Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Science
versão On-line ISSN 1996-7489
versão impressa ISSN 0038-2353
S. Afr. j. sci. vol.106 no.11-12 Pretoria Nov./Dez. 2010
http://dx.doi.org/10.4102/sajs.v106i11/12.423
RESEARCH LETTER
Hybrid wildebeest (Artiodactyla: Bovidae) provide further evidence for shared signatures of admixture in mammalian crania
Rebecca R. AckermannI; James S. BrinkII,III; Savvas VrahimisIV; Bonita de KlerkV
IDepartment of Archaeology, University of Cape Town, South Africa. email: becky.ackermann@uct.ac.za
IIFlorisbad Quaternary Research Department, National Museum, Bloemfontein, South Africa
IIICentre for Environmental Management, University of the Free State, Bloemfontein, South Africa
IVBiodiversity Planning & Research Component, Department of Economic Development, Tourism & Environmental Affairs, Bloemfontein, South Africa
VInstitute for Human Evolution, School of Geosciences, University of the Witwatersrand, Johannesburg, South Africa
ABSTRACT
The genus Connochaetes, Lichtenstein, 1814, contains two extant species, the blue wildebeest (C. taurinus, Burchell, 1823) and the black wildebeest (C. gnou, Zimmermann, 1780). In recent years, forced sympatry in confined areas within South Africa has led to interbreeding between these taxa and to fertile hybrid offspring. Here we report on a series of cranial characteristics of a hybrid wildebeest population culled at Spioenkop Dam Nature Reserve, KwaZulu-Natal, South Africa. Dental, sutural and horn morphological anomalies occur at high frequency within these animals. Similar cranial morphological anomalies have been shown in other mammalian hybrids and this study provides further evidence that such anomalies may characterise hybridisation more broadly across phylogenetically divergent mammalian groups, although the anomalies appear to differ in their expression across taxa. An increased ability to identify hybrids may also have important applications in the conservation of the endemic black wildebeest.
Keywords: Connochaetes; conservation; cranial anomalies; dental anomalies; gene flow; hybridisation; South Africa
INTRODUCTION
The genus Connochaetes, Lichtenstein, 1814, is part of the family Bovidae (Order: Artiodactyla), which includes antelope, cattle, goats and other even-toed horned ungulates. Within this genus, there are two extant species, the blue wildebeest (or the brindled gnu, C. taurinus, Burchell, 1823) and the black wildebeest (or white-tailed gnu, C. gnou, Zimmermann, 1780). Molecular analyses and estimates from the fossil record indicate that these two species diverged around one million years ago1,2,3,4,5 into northern and southern forms.2,6 Following this initial divergence, the lineage leading to the modern blue wildebeest changed little morphologically from the ancestral form, while the southerly black wildebeest adapted to an open grassland niche, during which time many morphological changes accumulated.6
Today, five subspecies of the blue wildebeest occur on the African continent,7 although C. t. taurinus is the only subspecies living in South Africa. The black wildebeest is endemic to South Africa and has no extant subspecies.6 Superficial morphological separation of the two species of wildebeest is based primarily on horn curvature and pelage, especially the colour and length of the fur on the tail, as well as facial, neck and limb length and overall size (Table 1). The blue wildebeest is the larger of the two species, with males ~130 cm tall at the shoulder8 and weighing between 210 kg and 260 kg.9 They are dark grey in colour (although they can have a silvery blue sheen), with brindled stripes and long hair on their manes, chin, throat and tails.8 In addition, the blue wildebeest has a large, long head, with horns that sweep laterally and slightly downwards, then curve up at the ends and sometimes back towards the skull.6 The black wildebeest, in contrast, is smaller (males ~120 cm, ~170 kg),10,11,12 is brown in colour with cream-to-black manes and a very long cream-tipped tail, and has characteristic horns with expanded bases that curve primarily forwards and downwards before curving up at the tips.6
It is uncertain whether the divergent evolutionary lineages that led to these two forms were geographically separated at the time of speciation of C. gnou, but following their initial divergence, climatic and environmental conditions allowed the sympatric occurrence of the two species of wildebeest in southern Africa.6 Today, the blue wildebeest occurs in a variety of savannah habitat types, including open woodland and grassland, while the black wildebeest is restricted to the open grasslands of the central inland plateau.6,10,13,14 Although their distributions overlapped during the Pleistocene15,16 and into historic times,14 habitat preferences and behavioural differences have kept them largely separate.6,17
Yet despite these different habitat preferences, these two species have come into frequent contact in recent years on many private game farms and on government-controlled reserves throughout South Africa.18 This forced sympatry in confined areas has led to interbreeding, and the production of fertile hybrids.8,19 Indeed, a substantial proportion of the black wildebeest population in South Africa may contain a significant number of introgressed genes from the blue wildebeest,20 although the actual extent of admixture between the two taxa is not known.21 Hybrids between these two species that have been studied phenotypically have been described as highly variable, displaying traits of both species, as well as intermediate traits (Table 1).8,22
Here we report on a series of anomalous cranial morphological characteristics displayed in a hybrid (blue x black) wildebeest population culled at Spioenkop Dam Nature Reserve, KwaZulu-Natal, South Africa. These hybrids were identified initially from their external phenotype,23 (Rushworth I 2000, personal communication, March 08), based primarily on horn morphology and pelage, and have been reported in detail elsewhere.22 The primary goal of this paper is to briefly describe unusual dental and sutural anomalies, as well as other unusual morphological traits present in these hybrids. Similar anomalies have been demonstrated in the skeletons of primate hybrids between lineages that have diverged over a comparable time frame (i.e. Pleistocene),24,25,26 as well as a handful of other mammals,27,28 and this study provides further evidence that cranial morphological anomalies characterise mammalian hybridisation more broadly. Because mammalian hybrids are also known to be polymorphic,29 a goal of this study was to qualitatively assess the range of phenotypic variation in the hybrid sample. These results may assist in identification of regions in South Africa with high versus low levels of hybridisation, as it has been shown that the identification of such anomalies allows detection of hybridisation on the landscape.25 This has important implications for conservation of the rarer, endemic black wildebeest.
MATERIALS AND METHODS
All examined hybrid wildebeest crania (n = 13) are housed at the Florisbad Quaternary Research Station of the National Museum, Bloemfontein (NMB), and are listed in Table 2. The sample is dominated by male adults, although there are two females and two sub-adult individuals. These crania were prepared and curated at the Florisbad Quaternary Research Station after a large culling of wildebeest hybrids in the Spioenkop Dam Nature Reserve near the northern Drakensburg Mountains of KwaZulu-Natal province, South Africa. The present study sample was selected in the field at the time of the culling, based on author observations of deviations from the known black wildebeest phenotype.22,23 Further details of the examined cranial sample, as well as its associated postcranial remains, have been reported previously.22
Individual hybrid wildebeest crania were examined and scored for the presence of a suite of qualitative (non-metric) cranial traits, with emphasis on dental and sutural morphometric anomalies comparable to what is known to be present in hybrid primates.24,25,26 Qualitative cranial traits scored included: supernumerary teeth, extra sutures or ossicles in the maxillary or premaxillary region, rotated teeth and dental crowding.26 The expression of supernumerary teeth was also recorded, as this has been shown to vary across mammalian hybrids.26,27 Additionally, the crania were examined for other evidence of developmental abnormalities or abnormal trait variation.
Data were compared to known trait variation in the blue and black wildebeest.6 The comparative unhybridised sample included 20 blue wildebeest (11 male, 9 female) and 20 black wildebeest (10 male, 10 female). The black wildebeest sample included only historic and subfossil specimens, which predate the present hybridisation problem, while the blue wildebeest sample included both historic and modern specimens.6,17,22
RESULTS AND DISCUSSION
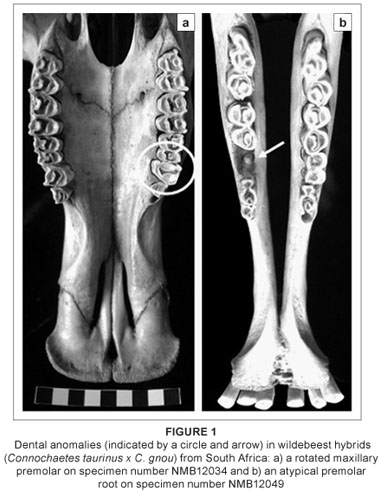
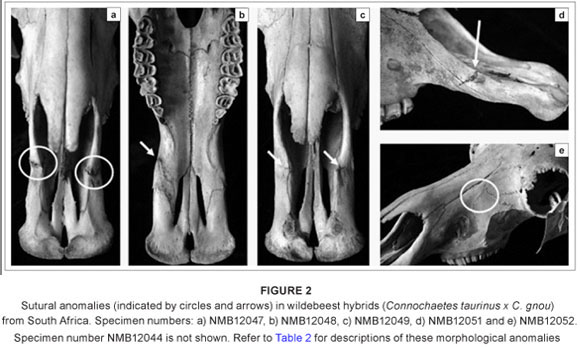
The dental and sutural morphological anomalies found in the 13 hybrid wildebeest are listed and briefly described in Table 2. None of the hybrids has supernumerary teeth, although one adult male individual (NMB12043) has a unilateral rotated premolar (Figure 1a). The only other dental anomaly is an unusual premolar root (Figure 1b), also in an adult male. This individual is old and it is possible that the root was damaged as a result of dental attrition. Sutural anomalies are more common in the hybrid wildebeest; six individuals (46%) had sutural anomalies, five of which were additional sutures in the premaxillary region. These sutural anomalies occur in both males and females. The additional premaxillary sutures do not represent sutures that are seen earlier in ontogeny, but are instead new atypical variants. It is possible that these premaxillary sutures provide some disadvantage in terms of fitness by altering the physical properties of the snout in these hybrids. No comparable dental or sutural anomalies were found in the non-hybridised blue wildebeest (n = 20) or black wildebeest (n = 20) samples,6,22 suggesting that their frequency across non-hybridised wildebeest is low or absent. Moreover, the high frequency of sutural morphological anomalies seen in the hybrids greatly exceeds 'normal' levels of atypical qualitative trait variation in mammals (generally less than 5%), and is comparable to what was observed for dental anomalies in known-pedigree hybrid male F1 baboons (50%)26 and for sutural anomalies in an identified hybrid zone within eastern lowland gorillas (45%).25 There is also considerable variation in the expression of these sutures across the hybrids (unilateral versus bilateral, superior versus inferior; see Figure 2).
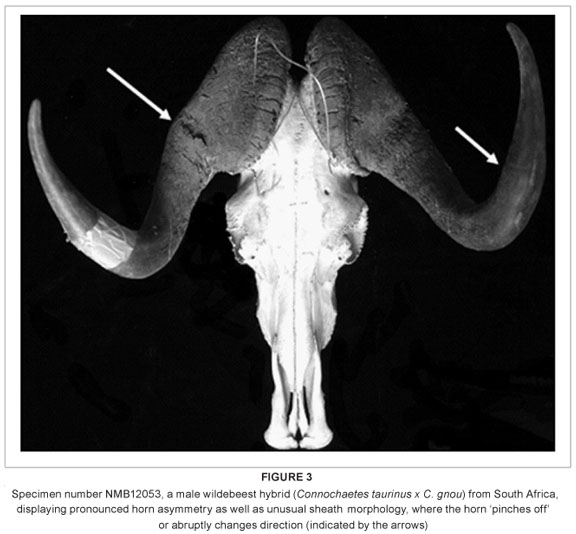
In addition to these dental and sutural morphological anomalies, there are also three individuals with abnormal horn sheath morphology, one of which also has a pronounced horn asymmetry (Figure 3). Although the presence of unusual and often intermediate horn morphology has been observed in hybrid wildebeest, and used to detect the presence of hybridisation in wild populations,21 the variation in horn morphology among the hybrids examined here is nonetheless striking.
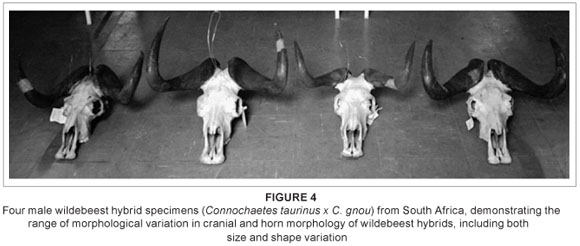
Substantial cranial variation is also present across the sample, both in terms of size and shape (Figure 4); this variation (both cranial and postcranial) has been more fully quantified and described elsewhere.22 While hybrid morphology is typically depicted as intermediate, in reality hybrid populations are highly variable, or polymorphic,29 with individuals showing a range of phenotypes that can be intermediate to the parental morphs, can resemble one parent or the other, or can fall outside of the parental range. The high variability seen here is consistent with such expectations.
Although this is a preliminary report documenting hybrid features in the crania of wildebeest, the implications of the results are nonetheless significant. Most importantly, these wildebeest provide additional evidence of dental and sutural morphological anomalies in mammalian hybrids. Previously, it has been hypothesised that such morphological traits, which have been observed in the hybrids of known pedigree and wild baboons,26 wild gorillas25 and recent and Pleistocene squirrels,27 are broadly characteristic of mammalian hybridisation.26,29 The presence of these morphological anomalies in the wildebeest provides further evidence in support of this hypothesis.
These results also indicate that anomalous morphological traits occur in fairly recently diverged lineages that have separated during the Pleistocene. Hybridisation is expected to have very different phenotypic effects in lineages that are distantly divergent, compared with those that are more recently separated.30,31 Extreme genetic differences (e.g. differentially fixed alleles and different diploid numbers of chromosomes) in the more divergent species could potentially result in extremely anomalous or even inviable hybrids, while limited differentiation in more recently separated species may lead to hybrids with relatively few signs of morphological anomalies. The range of phenotypic expression under such different scenarios remains, however, to be empirically demonstrated for most mammals.29
This study also demonstrates that the expression of such anomalies may differ across phylogenetically divergent mammalian groups. In primate skulls, the morphological anomalies associated with hybridisation were predominantly mandibular distomolars, although unusual zygomaxillary sutures were also fairly common.24,25,26 For squirrels, distomolars were also present, although they were maxillary.27 In both cases, the pattern of expression differed from the parental taxa. In the wildebeest sample, morphological anomalies were largely sutural, with limited evidence of dental anomalies, and considerable variation in horn shape. It will benefit our understanding of hybrid morphology to continue to examine morphological trait variation in skeletal collections of other mammals to more fully understand the range of variation in the mammalian hybrid phenotype.
Finally, this study shows the potential for identifying hybrid wildebeest and zones of introgression on the landscape when animal provenance is known.25 The identification of hybrid zones can provide insights into the dynamics of extant populations, as well as the recent historical and/or evolutionary past of the groups under investigation (sensu Ackermann and Bishop25), depending on when the skeletal material was collected. Identifying wildebeest hybrids in situ is also important from a conservation perspective. There is an ongoing effort to minimise the impact of introgression of blue wildebeest genes into black wildebeest populations and such increased powers of identification, applied either alone or in conjunction with molecular data, could play an important role in efforts to conserve this endemic South African species.
REFERENCES
1. Brink JS. The taphonomy of an Early/Middle Pleistocene hyaena burrow at Cornelia-Uitzhoek, South Africa. Rev de Palaéobiol 2004;23:731–740. [ Links ]
2. Brink JS. Postcranial evidence for the evolution of the black wildebeest Connochaetes gnou: An exploratory study. Palaeontol Afr. 1993;30:61–69. [ Links ]
3. Corbet SW. Genetic divergence in the wildebeest, Connochaetus taurinus and C. gnou: A molecular and cytogenetic study. MSc thesis, Pretoria, University of Pretoria, 1991. [ Links ]
4. Corbet SW, Grant WS, Robinson TJ. Genetic divergence in South African Wildebeest: Analysis of allozyme variability. J Hered. 1994;85:479–483. [ Links ]
5. Corbet SW, Robinson TJ. Genetic divergence in South African Wildebeest: Comparative cytogenetics and analysis of mitochondrial DNA. J Hered. 1991;82:447–452. [ Links ]
6. Brink JS. The evolution of the black wildebeest, Connochaetes gnou, and modern large mammal faunas in central southern Africa. DPhil dissertation, Stellenbosch, University of Stellenbosch, 2005. [ Links ]
7. IUCN SSC Antelope Specialist Group 2008. Connochaetes taurinus. In: IUCN 2010. IUCN Red List of Threatened Species. Version 2010.2 [homepage on the Internet]. c2010 [cited 2010 August 30]. Available from: http://www.iucnredlist.org [ Links ]
8. Fabricius C, Lowry D, van den Berg P. Fecund black wildebeest x blue wildebeest hybrids. S Afr J Wildl Res. 1988;18(1):35–37. [ Links ]
9. Attwell CAM. Reproduction and population ecology of the blue wildebeest Connochaetes taurinus taurinus in Zululand. PhD thesis, Pietermaritzburg, University of Natal, 1977. [ Links ]
10. Skinner JD, Chimimba CT. The mammals of the southern African subregion. Cambridge: Cambridge University Press; 2005. [ Links ]
11. Von Richter W. Observations on the biology and ecology of the black wildebeest (Connochaetus gnou). J S Afr Wildl Mgmt Assoc. 1971;1:3–16. [ Links ]
12. Von Richter W. Connochaetes gnou. Mamm Species. 1974;50:1–6. [ Links ]
13. Hirst SM. Ungulate-habitat relationships in a South African woodland/savanna ecosystem. Wildl Monogr. 1975;44:1–60. [ Links ]
14. Skead CJ. Historical mammal incidence in the Cape Province. Vol. 1. The western and northern Cape. Cape Town: Department of Nature and Environmental Conservation of the Provincial Administration of the Cape of Good Hope; 1980. [ Links ]
15. Brink JS, Berger LR, Churchill SE. Mammalian fossils from erosional gullies (dongas) in the Doring River drainage, central Free State Province, South Africa. In: Becker C, Manhart H, Peters J, Schibler J, editors. Historium animalium ex ossibus Beiträge zur Paläoanatomie, Archäologie, Ägyptologie, Ethnologie und Geschichte der Tiermedzin: Festschrift für Angela von den Driesch Verlag Marie Leidorf: Rahden/Westf, 1999; p. 79–90. [ Links ]
16. Plug I, Badenhorst S. The distribution of macromammals in Southern Africa over the past 30,000 years as reflected in animal remains from archaeological sites. Monographs of the Transvaal Museum. Pretoria: Transvaal Museum; 2001. [ Links ]
17. Codron D, Brink JS. Trophic ecology of two savanna grazers, blue wildebeest Connochaetes taurinus and black wildebeest Connochaetes gnou. Eur J Wildl Res. 2007;53:90–99. [ Links ]
18. Kok O, Vrahimis S. Black wildebeest territorial clearings. J Afr Zool. 1995;109:231–237. [ Links ]
19. Sidney J. The past and present distribution of some African ungulates. Vol 30. London: Zoological Society of London; 1961. [ Links ]
20. Grobler JP, Hartl GB, Grobler N, Kotze A, Botha K, Tiedemann R. The genetic status of an isolated black wildebeest (Connochaetes gnou) population from the Abe Bailey Nature Reserve, South Africa: Microsatellite data on a putative past hybridisation with blue wildebeest (C. taurinus). Mamm Biol. 2005;70:35–45. [ Links ]
21. Friedmann Y. Black wildebeest (Connochaetes gnou) hybridisation action plan. Final workshop report: Conservation Breeding Specialist Group (SSC/IUCN) / CBSG Southern Africa, Endangered Wildlife Trust; 2003. [ Links ]
22. De Klerk B. An osteological documentation of hybrid wildebeest and its bearing on black wildebeest (Connochaetes gnou). MSc thesis, Johannesburg, University of the Witwatersrand, 2007. [ Links ]
23. Langley N. Black wildebeest x blue wildebeest hybrid at Spioenkop Nature Reserve, unpublished Natal Parks Board report; 1995. [ Links ]
24. Ackermann RR. Craniofacial variation and developmental divergence in primate and human evolution. In: Lieberman D, Hall B, editors. Novartis Foundation Symposium 284 Tinkering: the micro-evolution of development. Chichester: Wiley, 2007; p. 262–279. [ Links ]
25. Ackermann RR, Bishop JM. Morphological and molecular evidence reveals recent hybridisation between gorilla taxa. Evolution. 2010;64:271–290. [ Links ]
26. Ackermann RR, Rogers J, Cheverud JM. Identifying the morphological signatures of hybridisation in primate and human evolution. J Hum Evol. 2006;51:632–645. [ Links ]
27. Goodwin HT. Supernumerary teeth in Pleistocene, recent, and hybrid individuals of the Spermophilus richardsonii complex (Sciuridae). J Mammal. 1998;79:1161–1169. [ Links ]
28. Heide-Jørgensen MP, Reeves RR. Description of an anomalous monodontid skull from West Greenland: A possible hybrid? Mar Mamm Sci. 1993;9:258–268. [ Links ]
29. Ackermann RR. The phenotype of primate hybrids: Recognizing admixture in the fossil past. Evol Anthropol. In press. [ Links ]
30. Falconer DS, Mackay T. Introduction to quantitative genetics. New York: Dover Publications; 1997. [ Links ]
31. Stelkens RB, Schmid C, Selz O, Seehausen O. Phenotypic novelty in experimental hybrids is predicted by the genetic distance between species of cichlid fish. BMC Evol Biol. 2009;9:283. [ Links ]
 Postal address:
Postal address:
Department of Archaeology, University of Cape Town
Private Bag X1
Rondebosch 7701, South Africa
Received: 11 Mar. 2010
Accepted: 07 Sept. 2010
Published: 12 Nov. 2010
Correspondence to: Rebecca Ackermann
This article is available at: http://www.sajs.co.za
© 2010. The Authors. Licensee: OpenJournals Publishing. This work is licensed under the Creative CommonsAttribution License.














