Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.107 n.1-2 Pretoria Jan./Feb. 2011
http://dx.doi.org/10.4102/sajs.v107i1/2.367
RESEARCH ARTICLES
Fusarium spp. and levels of fumonisins in maize produced by subsistence farmers in South Africa
Edson NcubeI; Bradley C. FlettI; Cees WaalwijkII; Altus ViljoenIII
IAgricultural Research Council-Grain Crops Institute, Potchefstroom, South Africa
IIPlant Research International BV, Wageningen, the Netherlands
IIIDepartment of Plant Pathology, University of Stellenbosch, South Africa
ABSTRACT
Fusarium spp. produce fumonisins - mycotoxins that are of importance to maize production in South Africa. Fumonisins have been associated with human oesophageal cancer and cause various diseases in animals that are of concern to the animal feed industry. Maize samples, collected from subsistence farm fields in the Eastern Cape, KwaZulu-Natal, Limpopo and Mpumalanga provinces of South Africa during the 2006 and 2007 growing seasons, were analysed for Fusarium spp. and contamination with fumonisins. Fusariumverticillioides was the most common Fusarium species in maize followed by F. subglutinans and F. proliferatum. Levels of contamination with fumonisins ranged from 0 µg/g to 21.8 µg/g, depending on the region where samples were collected. Levels of fumonisins were highest in northern KwaZulu-Natal (Zululand) where 52% and 17% of samples collected in 2006 and 2007, respectively, exceeded 2 µg/g. Regression analyses showed a positive correlation between fumonisin-producing Fusarium spp. determined by real-time polymerase chain reaction and concentration of fumonisins (r = 0.93). Many samples from Zululand, and some from Mokopane (Limpopo) and Lusikisiki (Eastern Cape), contained fumonisins at levels well above the maximum levels of 2 µg/g set by the Food and Drug Administration (USA) and therefore also the limit of 1 µg/g set by the European Union for food intended for direct human consumption. Regulations governing contamination of grain with fumonisins are not yet implemented in South Africa. The high incidence of fumonisins in subsistence farming systems indicates the need for awareness programmes and further research.
Introduction
Maize (Zea mays L.) is grown in two production systems in South Africa: intensive commercial farming systems and resource-poor subsistence farming systems. Average production amounts to approximately 9.2 million tonnes per annum, of which 8 million tonnes is utilised in the country as food and fodder.1 Depending on the surplus available, the remainder is exported to neighbouring countries.2 Because maize quality and yield is important to commercial producers in South Africa, they implement strategies that reduce losses. Subsistence farmers, however, lack the required resources to ensure production of quality grain from field planting through to consumption.3 With an insufficiency of hybrid seed, fertilisers and pesticides, their yield and product quality is often severely affected by poor soil fertility, fungal infections and pest damage.4
Over 600 000 households depend on subsistence farming in South Africa.5 With maize being a staple food, the average intake per person per day may exceed 300 g.6 The quality of maize consumed thus determines the quality of life. Maize produced in subsistence farming systems, however, is often affected by pre- and post-harvest damage, of which fungal infection can be considered one of the most significant problems. Most subsistence farmers plant their fields with seed retained from the previous harvest,4 thereby increasing the risk of systemic infection by plant pathogens.7 Late planting and practising maize monoculture also increases fungal inoculum and pest damage, thereby resulting in increased fungal infection of crops.8 Agricultural practices, such as retaining crop residues on the soil surface, may further increase the severity of diseases such as stem, ear and root rot in the following season.9
One of the most important fungal pathogens affecting maize in South Africa is Fusarium verticillioides (Sacc.) Nirenberg (syn = F. moniliforme Sheldon), a ubiquitous facultative endophyte found in most maize-producing countries of the world.10 Fusarium verticillioides can constitute up to 95% of all Fusarium strains recovered from maize fields in African countries.11 This fungus produces fumonisins, toxigenic secondary metabolites that are well recognised for their cancer-promoting effects.12 Other Fusarium spp. that produce mycotoxins in maize include F. graminearum Schwabe, that produces zearalenone and deoxynivalenol,13 F. proliferatum (Matsushima) Nirenberg, that produces fumonisins13 and F. subglutinans (Wollenw. and Reinking) P.E. Nelson, Toussoun and Marasas,14 a producer of moniliformin.15
Fumonisins have been associated with high rates of human oesophageal cancer worldwide16,17 and with increased incidences of neural tube defects in infants of mothers consuming maize-based products contaminated with fumonisins.18 Fumonisins are also toxic to livestock.19,20,21 Increased levels of fumonisins in mouldy maize kernels have been previously linked to the high incidence of human oesophageal cancer in several districts in the Transkei region of South Africa.17 Little, however, is known about contamination of maize with Fusarium spp. and fumonisins in other rural areas of the country. The aim of this study, therefore, was to determine the Fusarium spp. and levels of fumonisins associated with maize in subsistence farming systems in South Africa.
Materials and methods
Field sampling
Maize samples were collected from randomly selected subsistence farming localities in the Eastern Cape, KwaZulu-Natal, Limpopo and Mpumalanga provinces of South Africa (Table 1). These post-harvest samples were taken during storage. Because of different climatic conditions in the northern (subtropical coastal) and southern (temperate coastal) parts of KwaZulu-Natal, localities in KwaZulu-Natal were split into the two regions: southern and northern KwaZulu-Natal (Zululand). In total, 147 and 114 maize samples, each approximately 1.5 kg, were collected from different farmers in the same district in the 2006 and 2007 growing seasons, respectively. These samples were in storage for less than two months. Shelled maize or maize ears were placed in cloth bags to prevent condensation that might promote fungal growth and were labelled with the source or locality of the sample and the cultivar planted. Global positioning system (GPS) co-ordinates were recorded at each sampling point. The collected samples were then stored in a cold room at 4 ºC and 45% relative humidity until assayed.
Climatic data
Monthly rainfall and temperature data for weather stations closest to the sampling localities were obtained from the website of the Agricultural Research Council's Institute of Soil Climate and Water in Pretoria. Climatic data, stretching from October of the previous year to May in the year that maize samples were collected, were considered. These specific dates were chosen as they represent the growing season of maize in South Africa.
Isolation and enumeration of Fusarium spp.
Maize kernels were surface-sterilised by dipping them once in 70% ethanol, soaking them for 3 min in 1.6% NaOCl solution and rinsing them three times in sterile distilled water. The kernels were then plated out on Van Wyk agar, a Fusarium selective medium22 in Petri dishes (90 mm in diameter). Each Petri dish contained 4 kernels, equidistant from each, and a total of 50 Petri dishes were used to plate 200 seeds. After 7 days of incubation at 25 ºC, developing Fusarium colonies were identified morphologically to species level.23
Quantitative detection of fumonisin-producing Fusarium spp.
Maize samples were analysed for Fusarium spp. that produce fumonisins using quantitative real-time polymerase chain reaction (qPCR). A Cyclotec sample mill (Foss Tecator, Hoganas, Sweden) was used for grinding maize samples into a fine powder such that more than 75% of the ground material passed through a 20-mesh sieve. DNA was then isolated from 20 mg of each sample using Qiagen DNeasy Plant Mini Kits (Cat 69106, Qiagen, Hilden, Germany). The TaqMan method was used to detect Fusarium spp. that produce fumonisins using primers and probes designed for the polyketide synthase gene fum1.24 qPCR was performed using a MicroAmp Optical 96-well reaction plate and MicroAmp Optical Caps (Applied Biosystems, Foster City, USA). An ABI Prism 7700 Sequence Detection System (Applied Biosystems) was used to perform the PCR and assess fluorescence. Each amplification reaction consisted of 2 µl of DNA preparations, 1 x real-time PCR buffer (Applied Biosystems), 5 mM MgCl2, 83 nM of the FAM-labelled FUM-probe, 1.5 U of Hot Goldstar DNA polymerase (Eurogentec, Seraing, Belgium) and 333 nM of forward and reverse primers for the target DNA (Taqfum-2F in combination with Vpgen-3R, VertFum-3R or ProFum-3R). As an internal control, 100 pg of potato leaf roll virus (PLRV) DNA, forward primer PLRV-F and reverse primer PLRV-R (both at 333 nM) were included in the reaction along with 83 nM of the VIC-labelled PLRV probe.24
Analysis for fumonisins
Levels of fumonisins were quantified using the Veratox enzyme-linked immunosorbent assay (ELISA) quantitative fumonisin 5/10 test kit (Neogen Corp, Lansing, MI, USA) according to the manufacturer's instructions. Results above 6 µg/g were extrapolated from the standard curve (r = 0.99) using Veratox® software.25 An inter-laboratory comparison was done with the Division of Toxicology, Onderstepoort Veterinary Institute, South Africa to validate the method. Each analysis was repeated three times to determine reproducibility of the results.
Statistical analyses
Simple linear regression on Statgraphics 5 Plus26 (Manugistics Inc, Rockville, MD, USA) was used to determine the relationship between levels of fumonisins quantified by ELISA and target DNA of Fusarium spp. that produce fumonisins for the 2006 and 2007 seasons, and for both seasons combined. The correlation was done to determine whether the qPCR technique was a reliable method in detecting Fusarium spp. that produce fumonisins in maize as compared to morphological identification. Temperature and rainfall data were also correlated to the production fumonisins using simple linear regression analyses on Statgraphics 5 Plus.26
Results
Isolation and enumeration of Fusarium spp.
Three Fusarium spp. (Section: Liseola) were isolated from maize kernels collected from subsistence farmers' fields in South Africa. Fusarium verticillioides was the dominant Fusarium species in maize collected in Limpopo and Zululand and F.subglutinans was the dominant species in the Eastern Cape and Mpumalanga provinces (Table 1). Less than 5% of all maize kernels were infected by F. proliferatum, except those collected from Eshowe in Zululand, which yielded a mean infection level of 20.1% in 2006 (Table 1).
Quantitative determination of fumonisin-producing Fusarium spp.
Quantitative PCR results supported the seed isolation data, with Fusarium spp. that produce fumonisins being found in maize produced in Zululand at far greater levels than in any other province in both 2006 and 2007 (Table 1). The average amount of fungal DNA found in maize from Zululand was more than three times greater than that found in any other province (Table 1). Fusarium spp. that produce fumonisins were absent in maize kernels from Mqanduli (Eastern Cape) and several localities in Mpumalanga in 2006; Libode, Ngqeleni and Whittlesea in 2007 and Qunu (Eastern Cape) in both seasons (Table 1).
Analysis for fumonisins
Samples from Zululand and, to a lesser extent, those from Limpopo were contaminated with higher levels of fumonisins than those collected in the other provinces (Table 1). Levels of fumonisins in samples collected at Manguzi, Mbazwane, Pongola, Ulundi and Vryheid were generally in excess of 2 µg/g in 2006 (Table 1a). Maize samples from Manguzi had consistently high levels of fumonisins in both seasons. In Limpopo, it was only the samples collected in Mokopane and Venda that contained fumonisins at levels above 2 µg/g (Table 1). None of the samples collected in Mpumalanga were contaminated with more than 2 µg/g fumonisins. In the Eastern Cape, where contamination of maize with fumonisins has been reported previously,6,10,17 only samples collected from Lusikisiki had contamination levels above 2 µg/g in both 2006 and 2007 (Table 1).
Climatic data
The lower average daily temperatures were recorded in the mountainous district of Ngqeleni in the Eastern Cape while the higher average temperatures were recorded in the inland areas of Limpopo and Zululand (Table 1). Rainfall data in KwaZulu-Natal, Limpopo and Mpumalanga provinces varied notably among regions (Table 1).
Correlation between Fusarium spp., levels of fumonisins and climatic data
A significant positive correlation was obtained between target DNA of Fusarium spp. that produce fumonisins and levels of fumonisins for the 2006 (r = 0.74, p < 0.05) and 2007 (r = 0.93, p < 0.05) growing season (Figure 1) and for the combined seasons (r = 0.80, p < 0.05). A poor correlation, however, was obtained between F. verticillioides quantified by plating and target DNA of Fusarium spp. determined by qPCR (r = 0.14, p < 0.05). There was a significant positive correlation between the combined occurrence of F. verticillioides and F. proliferatum and fumonisins (r = 0.39, p > 0.05).

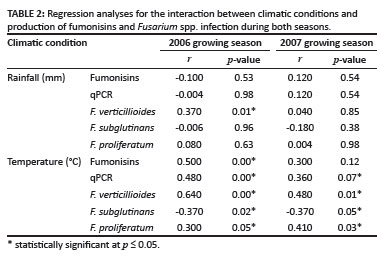
There was no correlation between temperature and level of fumonisins (r = 0.3, p > 0.1) in 2007. But there was a significant correlation between temperature and target DNA of Fusarium spp. determined by qPCR as well as Fusarium spp. quantified by plating in 2006 and in 2007 (Table 2). There was no correlation between rainfall data and the level of fumonisins in either season (Table 2).
Discussion
Levels of fumonisins in some samples found in Venda and Mokopane (Limpopo), Lusikisiki (Eastern Cape) and Mbazwane, Jozini, Pongola and Manguzi (Zululand) far exceeded the maximum levels of 2 µg/g set by the US Food and Drug Administration in the USA27 and the 1 µg/g set by the European Union28 for food intended for direct human consumption. This was possibly as a result of local agricultural practices, such as lack of fungus disease control, planting dates, harvesting dates, storage, crop residue disposal, land tillage methods, crop rotation, seed sources and maize stalk borer control, that might have promoted the growth of Fusarium spp. that produce fumonisins. In this study, levels of fumonisins in Butterworth and Centani and other areas in the Eastern Cape were found to be below 0.5 µg/g, possibly because of the provision of hybrid seed in the former Transkei by the Provincial Department of Agriculture.4 Because subsistence farmers produce maize for their own consumption, they could be at higher risk of exposure to fumonisins and concomitant mycotoxicoses in their diet than people living in urban areas of South Africa.
The high incidence of F. verticillioides and F.subglutinans in 2006 and 2007 can partly be explained by existing environmental conditions in the local rural maize production areas. F. verticillioides is known to proliferate in warm, humid regions,9 similar to those reported for Zululand, Venda in Limpopo and Lusikisiki and Engcobo districts in the Eastern Cape. F.subglutinans, however, is known to multiply more rapidly in temperate climates29 and was the dominant species found in the cooler districts of Mpumalanga and the Eastern Cape, as well as some mountainous districts in southern KwaZulu-Natal. Subsistence farmers in these areas might be at risk to moniliformin, which is the primary mycotoxin produced by F. subglutinans.15 As was reported in related studies on Fusarium spp. in maize,11 F. proliferatum proved to be a minor coloniser of maize kernels in subsistence farmer fields, except those collected from Eshowe in Zululand, possibly as a result of cross-over infections from sugarcane grown in and around Eshowe.30 High levels of co-infections of maize with F. verticillioides and F. subglutinans also occurred in all provinces, and subsistence farmers in these areas could be simultaneously exposed to both fumonisins and moniliformin.
More pronounced infections of maize in Zululand by F. verticillioides might have been caused by farmers planting seed of open pollinated maize varieties retained from the previous harvest,4 as such seed could lead to increased systemic infections and concomitant production of fumonisins.7 These infections could also have been caused by crop residues left on the lands, late harvesting and by practising maize monoculture.4 These practices are more predominant in Zululand than anywhere else in South Africa. The low levels of F. verticillioides found in the Eastern Cape and southern KwaZulu-Natal could be a result of cooler temperatures prevailing in those regions. Colonisation of maize kernels with Fusarium spp. that produce fumonisins sometimes differed considerably within and between districts in the same region, which could be attributed to different farming practices that were followed by individual farmers.
A positive correlation was found between the target DNA of Fusarium spp. that produce fumonisins and levels of fumonisins. However, there was a poor correlation between Fusarium spp. determined by qPCR and F. verticillioides quantified by plating on Fusarium-selective medium. This study, therefore, shows that fungal biomass, as measured by qPCR, shows a significant correlation with contamination of maize with fumonisins. This is in agreement with several studies, such as that of Waalwijk et al.31
The occurrence of fumonisins in subsistence farmer crops has previously been reported for maize produced in the Eastern Cape.6,17 This study, however, is the first to show that fumonisins are produced in maize in all the subsistence production regions of South Africa. Implementation of disease-management practices, such as planting of regionally adapted maize varieties, reducing insect damage, early harvesting and discarding mouldy kernels and farmer education in rural areas can result in the reduction of mycotoxin contamination. This is particularly important because subsistence farmers rely on their maize produce as the primary source of food and income, irrespective of its quality.
Acknowledgements
Funding from the Maize Trust and Agricultural Research Council is greatly acknowledged. We thank Ms Gugu Khali, Mr Moses Ramusi and extension officers of the Department of Agriculture in the provinces visited for assistance with sample collection. We also would like to thank Ineke de Vries (Plant Research International, Wageningen, the Netherlands) for assistance with qPCR and Belinda Janse van Rensburg for assistance with ELISA analysis. Thanks are also due to Maria Mahlobo for laboratory assistance.
References
1. Crop Estimates Committee. Calculated area and crop production figures of maize and sorghum: 2006/07 production season [document on the Internet]. c2007 [cited 2009 Dec 14]. Available from: http://www.nda.agric.za/doaDev/cropestimates/Calculated%20final%20crop%20of%20summer%20crops%20Nov%202007.doc [ Links ]
2. Northern Cape Department of Agriculture and Land Reform. Maize industry: Situation analyses, market indicators and outlook for 2008 season [document on the Internet]. c2007 [cited 2009 Dec 14]. Available from: http://www.agrinc.gov.za/docs/Maize%20Outlook%202008.pdf [ Links ]
3. Bankole SA, Adebanjo A. Mycotoxins in food in West Africa: Current situation and possibilities of controlling it. Afr J Biotech. 2003;2:254-263. [ Links ]
4. Ncube E. Mycotoxin levels in subsistence farming systems in South Africa [MSc dissertation]. Stellenbosch: University of Stellenbosch; 2008. [ Links ]
5. Watkinson E, Makgetla N. South Africa's food security crisis [document on the Internet]. c2002 [cited 2007 Apr 19]. Available from: http://www.sarpn.org.za/documents/0000077/P93_safscrisis.pdf [ Links ]
6. Shephard GS, Marasas WFO, Burger H-M, et al. Exposure assessment for fumonisins in the former Transkei region of South Africa. Food Add Contam. 2007;24:621-629. [ Links ]
7. Wilke AL, Bronson CR, Tomas A, Munkvold GP. Seed transmission of Fusarium verticillioides in maize plants grown under three different temperature regimes. Plant Dis. 2007;91:1109-1115. [ Links ]
8. Meissle M, Mouron P, Musa T, et al. Pests, pesticide use and alternative options in European maize production: Current status and future prospects. J Appl Entomol. 2010;134:357-375. [ Links ]
9. Munkvold GP, Desjardins AE. Fumonisins in maize. Can we reduce their occurrence? Plant Dis. 1997;81:556-565. [ Links ]
10. Marasas WFO. Fumonisins: History, world-wide occurrence and impact. Adv Exp Med Biol. 1996;392:1-17. [ Links ]
11. Leslie JF. Diversity in toxigenic Fusarium species in Africa. Paper presented at: Myco-Globe 2005. Proceedings of the Conference on Reducing Impact of Mycotoxins in Tropical Agriculture with Emphasis on Health and Trade in Africa; 2005 Sept 13-26; Accra, Ghana. [homepage on the Internet]. c2005 [cited 2010 Oct 18]. Available from: http://www.ispa.cnr.it/mycoglobe/index.php?page=result&id=18 [ Links ]
12. Gelderblom WCA, Jaskiewicz K, Marasas WFO, et al. Fumonisins - novel mycotoxins with cancer promoting activity produced by Fusarium moniliforme. Appl Environ Microbiol. 1988;54:1806-1811. [ Links ]
13. Shephard GS. Mycotoxins in the context of food risks and nutrition issues. In: Barug A, Bhatnagar D, van Egmond HP, van der Kamp JW, van Osenbruggen WA, Visconti A, editors. The mycotoxin factbook. Wageningen: Wageningen Academic Publishers, 2006; p. 21-36. [ Links ]
14. Summerell BA, Salleh B, Leslie JF. A utilitarian approach to Fusarium identification. Plant Dis. 2003;87:117-128. [ Links ]
15. Thiel PG, Meyer CJ, Marasas WFO. Natural occurrence of moniliformin together with deoxynivalenol and zearalenone in Transkeian corn. J Agric Food Chem. 1982;30:308-317. [ Links ]
16. Chu FS, Li GY. Simultaneous occurrence of fumonisin B1 and other mycotoxins in mouldy corn collected from the People's Republic of China in regions with high incidences of oesophageal cancer. Appl Environ Microbiol. 1994;60:847-852. [ Links ]
17. Rheeder JP, Marasas WFO, Thiel PG, Sydenham EW, Shephard GS, Van Schalkwyk DJ. Fusarium moniliforme and fumonisins in corn in relation to human oesophageal cancer in Transkei. Phytopathology. 1992;82:353-357. [ Links ]
18. Missmer SA, Suarez L, Felkner M, et al. Exposure to fumonisins and the occurrence of neural tube defects along the Texas-Mexico border. Environ Health Persp. 2006;114:237-241. [ Links ]
19. Glenn AE. Mycotoxigenic Fusarium species in animal feed. Anim Feed Sci Technol. 2007;137:213-240. [ Links ]
20. Kellerman TS, Marasas WFO, Thiel PG, Gelderblom WCA, Cawood ME, Coetzer JA. Leukoencephalomalacia in two horses induced by oral dosing of fumonisin B1. Onderst J Vet Res. 1990;57:269-275. [ Links ]
21. Haschek WM, Gumprecht LA, Smith G, Tumbleson ME, Constable PD. Fumonisin toxicosis in swine: An overview of porcine pulmonary edema and current perspectives. Environ Health Persp Suppl. 2001;109:251-257. [ Links ]
22. Van Wyk PS, Scholtz DJ, Los O. A selective medium for the isolation of Fusarium spp. from soil debris. Phytophylactica. 1986;18:67-69. [ Links ]
23. Leslie JF, Summerell BA. The Fusarium laboratory manual. Ames: Blackwell Publishing; 2006. [ Links ]
24. Waalwijk C, Koch S, Ncube E, et al. Quantitative detection of fumonisin-producing Fusarium spp. and its correlation with fumonisin content in maize from South African subsistence farmers. World Mycotoxin J. 2008;1:39-47. [ Links ]
25. Neogen Corporation [DVD]. Veratox® Software for Windows. Log/Logit and Single Test Format. Version 3.0.1 EU. Lansing, MI, USA. 2008. [ Links ]
26. Manugistics Incorporated. Statgraphics® 5 Plus. Rockville, MD, USA. 2000. [ Links ]
27. Food and Drug Administration (FDA). Guidelines for industry: Fumonisin levels in human foods and animal feeds. Rockville: FDA; 2001. [ Links ]
28. European Commission (EC). Commission Regulation (EC) No 1126/2007 of 28 September 2007 amending Regulation (EC) No 1881/2006 setting maximum levels for certain contaminants in foodstuffs as regards Fusarium toxins in maize and maize products. Off J Eur Union. 2007;L255:14-17. [ Links ]
29. Marasas WFO, Kriek NPJ, Wiggins VM, Steyn PS, Towers DK, Hastie TJ. Incidence, geographic distribution, and toxicigenicity of Fusarium species in South Africa. Phytopathology. 1979;69:1181-1185. [ Links ]
30. McFarlane SA, Govender P, Rutherford RS. Interactions between Fusarium species from sugarcane and the stalk borer Eldana saccharina (Lepidoptera: Pyralidae). Ann Appl Biol. 2009;155:349-359. [ Links ]
31. Waalwijk C, Van der Heide R, De Vries PM, et al. Quantitative detection of Fusarium species in wheat using TaqMan. Eur J Plant Path. 2004;110:481-494. [ Links ]
 Correspondence to:
Correspondence to:
Edson Ncube
Postal address: Agricultural Research Council-Grain Crops Institute
Potchefstroom 2520
Private Bag X1251, South Africa
email: ncubee@arc.agric.za
Received: 21 Jul. 2010
Accepted: 18 Nov. 2010
Published: 28 Jan.2011














