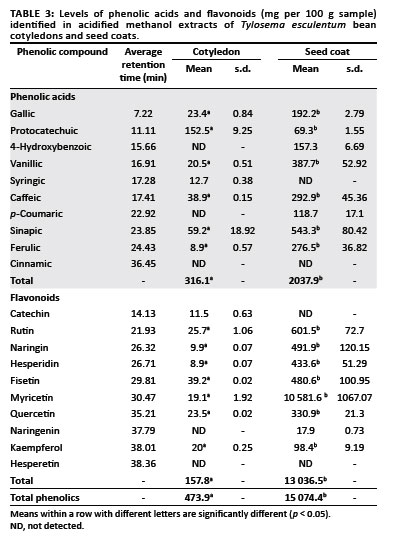
The phenolic acids and flavonoids identified in the T. esculentum bean cotyledon and seed coat acidified methanol fractions and their concentrations are shown in Table 3. There were significant differences (p < 0.05) in the concentrations of all the phenolic compounds detected between the cotyledon and seed coat samples. The total phenolic acid content in the seed coat (2037.9 mg/100 g) was significantly higher than that of the cotyledon (316.1 mg/100 g). The total flavonoid content of the seed coat (13 036.5 mg/100 g) was also significantly higher than that of the cotyledon (157.8 mg/100 g). On the whole, flavonoids constituted the major class of phenolic substances in the T. esculentum bean.
For the phenolic acids, protocatechuic acid (152.5 mg/100 g) and sinapic acid (543.3 mg/100 g) were present in highest concentrations in the cotyledon and seed coat, respectively. The seed coat contained higher amounts of each individual phenolic acid detected than the cotyledon, except for syringic acid (which has not been detected in the seed coat) and protocatechuic acid (which was higher in the cotyledon). For the flavonoids, fisetin (39.2 mg/100 g) and myricetin (10 581.6 mg/100 g) were present in highest concentrations in the cotyledon and seed coat, respectively. The seed coat contained higher amounts of each individual flavonoid detected than the cotyledon, except for catechin (which was not detected in the seed coat) and hesperetin (which was not detected in either the seed coat or the cotyledon).
Overall, the seed coat had 13, 1.6 and 3.4 times the amount of benzoic acid derivatives, cinnamic acid derivatives and flavonoids, respectively, than the cotyledon. The main phenolic acids present in the seed coat were the benzoic acid derivatives which comprised 56% of the total phenolic substances whereas the flavonoids comprised 33%. The cotyledon had higher levels of cinnamic acid in total (31% of the total phenolic substances) compared to the amount of benzoic acid derivatives (21% of the total phenolic substances). Flavonoids comprised 48% of the total phenolic substances in the cotyledon.
Concentrations of most of the phenolic acids and flavonoid compounds in the free and bound solvent fractions were generally higher in seed coat fractions than in cotyledon fractions (Table 4). The phenolic composition in the beans was dominated by gallic acid, caffeic acid, catechin and myricetin, especially in the free phenolic and insoluble-bound fractions.
Tylosema esculentum tubers were found to contain high concentrations of gallic acid, and other unspecified phenolic substances (results not shown).
Antibacterial and anticandidal activity of T. esculentum extracts
Results presented in Table 5 show the antibacterial activity (against MRSA, C. diphtheriae and M. terrae) and the anticandidal activity (against C. albicans) of T. esculentum extracts, cranberry extract and the control antimicrobials. Antimicrobial activity is given in terms of MIC values, which were obtained using the broth microdilution method in which the microorganisms were exposed to 10-fold dilutions of T. esculentum extracts.
Inhibition of colonial growth was determined after co-incubation of T. esculentum extracts, which were diluted at selected/achievable concentrations, in nutrient broth for 3 h and plating of the treated inocula on solid agar. The level of inhibition (that is, the average number of colony forming units (CFU/mL) ± s.d.) is shown in Table 6. Percentage (%) inhibition of colonial formation was determined at selected T. esculentum extract concentrations, typically the determined MIC. Cranberry extracts were used as comparative antimicrobial plant extracts; cranberry has been widely shown to have low MIC values against different microorganisms, especially in the treatment of urinary tract infections, and has been widely recommended as an antimicrobial.
Using the colony counting method, it was shown that all T. esculentum polyphenolic fractions (MSCAM, MCAM, MSCS, MCS, MSCFP, MCFP, MSCIB and MCIB) and crude T. esculentum extracts (MCW, MSCW, MTW, MCE and MSCE) resulted in high inhibition of colonial growth (at least 90% - 100%) with co-incubation (Table 6). The crude T. esculentum extracts (MCW, MSCW, MTW, MCE and MSCE) had relatively high antimicrobial activity against MRSA, C. diphtheriae, and M. terrae, and low inhibitory effect against C. albicans (Table 5).
The results in Table 5 show that most of the polyphenolic fractions, namely MCAM, MSCAM, MSCIB, MSCS and MCS, had high antimicrobial activity (against C. diphtheriae, M. terrae and C. albicans), comparable to that of the standard antimicrobials tested. Only ethanolic extracts, among the more crude extracts, had anticandidal effects; crude extracts generally had low antimicrobial activity. The aqueous extracts frequently resulted in enhanced growth of the microorganisms tested. The respective t-values are shown in Table 7.
As shown in Table 7, the following extracts/fractions had MIC values close to those of control drugs (with t-values between -5 and 5): MCW (compared to streptomycin and gentamicin against M. terrae; t = -1.74 and t = 3.29, respectively), MCAM (compared to streptomycin against M. terrae; t = -1.29), MSCE (compared to streptomycin against C. albicans; t = 2.88), MSCIB (compared to streptomycin and gentamicin against M. terrae; t = -0.68 and t = 3.61, respectively), MCS and MSCS (compared to streptomycin against M. terrae; t = -1.36 and t = -1.88, respectively), MCFP (compared to gentamicin against M. terrae; t = 3.25), MSCFP (compared to amphotericin (t = 2.41); cefsulodin (t = 3.37); streptomycin (t = 3.18) and gentamicin (t = 3.37)), cranberry water extract (against C. albicans compared to amphotericin (t = 2.38); streptomycin (t = 3.46) and gentamicin (t = 3.46)). MSCIB and MSCFP showed comparable activity (-10 < t < 10) to that of most control antibiotics against C. diphtheria and M. terrae. MCW also showed comparable activity to control antibiotics tested against M. terrae (-10 < t < 10).
Exposure of microorganisms to T. esculentum extracts using the microdilution method resulted in low MIC values, comparable to those of cranberry or some of the conventional antibiotic drugs used. The phenolic extracts, MSCAM, MCS and MSCS, had low MIC values (0.44 mg/mL - 0.9 mg/mL), comparable to that for amphotericin B (0.5 mg/mL) against MRSA. MIC values of MSCAM, MSCIB, MCS and MSCS (0.1 mg/mL - 0.9 mg/mL) were comparable to those for cranberry water extract (0.16 mg/mL) and amphotericin B (0.5 mg/mL) against C. diphtheriae. MIC values of MCW (0.78 mg/mL), MCAM (0.9 mg/mL), MSCIB (0.9 mg/mL), MCS (0.9 mg/mL) and MSCS (0.9 mg/mL) were close to those for cranberry water extract (0.16 mg/mL) and streptomycin (1.0 mg/mL) against M. terrae. MIC values of MSCE (0.01 mg/mL), MCE (0.01 mg/mL), MSCAM (0.9 mg/mL) and MCFP (0.9 mg/mL) were comparable to those for cranberry water extract (0.16 mg/mL) and amphotericin B (0.05 mg/mL) against C. albicans. The extracts MSCAM, MCS, MSCS and MSCIB, were generally highly effective against most of the microorganisms tested; however the crude extracts (MCE and MSCE) were more effective against C. albicans. All the polyphenolic fractions (MCAM, MSCAM, MSCIB, MCIB, MCFP, MSCFP, MSCS and MCS), showed high inhibition of all the microorganisms tested; these fractions inhibited growth of all the microorganisms tested (S. aureus, C. diphtheriae, M. terrae and C. albicans) by at least 100% compared to control treatments. Co-incubation of the microorganisms with some crude extracts, especially aqueous extracts (MSCW and MCW), resulted in increased microbial counts.
Discussion
To our knowledge, this study is the first to determine the biological activity of the marama plant (T. esculentum) in terms of its antimicrobial (antibacterial and anticandidal) effects. In addition to the known ethnomedicinal and nutritional uses,2 we have shown here that T. esculentum extracts have a relatively high antimicrobial potential, especially the crude bean ethanolic extracts and polyphenolic fractions. Tylosema esculentum bean seed coat and cotyledon polyphenolic fractions had relatively high antibacterial and anticandidal activity, implying their possible use as new and potent antimicrobial drugs. While MIC values of the extracts against all microorganisms tested here (bacteria and candida) were generally higher than those of the control drugs conventionally used, some of the extracts, especially the polyphenolic fractions, had MIC values approximating those of the control drugs.
The fractions MSCAM, MCAM, MSCIB, MCS and MSCS had high antibacterial and anticandidal activities, having MIC values between 0.1 mg/mL and 1 mg/mL, which were comparable to those of some of the control antibiotics used (Table 5). The following extracts/fractions had MIC values similar to those of the respective control antibiotics (as determined by the t-values): MCW, MSCIB and MSCFP had t-values between -10 and 10 when MICs were compared to those of most of the control antibiotics tested against C. diphtheria and M. terrae; MCAM activity compared favourably to that of streptomycin against M. terrae (t = -1.29); MSCE activity compared favourably to that of streptomycin against C. albicans (t = 2.88); MCS and MSCS had MICs comparable to that of streptomycin against M. terrae (t = -1.36 and t -1.88, respectively); and MCFP activity compared favourably to that of gentamicin against M. terrae (t = 3.25) (Table 6). The polyphenolic fractions, especially MCAM, MSCAM, MCS and MSCS, had MIC values close to those for cranberry water extract against all test microorganisms. Cranberry was used in this test as it is widely accepted as an 'alternative and complementary' medicine for the treatment of urinary infections.41
All T. esculentum polyphenolic fractions tested resulted in total inhibition of bacterial and fungal growth at selected concentrations, especially those above MIC values (Table 5). Among the crude T. esculentum bean extracts, MCW had relatively high inhibitory effects (as shown by low MIC values) against M. terrae (MIC = 0.78 mg/mL) (Table 5). After in vitro co-incubation of bacteria and yeast for 3 h, it was shown that all T. esculentum polyphenolic fractions (MSCAM, MCAM, MSCS, MCS, MSCFP, MCFP, MSCIB and MCIB) and crude T. esculentum extracts (MCW, MSCW, MTW, MCE and MSCE) resulted in high inhibition of colonial growth (at least 90% to 100%) (Table 6). The crude T. esculentum extracts (MCW, MSCW, MTW, MCE and MSCE) had relatively high antimicrobial activity against MRSA, C. diphtheriae and M. terrae, and low inhibitory effect against C. albicans (Table 5 and Table 6). This finding indicates that the duration of exposure of the microorganism to the extract may be an important factor in determining antimicrobial efficacy - most of the extracts, especially the phenolic ones, were effective in killing the bacterial and fungal cells within 3 h of exposure.
Higher antimicrobial activity was found with the T. esculentum seed coats, particularly the polyphenolic fractions (MSCS and MSCIB), than the cotyledon extracts (Table 5 and Table 6). Tylosema esculentum bean seed coat polyphenolic fractions, especially seed coat soluble-bound extract (MSCS), were highly active against M. terrae, C. diphtheriae and C. albicans (Table 5 and Table 6). MSCS had an MIC value comparable to that of cefsulodin against M. terrae. Because polyphenolic fractions contain high concentrations of phenolic substances, their high antimicrobial activity shows that phenolic substances contribute to this antimicrobial activity. Phenolic fractions are known to accumulate during ethanolic and methanolic extraction of crude extracts.42,43 Tylosema esculentum seed coats have been shown to contain higher amounts of phenolic substances than cotyledons (Table 2, Table 3 and Table 4), especially catechin, rutin, naringin, hesperidin and myricetin; while cotyledons contain higher amounts of naringenin and kaempferol than seed coats (Table 3 and Table 4). Quercetin was shown to be equally distributed between cotyledons and seed coats (Table 3 and Table 4).
The observed high antimicrobial activity of the bean polyphenolic fractions could also be as a consequence of the high content of gallic acid (Table 3 and Table 4); certain fatty acids existing in high concentrations such as palmitic acid and oleic acid21,22;or the high phytosterol concentration.14 Fatty acid combinations in apple oil were reported to contribute to the high antimicrobial activity of the oil.44 Apple seed oils have been shown to consist of linoleic acid (50.7 g/100 g - 51.4 g/100 g), oleic acid (37.49 g/100 g - 38.55 g/100 g), palmitic acid (6.51 g/100 g - 6.60 g/100 g), stearic acid (1.75 g/100 g - 1.96 g/100 g) and arachidic acid (1.49 g/100 g - 1.54 g/100 g)44; the same fatty acids are also present in T. esculentum oil. Tylosema esculentum oil however contains more oleic acid (34% - 49%) than linoleic acid (22% - 26.4%).21,22 Comparably, apple seed oil was found to almost completely inhibit bacteria, with reduced inhibitory effects against fungi; the MIC of apple seed oil ranged from 0.3 mg/mL to 0.6 mg/mL.44
Phytosterols present in T. esculentum beans could also have contributed to the observed antimicrobial activity shown previously.45 Lignans, also known to be present in the beans, have been shown to have both antibacterial46,47 and anticandidal activity.46,48 Phytosterols, such as beta-sitosterol and stigmasterol present in T. esculentum beans in high concentrations, are also known to have antimicrobial activity against Gram-positive and Gram-negative bacteria and a yeast strain.49 Phytosterols, particularly 19-acetoxy-7,15-isopimaradien-3beta-ol and 7,15-isopimaradien-19-ol in Aeollanthus rydingianus showed MIC values between 3.90 µg/mL and 15.62 µg/mL for S. aureus and 7.81 µg/mL for Enterococcus hirae.49 Tylosema esculentum tubers may also contain tannins, previously shown to have antimicrobial activity,42 as previously reported in the roots of Bauhinia petersiana, another member of Leguminosae.49
Additionally, T. esculentum beans are also known to be rich in trypsin inhibitors (of the Kunitz type).50 Some trypsin inhibitors, including serine protease inhibitors, have been shown to contribute to the antimicrobial effects of known antimicrobial peptide cocktails.51
In summary, we have shown that some T. esculentum bean and tuber extracts have high antibacterial and anticandidal effects in vitro, depending on the extraction procedure and microorganism. Polyphenolic fractions, in particular, contained high concentrations of different phenolic substances; higher antimicrobial activity in the seed coat extracts was as a result of higher phenolic concentration than in cotyledon extracts. Crude water and ethanolic extraction did not prove useful in extracting the high antimicrobial effects observed with polyphenolic fractions.
Conclusion
Our study is the first to show the importance of T. esculentum beans and tubers against diseases caused by certain bacteria and a strain of candida. We therefore conclude that T. esculentum bean extracts, especially phenolic cotyledon (MCAM, MCFP and MCIB) and seed coat fractions (MSCAM, MSCFP and MSCIB), have high antimicrobial activity. Our findings therefore suggest T. esculentum beans as an important source of microbicides against the tested bacteria and yeast. Tylosema esculentum extracts have exhibited broad spectrum antibacterial activity, mostly harboured in polyphenolic fractions.
The method of extraction and the microorganism to be targeted would be important factors to consider for optimal activity of the extract, if T. esculentum extracts were to be used as antimicrobial agents. Most T. esculentum bean seed coat extracts generally had greater inhibitory effect against bacteria and yeast because of their higher phenolic content than the cotyledon extracts. Phenolic substances such as gallic acid, phytosterols, lignans, certain fatty acids, peptides (specifically protease inhibitors) and amino acids are among the many chemicals in T. esculentum thought to be responsible for the antimicrobial activities of the T. esculentum extracts.
Acknowledgements
We would like to thank the technical team in the Department of Chemistry, University of Botswana (A. Muzila, C. Ntwaagae, O. Oyentunji, O. Mazimba, E. Yeboah and S. Ditlhabi) for providing the bean ethanolic extracts and for processing the tuber; L. Gradisnik for her excellent technical support; and Dr B. Filipič (University of Ljubljana, Slovenia), for supplying the microbial strains used. This work was supported by the EU FP-6 grant (FP6-2004-INCO-DEV-3-MARAMA II - 032059), Development of Innovative and Healthful Marama Bean (TE) Products Targeting Niche Markets.
References
1. Van der Maesen LJG. Tylosema esculentum (Burch.) A.Schreib. In: Brink M, Belay G, editors. PROTA 1: Cereals and pulses/Cereales et legumes secs. [book on CD-ROM]. Wageningen: PROTA; 2006. [ Links ]
2. McMurdo ME, Argo I, Phillips G, et al. Cranberry or trimethoprim for the prevention of recurrent urinary tract infections? A randomized controlled trial in older women. J Antimicrob Chemother. 2009;63(2):389-395. doi:10.1093/jac/dkn489, PMid:19042940, PMCid:2639265 [ Links ]
3. Chan KH, Pan RN, Hsu MC, Hsu KF. Urinary elimination of ephedrines following administration of the traditional Chinese medicine preparation Kakkon-to. J Anal Toxicol. 2008;32(9):763-767. PMid:19021932 [ Links ]
4. Chomnawang MT, Trinapakul C, Gritsanapan W. In vitro antigonococcal activity of Coscinium fenestratum stem extract. J Ethnopharmacol. 2009;122(3):445-449. doi:10.1016/j.jep.2009.01.036, PMid:19429310 [ Links ]
5. Tomczykowa M, Tomczyk M, Jakoniuk P, Tryniszewska E. Antimicrobial and anticandidal activities of the extracts and essential oils of Bidens tripartita. Folia Histochem Cyto. 2008;46(3):389-393. doi:10.2478/v10042-008-0082-8, PMid:19056546 [ Links ]
6. Sofowora A. Recent trends in research into African medicinal plants. J Ethnopharmacol. 1993;38(2-3):209-214. doi:10.1016/0378-8741(93)90017-Y [ Links ]
7. Chingwaru W, Faria ML, Saravia C, Cencic A. Indigenous knowledge of health benefits of marama plant among respondents in Ghantsi and Jwaneng of Botswana. Afr J Food Agric Nutr Dev. 2007;7(6). Available from: http://www.ajfand.net/Issue17/Issue17commentary.htm [ Links ]
8. Amarteifio J, Moholo D. The chemical composition of four legumes consumed in Botswana. J Food Compos Anal. 1998;11(4):329-332. doi:10.1006/jfca.1998.0595 [ Links ]
9. Bower N, Hertel K, Storey R, Storey R. Nutritional evaluation of marama bean (Tylosema esculentum, Fabaceae): Analysis of the seed. Econ Bot. 1988;42(4):533-540. doi:10.1007/BF02862798 [ Links ]
10. Keegan AB, Van Staden J. Marama bean, Tylosema esculentum, a plant worthy of cultivation. S Afr J Sci. 1981;77(9):387. [ Links ]
11. Holse M, Husted S, Hansen A. Chemical composition of marama bean (Tylosema esculentum) - a wild African bean with unexploited potential. J Food Compos Anal. 2010, in press. doi:10.1016/j.jfca.2010.03.006 [ Links ]
12. Dubois M, Lognay G, Baudart E, et al. Chemical characterization of Tylosema-Fassoglensis (Kotschy) Torre and Hillc oilseed. J Sci Food Agric. 1995;67(2):163-167. doi:10.1002/jsfa.2740670204 [ Links ]
13. Francis CM, Campbell MC. New high quality oil seed crops for temperate and tropical Australia. Canberra: Rural Industries Research and Development Corporation; 2003. [ Links ]
14. Mitei YC, Ngila JC, Yeboah SO, et al. NMR, GC-MS and ESI-FTICR-MS profiling of fatty acids and triacylglycerols in some Botswana seed oils. J Am Oil Chem Soc. 2008;85:1021-1032. doi:10.1007/s11746-008-1301-3 [ Links ]
15. Wehmeyer AS, Lee M, Whiting M. The nutrient composition and dietary importance of some vegetable foods eaten by the !Kung Bushmen. S Afr Med J. 1969;43:1529. PMid:5364781 [ Links ]
16. Gueguen J. Legume seed protein extraction, processing, and end product characteristics. Qual Plant. 1983;32(3-4):267-303. doi:10.1007/BF01091191 [ Links ]
17. Nassar AG, Mubarak AE, El-Beltagy AE. Nutritional potential and functional properties of tempe produced from mixture of different legumes. 1: Chemical composition and nitrogenous constituent. Int J Food Sci Technol. 2008;43(10):1754-1758. doi:10.1111/j.1365-2621.2007.01683.x, PMid:20548306 [ Links ]
18. Belitz HD, Grosch W, Schieberle P. Food chemistry. Berlin: Springer-Verlag, 2004; p. 1070. [ Links ]
19. Hymowitz T, Collins FI, Walker WM, Walker WM. Relationship between content of oil, protein, and sugar in soybean seed. Agron J. 1972;64(5):613-616. doi:10.2134/agronj1972.00021962006400050019x [ Links ]
20. Maruatona GN, Duodu KG, Minnaar A. Physicochemical, nutritional and functional properties of marama bean flour. Food Chem. 2010;121(2):400-405. doi:10.1016/j.foodchem.2009.12.054 [ Links ]
21. Ripperger-Suhler JA, Longenecker JB. Assessment of the nutritional value of the marama bean. Report to the Center for the Study of Human Adaptation, Division of Graduate Nutrition, University of Texas, Austin; 1982. [ Links ]
22. Biesele M, Murray RE. Alternative food plants for arid regions: Marama and other plant foods of Kalahari foragers. An applied ethno-botanical study. Report to the Center for the Study of Human Adaptation, Division of Graduate Nutrition, University of Texas, Austin; 1983. [ Links ]
23. Parlatan A, Saricoban C, Ozcan MM. Chemical composition and antimicrobial activity of the extracts of Kefe cumin (Laser trilobum L.) fruits from different regions. Int J Food Sci Nutr. 2009;60(7):606-617. doi:10.3109/09637480801993938, PMid:19817640 [ Links ]
24. Mazur W, Fotsis T, Wahala K, et al. Isotope dilution gas chromatographic-mass spectrometric method for the determination of isoflavonoids, coumestrol, and lignans in food samples. Anal Biochem. 1996;233:169-180. doi:10.1006/abio.1996.0025, PMid:8789715 [ Links ]
25. Penalvo JL, Heinonen SM, Nurmi T, et al. Plant lignans in soy-based health supplements. J Agric Food Chem. 2004;52(13):4133-4138. doi:10.1021/jf0497509, PMid:15212459 [ Links ]
26. Kang MS, Oh JS, Kang IC, et al. Inhibitory effect of methyl gallate and gallic acid on oral bacteria. J Microbiol. 2008;46(6):744-750. doi:10.1007/s12275-008-0235-7, PMid:19107406 [ Links ]
27. O'Dell BL, De Boland A. Complexation of phytate with proteins and cations in corn germ and oilseed meals. J Agric Food Chem. 1976;24(4):804-808. doi:10.1021/jf60206a034 [ Links ]
28. Chang RS, Yeung HW. Inhibition of growth of human immunodeficiency virus in vitro by crude extracts of Chinese medical herbs. Antiviral Res. 1988;9:163-176. doi:10.1016/0166-3542(88)90001-0 [ Links ]
29. Vilhjalmsdottir L, Fisher H. Castor bean meal as a protein source for chickens: Detoxification and determination of limiting amino acids. J Nutr. 1971;101:1185. PMid:5106935 [ Links ]
30. Yu YS, Hsu CL, Yen GC. Anti-inflammatory effects of the roots of Alpinia pricei Hayata and its phenolic compounds. J Agric Food Chem. 2009;57(17):7673-7680. doi:10.1021/jf901327g, PMid:19685877 [ Links ]
31. Krygier K, Sosulski F, Hogge L. Free, esterified, and insoluble-bound phenolic acids. 1. Extraction and purification procedure. J Agric Food Chem. 1982;30:330-334. doi:10.1021/jf00110a028 [ Links ]
32. Naczk M, Shahidi F. Extraction and analysis of phenolic substances in food. J Chromatogr A. 2004;1054:95-111. doi:10.1016/S0021-9673(04)01409-8, doi:10.1016/j.chroma.2004.08.059, PMid:15553136 [ Links ]
33. BreezeTM. Part number WA01016. Milford, MA: Waters Corporation; 2000. [ Links ]
34. Kuzina LV, Miller TA, Cooksey DA. In vitro activities of antibiotics and antimicrobial peptides against the plant pathogenic bacterium Xylella fastidiosa. Lett Appl Microbiol. 2006;42:514-520. doi:10.1111/j.1472-765X.2006.01898.x, PMid:16620212 [ Links ]
35. Ali-Shtayeh MS, Yaghmour RM, Faidi YR, et al. Antimicrobial activity of 20 plants used in folkloric medicine in the Palestinian area. J Ethnopharmacol. 1998;60(3):265-271. doi:10.1016/S0378-8741(97)00153-0 [ Links ]
36. National Committee for Clinical Laboratory Standards (NCCLS). Reference method for broth dilution antifungal susceptibility testing of yeasts: Approved Standard M27-A. Wayne, PA: NCCLS; 1997. [ Links ]
37. Swenson JM, Thornsberry C, Silcos VA. Rapidly growing mycobacteria: Testing of susceptibility to 34 patients. Antimicrob Agents Chemother. 1982;22:186-192. PMid:6927280, PMCid:183707 [ Links ]
38. Zasada AA, Zaleska M, Podlasin RB, Seferynska I. The first case of septicemia due to nontoxigenic Corynebacterium diphtheriae in Poland: Case report. Ann Clin Microbiol Antimicrob. 2005;4:8. doi:10.1186/1476-0711-4-8, PMid:15876349, PMCid:1156865 [ Links ]
39. STATISTICA. Version 7.1. Tulsa, OK: StatSoft Inc.; 2005. [ Links ]
40. Stermitz FR, Lorenz P, Tawara JN, et al. Synergy in a medicinal plant: Antimicrobial action of berberine potentiated by 5*-methoxyhydnocarpin, a multidrug pump inhibitor. Proc Natl Acad Sci. USA. 2000;97(4):1433-1437. doi:10.1073/pnas.030540597, PMid:10677479 [ Links ]
41. Del Mar C. Urinary tract infections in healthy women: A revolution in management? BMC Fam Pract. 2010;11:42. doi:10.1186/1471-2296-11-42, PMid:20504297, PMCid:2885322 [ Links ]
42. Akinyemi KO, Oluwa OK, Omomigbehin EO. Antimicrobial activity of crude extracts of three medicinal plants used in south-west Nigerian folk medicine on some food borne bacterial pathogens. Afr J Tradit Complement Altern Med. 2006;3(4):13-22. [ Links ]
43. Cowan MM. Plant products as antimicrobial agents. Clin Microbiol Rev. 1999;12:564-582. PMid:10515903, PMCid:88925 [ Links ]
44. Tian HL, Zhan P, Li KX. Analysis of components and study on antioxidant and antimicrobial activities of oil in apple seeds. Int J Food Sci Nutr. 2010;61(4):395-403. doi:10.3109/09637480903535772, PMid:20128637 [ Links ]
45. Sanchez Perera LM, Varcalcel L, Escobar A, Noa M. Polyphenol and phytosterol composition in an antibacterial extract from Rhizophora mangle L. bark. J Herb Pharmacother. 2007;7(3-4):107-128. PMid:18928137 [ Links ]
46. Kawaguchi Y, Yamauchi S, Masuda K, et al. Antimicrobial activity of stereoisomers of butane-type lignans. Biosci Biotechnol Biochem. 2009;73(8):1806-1810. doi:10.1271/bbb.90167 [ Links ]
47. Akiyama K, Yamauchi S, Maruyama M, et al. Antimicrobial activity of stereoisomers of morinols A and B, tetrahydropyran sesquineolignans. Biosci Biotechnol Biochem. 2009;73(1):129-133. doi:10.1271/bbb.80536 [ Links ]
48. Nakato T, Yamauchi S, Tago R, et al. Syntheses and antimicrobial activity of tetrasubstituted tetrahydrofuran lignan stereoisomers. Biosci Biotechnol Biochem. 2009;73(7):1608-1617. doi:10.1271/bbb.90107 [ Links ]
49. Rijo P, Simoes MF, Duarte A, Rodriguez B. Isopimarane diterpenoids from Aeollanthus rydingianus and their antimicrobial activity. Phytochemistry. 2009;70(9):1161-1165. doi:10.1016/j.phytochem.2009.06.008, PMid: 19631355 [ Links ]
50. Nadaraja D, Weintraub ST, Hakala KW, et al. Isolation and partial sequence of a Kunitz-type elastase specific inhibitor from marama bean (Tylosema esculentum). J Enzyme Inhib Med Chem. 2010;25(3):377-382. doi:10.3109/14756360903179500, PMid:19883219 [ Links ]
51. Brown SE, Howard A, Kasprzak AB, et al. A peptidomics study reveals the impressive antimicrobial peptide arsenal of the wax moth Galleria mellonella. Insect Biochem Mol Biol. 2009;39(11):792-800. doi:10.1016/j. ibmb.2009.09.004, PMid:19786100 [ Links ]
 Correspondence to:
Correspondence to:
Avrelija Cencic
Postal address:
Department of Microbiology, Biochemistry, Molecular Biology and Biotechnology, Faculty of Agriculture and Life Sciences, University of Maribor
Pivola 10, 2311 Hoče, Slovenia
email: avrelija.cencic@uni-mb.si
Received: 20 July 2010
Accepted: 29 Nov. 2010
Published: 03 Mar. 2011
© 2011. The Authors. Licensee: OpenJournals Publishing. This work is licensed under the Creative Commons Attribution License.














