Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.107 n.1-2 Pretoria Jan./Feb. 2011
http://dx.doi.org/10.4102/sajs.v107i1/2.269
RESEARCH ARTICLES
Spatial and temporal assessment of gaseous pollutants in the Highveld of South Africa
Alexandra S. LourensI; Johan P. BeukesI; Pieter G. van ZylI; Gerhardus D. FourieII; Johanna W. BurgerII; Jacobus J. PienaarI; Colin E. ReadI; Johan H. JordaanI
ISchool of Physical and Chemical Sciences, North West University, Potchefstroom campus, South Africa
IISasol Technology, Sasolburg, South Africa
ABSTRACT
Diverse natural and anthropogenic activities in the Highveld of South Africa contribute to elevated levels of inorganic and organic gaseous pollutant species. The primary aims of this investigation were to determine spatial and temporal distributions of nitrogen dioxide (NO2), sulphur dioxide (SO2) and ozone (O3), as well as benzene, toluene, ethylbenzene and xylene (BTEX) in this area. Sampling was conducted on a monthly basis for a year at eight strategically selected sites. The highest NO2, SO2 and BTEX concentrations were measured at sites with significant local sources and/or sites impacted by more remote sources, as indicated by overlay back trajectories. O3 concentrations were found to be lower at sites with high levels of NO2, SO2 and BTEX. NO2 and SO2 peaked during winter, while O3 peaked in spring. NO2 and SO2 temporal concentration variations were ascribed to differences in seasonal meteorological conditions, as well as additional sources in winter. The O3 peak coincided with a seasonal CO peak, which was identified as an important precursor for O3 formation. No distinct seasonal trend was observed for BTEX. The annual average concentrations for SO2, NO2, O3 and benzene were below the South African annual standards at all sites.
Introduction
The South African Highveld is known for its diverse anthropogenic activities, which include agriculture, metallurgical and mining operations, power generation (coal-fired power stations), petrochemical operations, coal dumps and transportation.1,2 The dry and cold winters (June to August) experienced in the Highveld also result in additional combustion of coal and wood for domestic heating, as well as increased occurrences of biomass burning (veld fires) (July-September). Anthropogenic activities contribute to elevated levels of gaseous pollutants in the atmosphere, which include the inorganic gaseous species nitrogen dioxide (NO2), sulphur dioxide (SO2) and ozone (O3), as well as volatile organic compounds (VOCs) such as benzene, toluene, ethylbenzene and xylene (BTEX). Primary pollutants emitted into the atmosphere can also react and lead to the formation of secondary pollutants. VOCs, for instance, play a significant role in particle formation.3 VOCs can also act as precursors for ozone formation during photochemical reactions with NOx,4 hence modifying the oxidising capacity of the atmosphere.5,6
There are adverse human health effects associated with the abovementioned atmospheric pollutants and they are also harmful to ecosystems.7 Ozone is highly phytotoxic and can cause damage to crops and native vegetation.8 NOx and SO2 could have long-term effects such as pulmonary asthma and chronic bronchitis respectively.9,10 Benzene is a known carcinogen and is associated with leukemia whereas toluene can cause problems such as foetal malformation.11
In the last quarter of 2007 the South African Minister of Environmental Affairs and Tourism proclaimed eastern Gauteng and western Mpumalanga, in terms of the National Environmental Management: Air Quality Act, as a national air pollution hotspot termed the Highveld Priority Area (HPA). The HPA covers a surface of 31 106 km2 and has a population of 3.6 million people. This declaration implies that national government takes responsibility for assessing levels of pollution and putting in place emission reduction plans, while local and provincial governments participate in the process.12 Apart from the local and regional importance of the HPA, satellite-based measurements have also indicated that this area has some of the highest NO2 concentrations in the world,13 emphasising the global importance of this region.
The monitoring of inorganic gaseous species with active and passive samplers is well established in South Africa,14,15 whereas very little data exist for VOCs. Currently, measurements of air pollutants usually are carried out by local authorities (municipalities) and certain industries, which do not necessarily give a good spatial representation of larger areas. Most of these data sets are also not published in peer-reviewed journals, which makes it difficult to evaluate the data quality. Therefore, the primary aims of this investigation were to determine spatial and temporal distributions of ambient concentrations of NO2, SO2, O3 and BTEX in the Highveld for one year.
Materials and methods
Sampling sites
Eight sampling sites were selected within a sampling grid of 600 km2 as shown in Figure 1. The sites were selected to be representative of the Highveld. The sites were located in Vanderbijlpark, Delmas, Witbank, Ermelo, Carolina, Amersfoort, Standerton and Balfour. The site locations ranged from highly industrialised areas such as Witbank and Vanderbijlpark to the remote rural areas of Carolina and Amersfoort. Apart from localised pollution sources such as traffic emissions, household combustion and small industries, there are also larger pollution sources in this area (Figure 1), which include power stations, a petrochemical plant (Secunda) and metallurgical operations (based mainly in Witbank, Middelburg and Vanderbijlpark). Coal mining activities scattered across the entire sampling area could also be regarded as pollution sources (e.g. from materials handling and spontaneous combustion of coal). Additionally, emissions from the Johannesburg conurbation could also affect the measurement area.
The relevance of this investigation was further enhanced by a recent regional study conducted during 2005-2007, which identified the area under investigation as a pollution hotspot for SO2 and NO2.16 The eight sites selected for this study gave a higher monitoring resolution for the abovementioned hotspot.
Sampling equipment
Sampling commenced in August 2007 and was conducted for a year. Samples were exposed for a full month for which monthly averages were calculated.
Passive samplers developed and used by the Atmospheric Chemistry Research Group at the North West University were used for NO2, SO2 and O3 measurements.14,17 The passive samplers consisted of two plastic snap-on caps with a stainless steel mesh (25 mm o.d., thickness of 160 µm and porosity of 40%), a 175-µm Teflon PTFE filter (GIC, Johannesburg, South Africa) with an outer diameter of 25 mm, a porosity of 85% and an impregnated paper disc (Whatman filter, GIC, Johannesburg, South Africa) at the rear end. The plastic snap-on caps and the stainless steel mesh were manufactured by the instrument makers of the North West University. The mesh and PTFE filter prevented aerosols from diffusing into the sampler. The Whatman filter was impregnated with a specific chemical to react with and trap the pollutant of interest.14 Samplers were prepared and stored in a freezer prior to sampling. Exposed samplers were stored similarly before analysis.
Tenax TA adsorbent tubes (Sigma-Aldrich, Johannesburg, South Africa), which are commonly used for BTEX sampling,18,19 were used in accordance with the US EPA TO-17 Compendium method.18 The tubes were tested for leaks and preconditioned with helium for 30 min at 350 ºC at a flow rate of 50 cm3/min before they were used. After treatment, the tubes were sealed with Swagelok® brass ¼" caps (P/N 0990-8851, Sigma-Aldrich, Johannesburg, South Africa) and stored in a freezer. The Swagelok® caps were removed before exposure in the field and replaced with diffusive caps for effective air diffusion into the tubes. In order to attain reproducibility, two samplers for each station were exposed for an entire month. After sampling, the tubes were removed and closed with the Swagelok® caps. Each tube was separately wrapped in aluminium foil and stored in a freezer before analysis.
Chemical analysis
Inorganic gaseous species were analysed using two different analytical methods as previously described.14 SO2 and O3 samples were analysed on a Dionex Ion Chromatograph System (ICS-3000, Anatech Instruments, Johannesburg, South Africa) fitted with an IonPac AS18 (2 mm x 50 mm) analytical column, IonPac AG18 (2 mm x 50 mm) guard column and ASRS Ultra II 2-mm self-regulating suppressor. NO2 samples were analysed on a Varian Cary® 50 UV-visible spectrophotometer (SMM Instruments, Johannesburg, South Africa).
VOC adsorbent tubes were analysed with a Hewlett Packard Agilent 6899 gas chromatograph (SMM Instruments, Johannesburg, South Africa) connected to a Micromass AutoSpecTM time-of-flight mass spectrometer (TOF-MS) detector. The samples were introduced into the gas chromatograph using a PerkinElmer® TurboMatrixTM desorber (Shelton, Connecticut, USA). The equipment was set up and used as prescribed by the US EPA TO-14 Compendium method.18 A suitable calibration standard (Cat no: 41900-U, Supelco, Taipei, Taiwan), which contained 39 VOC compounds (100 ppb), was used as an external standard.
Overlay back trajectories
Individual hourly back trajectories with an arrival height of 100 m were compiled in HYSPLIT 4.820 for each site. The duration of each back trajectory was taken as 96 h (i.e. 4 days). An arrival height of 100 m was chosen because the orography in HYSPLIT is not very well defined, thus lower arrival heights could result in increased error margins on individual trajectory calculations. Considering the above, 24 back trajectories per day per site were obtained for the monitoring duration of one year. For each site individual back trajectories generated in HYSPLIT (24 x 365 days) were superimposed in MATLAB 7.8.0.21 In the overlay back-trajectory graph compiled with MATLAB 7.8.0 a colour code indicates the percentage of trajectories passing over 0.2º x 0.2º grid cells, with red being the highest percentage.
Results and discussion
Spatial distribution
The spatial distribution of the annual average concentrations of the measured inorganic gases is presented in Figure 2. NO2 and SO2 levels were the highest at sites situated in or near industrialised areas. The highest O3 concentrations were recorded at sites positioned in more rural areas. It is also evident that sites with the highest SO2 and NO2 concentrations exhibited the lowest O3 levels, while higher O3 concentration coincided with lower SO2 and NO2 levels.
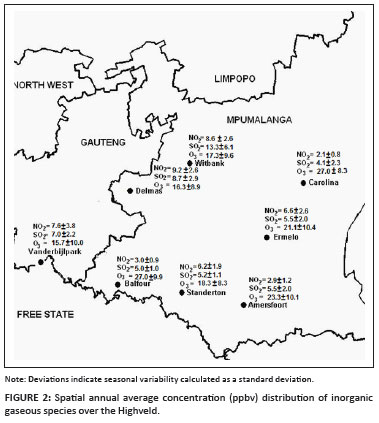
Although NO2 is a precursor for the formation of tropospheric O3, high NO2 in polluted areas does not necessarily imply high O3 levels, because O3 is rapidly consumed during oxidation of other pollutants. When the pollution dilutes through transportation, O3 levels will increase if NO2 and radiation are not limiting factors. For the same reason, virtually no O3 is observed in a plume close to a point source, while O3 increases as dispersion occurs.22
In an effort to determine the link between sources and measured pollutant species concentrations, overlay back trajectories for the entire 12-month sampling period for every measuring station were generated (Figure 3). Each monitoring station in these figures is indicated by the point where two perpendicular white lines cross. In addition, these white lines also divide the area into sectors aiding the identification of dominating air mass movements towards sampling sites. The black lines in Figure 3 are contour timelines that indicate the average trajectory position in each direction at a given time. The overlay back trajectories obtained indicate that the predominant movement of air masses were from a northerly to easterly direction at most of the measurement sites during the sampling period.
As can be seen in Figure 2, the combined annual average concentrations of NO2 and SO2 were the highest at Witbank, which can be partially attributed to industrial and mining activities in the immediate proximity of this highly industrialised area. Major point sources in the immediate vicinity include a host of metallurgical operations i.e. ferrochrome, vanadium, manganese and steel smelters, as well as coal-based industries such as char and electrode paste production. In addition to local sources, anthropogenic activities further away from the site could also contribute to increased NO2 and SO2 concentrations. Overlay back trajectories (Figure 3a) for this site show dominant incoming air mass movements from the sector between the north and south-east. In this sector there are several additional major point sources i.e. three coal-fired power stations, as well as ferrochrome and stainless steel plants at Middelburg (about 27 km from Witbank) that could contribute to measured pollutant levels. It can also be expected that smaller industrial, traffic and household sources would contribute to the presence of pollutant species at this site.
The second highest combined annual average concentrations of NO2 and SO2 were measured at Delmas. In contrast to Witbank, Delmas is set in rural surroundings suggesting that the relatively high pollutant levels could not be ascribed to major local sources. Overlay back trajectories (Figure 3b) for this site show dominant incoming air mass movements from the sector between the north and south-east. This sector includes Witbank, which implies that the same sources that had an impact on Witbank would have an effect on Delmas, with some dilution, as the two sites are about 62 km apart. In the north to south-easterly sector of Delmas there are, however, also three additional coal-fired power stations and a petrochemical plant that could also contribute to higher levels of NO2 and SO2. It can therefore be concluded that the relatively high pollutant concentrations at Delmas were a direct result of air mass being swept over an area with several major sources.
Relatively high levels of NO2 and SO2 were also determined at Vanderbijlpark. This is also a highly industrialised area with numerous significant local sources such as petrochemical industries, steel smelters and a coal-fired power station. Figure 3c shows overlay back trajectories for this site, which indicate that the predominant air mass movement was from the sector between the north and east. The Johannesburg conurbation is within the 12-hour back-trajectory contour to the north-north-east of the measurement site in this sector, which indicates that it could contribute to the relatively high pollutant concentrations. Also, within this dominant sector of air mass movement there are several coal-fired power stations, a petrochemical plant and the metallurgical operations in the Witbank-Middelburg area within the 24-hour back-trajectory contour line.
In contrast to the abovementioned sites (Witbank, Delmas and Vanderbijlpark), the NO2 and SO2 concentrations at Carolina were the lowest. Carolina is a small rural town with no major local sources. The overlay back trajectories (Figure 3d) show that there are no major sources (Figure 1) within the 24-hour contour line in the dominant air mass movement sector between the north and south-east. It cannot, however, be regarded as an unpolluted background site because NO2 and SO2 levels are higher than those reported for South African background sites.23 As previously mentioned, the relatively high O3 concentrations also indicate that this site is possibly impacted by aged polluted air masses.
Combined SO2 and NO2 concentrations at Balfour, Standerton, Amersfoort and Ermelo were lower than those recorded at Witbank, Delmas and Vanderbijlpark, but higher than those measured at Carolina. In general this can be explained by the fact that these stations do not have many significant local sources and are also further removed from most of the major point sources on the Highveld. Overlay back trajectories for these sites (Figure 3e-h) show dominant incoming air mass movements from the sector between north and east for Balfour and Standerton and from the north to south-east sector for Amersfoort and Ermelo.
Similar SO2 concentrations were measured at Balfour and Standerton, while the annual average NO2 concentration at Standerton was more than double that determined at Balfour. This can be attributed to the proximity of a single major point source 23 km north-north-east of Standerton (Figure 1). NO2 has a shorter lifetime in the atmosphere than SO2.24 As similar emission concentration ratios of large point sources (especially coal-fired power plants) impacting on Balfour and Standerton are present, the longer lifetime of SO2 results in similar SO2 levels at both sites. In contrast, the nearby source at Standerton results in higher NO2 levels. This explanation is supported by the finding that O3 concentrations were higher at Balfour than at Standerton.
Ermelo and Amersfoort had the same annual SO2 concentration for the sampling period. The relatively low concentrations measured at Amersfoort were unexpected because there is a point source in close proximity to the measurement site. A plausible explanation is that this point source is not situated in the dominant incoming air mass movements sector, but in the south-west sector. To some extent NO2 and SO2 concentrations at Ermelo were anomalies because there are no sources in the dominant incoming air mass sector. The 12-hour back-trajectories contour line does, however, indicate some air mass movements from the westerly region, hence implying impact from the major sources in this area. This is confirmed by the slightly higher concentration of NO2, which has a shorter lifetime than SO2.
The spatial distribution obtained during this investigation can be compared to results obtained during the previous study conducted during 2005-2007 over a larger area in the northern and eastern parts of South Africa.16 Both studies included sampling sites at Amersfoort and Standerton. The results obtained at these sites during 2005-200716 are comparable to those measured in this study. Furthermore the 2005-2007 study identified a pollution hotspot in the Highveld. As mentioned previously, the eight sampling sites of the current investigation were situated within this hotspot. The enhanced sampling spatial resolution enabled the identification of a more localised hotspot i.e. the Witbank area.
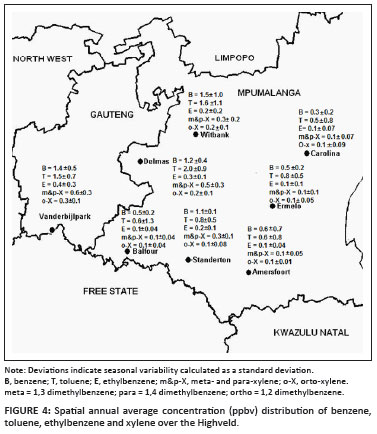
The spatial distribution of the annual average concentrations of BTEX in the Highveld is shown in Figure 4. A similar spatial trend observed for NO2 and SO2, is evident for BTEX levels. BTEX concentrations were the highest at Witbank, Delmas and Vanderbijlpark. The concentrations measured at Balfour, Standerton, Amersfoort and Ermelo were lower, with Carolina registering the lowest levels. As indicated previously, the overlay back trajectories in Figure 3 can be used to clarify the link between measured levels, sources and the impact of meteorological conditions. The resemblance between the spatial distributions of BTEX and NO2/SO2 therefore indicate possible corresponding sources for all these species. As the spatial BTEX distribution and linkage to sources (industrialised areas) were similar to that already discussed for SO2 and NO2, in depth discussion on spatial distribution of BTEX is not given.
Temporal distribution
The monthly mean concentrations for inorganic gaseous species over the Highveld are shown in Figure 5. The error bars on the graph indicate the maximum and minimum concentrations measured at the different sites during the entire sampling period.
Seasonal trends were observed, with NO2 and SO2 peaking during winter while O3 peaked in spring. Similar trends were observed in a previous investigation.14 NO2 and SO2 concentration variations can be explained by the difference in meteorological conditions experienced in the Highveld in winter and summer. The warm and wet summers are associated with unstable meteorological conditions, which increase the vertical motion and dispersion in the atmosphere. In addition, rain also scavenges pollutants more frequently from the atmosphere during summer. In contrast, winters are characterised by the formation of surface inversion layers inhibiting vertical atmospheric mixing and effectively trapping the primary pollutants.2,25 Increased occurrences of combustion for domestic heating during winter also contribute to elevated levels of SO2 and NO2.
The seasonal variation of O3 can possibly be ascribed to a combination of various contributing factors. During spring relatively stable conditions (pre-rain season) still prevail, while there is an increase in photochemical activity (longer days). Several compounds, which are considered to be O3 precursors, participate in the photochemical reaction with NO2 in O3 formation. However, it seems as if CO is probably the most important precursor in the South African context. The importance of CO as a precursor for O3 formation is well described in the literature.26 Other authors27 also found CO and O3 to peak at the same time during the year, i.e. spring, in South Africa. The origin of the CO peak during late winter and early spring can be attributed to the regional increase in biomass burning (veld fires).28 Previous investigations29,30 also ascribed seasonal trends to these factors, but did not specifically point out the significance of CO as a precursor.
Figure 6 shows the monthly mean concentration for BTEX measured in the Highveld. The error bars indicate the maximum and minimum concentrations for the eight stations. No distinct seasonal trend was observed for BTEX in the Highveld. A possible reason for the low levels of ethylbenzene and xylene can be related to their photochemical reactivity, which leads to faster degradation.5
Comparison to Ambient Air Quality Standards
South African National Ambient Air Quality Standards have been established for criteria pollutants, which include SO2, NO2, O3 and benzene.31 A list of the relevant standards is given in Table 1.

By comparison of the annual average concentrations for all gaseous species measured (Figure 2 and Figure 4) with the standards,31 it is evident that levels at all sites were below the annual standards. The results are of particular significance with regard to benzene, which is the only hydrocarbon species included in the new legislation. No national monthly average standards exist for the gaseous pollutant species considered during this investigation. Therefore, no conclusions can be made with regard to the monthly measured values in comparison to a standard.
A comprehensive investigation of benzene levels in the Highveld has been lacking up to now. The results indicate that there were months when values higher than 1.6 ppb (2015 standard31) were measured at individual sites, with the highest value of 6 ppb measured in August 2007 at Standerton. The monthly benzene concentrations are, however, difficult to interpret within the South African legislative context, because no monthly standard for benzene exists in the local legislation. Benzene is currently the only VOC listed as a criteria pollutant in the South African Air Quality Act. It was evident that toluene was significantly higher than benzene in all instances. Toluene also has negative effects on human health11 and is a precursor for O3 and secondary organic aerosols.
Conclusions
The highest NO2, SO2 and BTEX concentrations were measured at sites with significant local sources and/or sites located downwind from major point sources as indicated by dominant air mass movements. The measured O3 concentrations were found to be inverse of the abovementioned species, that is, sites with high concentrations of NO2, SO2 and BTEX exhibited low levels of O3. Witbank had the highest NO2, SO2 and BTEX concentrations, while Carolina had the lowest. A NO2, SO2 and BTEX hotspot was identified in the Witbank area.
NO2 and SO2 peaked during winter while O3 peaked in spring. NO2 and SO2 seasonal concentration variations were ascribed to differences in meteorological conditions, that is, the conditions in winter are more stable than in summer. The O3 peak coincided with a peak in veld fires, which resulted in elevated levels of CO - an important precursor for O3. No distinct seasonal trend was observed for BTEX.
The annual average concentrations for SO2, NO2, O3 and benzene were below the South African annual standards at all sites. Although BTEX measurements were previously conducted in the Highveld,32 the BTEX data presented in this article is the first comprehensive published temporal and spatial data set.
Acknowledgements
We thank Sasol Research & Development and the National Research Foundation, South Africa for financial support. We also thank Mr Ville Vakkari for his guidance with the generation of overlay back trajectories.
References
1. Dabrowski JM, Ashton PJ, Murray K, Leaner JJ, Mason RP. Anthropogenic mercury emissions in South Africa: Coal combustion in power plants. Atmos Environ. 2008;42(27):6620-6626. [ Links ]
2. Freiman MT, Piketh SJ. Air transport into and out of the industrial Highveld Region of South Africa. J Appl Meteorol. 2002;42(7):994-1002. [ Links ]
3. Reisell A, MacDonald C, Roberts P, Arey J. Characterization of biogenic volatile organic compound and meteorology at Azusa during the SCOS97-NARSTO. Atmos Environ. 2003;37(Suppl 2):181-196. [ Links ]
4. Atkinson R. Atmospheric chemistry of VOCs and NOx. Atmos Environ.2000;34(12-14):2063-2101. [ Links ]
5. Parra MA, Elustondo D, Garrigo J. Spatial and temporal trends of volatile organic compounds (VOC) in a rural area of northern Spain. Sci Tot Environ. 2006;370(1):157-167. [ Links ]
6. Hoque RR, Khillare PS, Agarwal T, Shridhar V, Balachandran S. Spatial and temporal variation of BTEX in the urban atmosphere of Delhi, India. Sci Tot Environ. 2007;392(1):30-40. [ Links ]
7. Paoletti E, Bytnerowicz A, Andersen C, Augustatitis A. Impacts of air pollution and climate change on forest ecosystems - emerging research needs. Sci World J. 2007;7(Suppl 1):1-6. [ Links ]
8. Felzer BS, Cronin T, Reilly JM, Melillo JM, Wang X. Impacts of ozone on trees and crops. C R Geoscience. 2007;339:784-798. [ Links ]
9. Bernstein JA, Alexis N, Barnes C, et al. Health effects of air pollution. J Allergy Clin Immunol. 2004;114(5):1116-1123. [ Links ]
10. Kampa M, Castanas E. Human health effects of air pollution. Environ Pollut. 2007;151(2):362-367. [ Links ]
11. Ma W, Shaffer KM, Pancrazio JJ, et al. Toluene inhibits muscarinic receptor-mediated cytosolic Ca2+ responses in neutral precursor cells. Neurotoxicol. 2002;23(1):61-68. [ Links ]
12. Notice of intention to declare the Highveld Priority Area in terms of Section 18(1) of the National Environmental Management: Air Quality Act, 2004. Act no. 39 of 2004. Proclamation no. 396, 2007. [ Links ]
13. Toenges-Schuller N, Stein O, Rohrer F, et al. Global distribution pattern of anthropogenic nitrogen oxide emissions: Correlation analysis of satellite measurements and model calculations. J Geophys Res. 2006;111(D05312):1-15. [ Links ]
14. Martins JJ, Dhammapala RS, Lachmann G, Galy-Lacaux C, Pienaar JJ. Long-term measurements of sulphur dioxide, nitrogen dioxide, ammonia, nitric acid and ozone in southern Africa using passive samplers.S Afr J Sci. 2007;103(7-8):336-342. [ Links ]
15. Carmichael GR, Ferm M, Thongvoonchoo N, et al.Measurements of sulfur dioxide, ozone and ammonia concentrations in Asia, Africa and South America using passive samplers. Atmos Environ. 2003;37(9-10):1293-1308. [ Links ]
16. Josipovic M. Acidic deposition emanating from the South African Highveld - a critical levels and critical loads assessment. PhD thesis, Johannesburg, University of Johannesburg, 2009. [ Links ]
17. Dhammapala RS. Use of diffusive samplers for the sampling of atmospheric pollutants. MSc thesis, Potchefstroom, North West University, 1996. [ Links ]
18. United States Environmental Protection Agency. Compendium methods for the determination of toxic organic compounds in ambient air. 2nd ed. Compendium method TO-17. Determination of volatile organic compounds (VOCs) in ambient air using active sampling unto sorbent tubes. Cincinnati: United States Environmental Protection Agency; 1999. [ Links ]
19. Skov H, Lindskog A, Palmgren F, Christensen CS. An overview of commonly used methods for measuring benzene in ambient air. Atmos Environ. 2001;35(Suppl 1):141-148. [ Links ]
20. Hybrid Single-Particle Lagrangian Integrated Trajectory (HYSPLIT), Version 4.8, [cited 2009 Dec 4]. Available from: http://www.arl.noaa.gov [ Links ]
21. MATLAB 7.8.0, Mathworks, Massachusetts, USA, [cited 2009 Nov 21]. Available from: http://www.mathworks.com [ Links ]
22. Valente RJ, Imhoff RE, Tanner RL, et al. Ozone production during an urban air stagnation episode over Nashville, Tennessee. J Geophys Res. 1998;103(D17):22555-22568. [ Links ]
23. Martins JJ. Concentrations and deposition of atmospheric species at regional sites in southern Africa. PhD thesis, Potchefstroom, North West University, 2008. [ Links ]
24. Seinfeld JH, Pandis SP. Atmospheric chemistry and physics: From air pollution to climate change. New York: Johan Wiley & Sons, 1998; p. 66259. [ Links ]
25. Garstang M, Tyson PD, Swap R, Edwards M, Kallberg P, Lindesay JA. Horizontal and vertical transport of air over southern Africa. J Geophys Res. 1996;101(D19):23721-23736. [ Links ]
26. Jacob DJ. Heterogeneous chemistry and tropospheric ozone. Atmos Environ. 2000;34:2131-2159. [ Links ]
27. Laakso L, Laakso H, Aalto PP, et al. Basic characteristics of atmospheric particles, trace gases and meteorology in a relatively clean southern African savannah environment. Atmos Chem Phys. 2008;8:4823-4839. [ Links ]
28. Swap RJ, Annegarn HJ, Suttles JT, et al. Africa burning: A thematic analysis of the South African Regional; Science Initiative (SAFARI 2000). J Geophys Res. 2003;108(D13):8465-8480. [ Links ]
29. Zunckel M, Venjonoka K, Pienaar JJ. Surface ozone over South Africa: Synthesis of monitoring results during the cross border air pollution impact assessment project. Atmos Environ. 2004;38:6139-6147. [ Links ]
30. Diab RD, Thompson AM, Mari K, Ramsay L, Coetzee GJR. Troposheric ozone climatology over Irene, South Africa, from 1990 to 1994 and 1998 to 2002. J Geophys Res. 2004;109(D20301):1-11. [ Links ]
31. National Environmental Management Air Quality Act of 2004 section 63. Government Gazette Republic of South Africa. 534:8-9. c2004 [cited 2009 Dec 24]. Available from: http://www.info.gov.za [ Links ]
32. Van der Walt HJ. The impact of hydrocarbon emissions on regional air quality in a South African metropolitan area. PhD thesis, Potchefstroom, North West University, 2009. [ Links ]
 Correspondence to:
Correspondence to:
Pieter van Zyl
Postal address: School of Physical and Chemical Sciences, North West University, Potchefstroom campus
Potchefstroom 2520
Private Bag X6001, Box 179, South Africa
email: pieter.vanzyl@nwu.ac.za
Received: 28 May 2009
Accepted: 21 Nov. 2010
Published: 28 Jan. 2011














