Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Science
versión On-line ISSN 1996-7489
versión impresa ISSN 0038-2353
S. Afr. j. sci. vol.106 no.9-10 Pretoria sep./oct. 2010
RESEARCH ARTICLE
Palladium mixed-metal surface-modified AB5-type intermetallides enhance hydrogen sorption kinetics
Mario WilliamsI; Mykhaylo LototskyI; Alexander NechaevII; Volodymyr YartysIII; Jan K. SolbergIV; Roman V. DenysV; Vladimir M. LinkovI
ISouth African Institute for Advanced Materials Chemistry, Department of Chemistry, University of the Western Cape, South Africa
IIShubnikov Institute of Crystallography, Russian Academy of Science, Moscow, Russia
IIIInstitute for Energy Technology, Kjeller, Norway
IVNorwegian University of Science and Technology, Trondheim, Norway
VPhysico-Mechanical Institute, Lviv, Ukraine
ABSTRACT
Surface engineering approaches were adopted in the preparation of advanced hydrogen sorption materials, based on 'low-temperature', AB5-type intermetallides. The approaches investigated included micro-encapsulation with palladium and mixed-metal mantles using electroless plating. The influence of micro-encapsulation on the surface morphology and kinetics of hydrogen charging were investigated. It was found that palladium-nickel (Pd-Ni) co-deposition by electroless plating significantly improved the kinetics of hydrogen charging of the AB5-type intermetallides at low hydrogen pressure and temperature, after long-term pre-exposure to air. The improvement in the kinetics of hydrogen charging was credited to a synergistic effect between the palladium and nickel atoms in the catalytic mantle and the formation of an 'interfacial bridge' for hydrogen diffusion by the nickel atoms in the deposited layer. The developed surface-modified materials may find application in highly selective hydrogen extraction, purification, and storage from impure hydrogen feeds.
Keywords: AB5-type intermetallides; electroless plating; kinetics; mixed-metal; palladium
INTRODUCTION
Breakthroughs in hydrogen separation, purification, and storage technologies require composite materials that meet the scientific and technological requirements of hydrogen processing, including excellent sorption stability, resistance to surface poisoning in impure hydrogen and fast reaction kinetics. It has been recognised that the key elements for substantial advancement in this field will be provided by new strategies in the preparation of novel composite materials wherein the surface composition and structure are specifically tailored for their desired application.
Technologically flexible 'low-temperature', rare earth metal AB5-type intermetallides are potentially attractive materials for the sorption of hydrogen, because they have good reversibility in hydrogen sorption, fast kinetics in hydrogen sorption under mild conditions, tunable plateau pressures, reasonable cycle lifetimes, and low hysteresis.1,2,3,4 The hydrogen charging/discharging capacities of the AB5-type intermetallides exhibit substantial sensitivity to the chemical state of the surface. In particular, exposure of the AB5-type intermetallides to air prior to hydrogen absorption results in selective surface oxidation of the rare earth metals. Surface oxidation yields friable, hydrogen-permeable rare earth oxides and simultaneous segregation of metallic nickel. Segregated clusters promote the processes of dissociative chemisorption and associative desorption of hydrogen (H2) molecules, which improves the hydrogen sorption performance of the material.5
As hydrogen sorption from impure hydrogen feed streams is likely to involve exposure to chemically aggressive electrophilic gaseous components (e.g. hydrogen sulphide [H2S], carbon monoxide [CO], oxygen [O2] and water [H2O]), the rationale for improving their hydrogen sorption kinetics and surface poisoning resistance, by surface modification of AB5-type intermetallides, emerges. AB5-type intermetallides tend to exhibit susceptibility to these gases, passivating the surface, and thus inhibiting hydrogen dissociation/recombination processes.
There are several methods to eliminate the negative effects of impurities on hydrogen sorption properties of intermetallides: creation of multi-phase materials,6 introduction of deoxidisers,7 fluorination,8,9 alkaline ion implantation,10 and micro-encapsulation.11,12,13 The last method is especially efficient when platinum group metals are used as a modifier, because it facilitates an increase in the resistance of intermetallides, even to strong gaseous poisons, such as CO.14 The surface coatings of platinum group metals enable rapid dissociation of hydrogen molecules, while still maintaining the charging activity of the intermetallide, even after extended exposure to contaminated hydrogen. In particular, palladium has the ability to catalyse hydrogen exchange reactions, promote easy activation, and increase poisoning tolerance.13,14,15,16 It is impermeable to larger molecules, and as a result palladium also exhibits an infinitely high selectivity towards hydrogen. Palladium micro-encapsulation also gives intermetallides the ability to absorb hydrogen at low temperature, without a special activation step, and enhanced kinetics of hydrogen charging. Palladium catalytic layers can typically be deposited using a wet chemical technique such as electroless plating, which is known for its ability to plate irregular surfaces and prepare metal layers of uniform thickness.
Palladium surface-modified AB5-type intermetallides are, however, still prone to low levels of surface poisoning after long periods of exposure to air; such poisoning manifests in the gradual deterioration of hydrogen sorption capacity with repeated cycling.13 To address this problem, it was suggested that surface modification be conducted using mixed-metal coatings instead of pure palladium coatings. Palladium alone is also not suitable for hydrogen uptake, because of its unfavourable equilibrium pressure, high cost, and susceptibility to sulphurous gases.17 There have been few known attempts to study the influence of micro-encapsulation of palladium-based mixed-metals on the hydrogen charging performances of intermetallides.13,15,16,18,19 It also is known that employment of mixed-metal catalysts instead of single-metal catalysts can enhance activity and/or selectivity for a given reaction.reasons, the use of palladium mixed-metal coatings to enhance the kinetics of hydrogen charging of AB5-type intermetallides was investigated.
Mixed-metal coatings of interest include Pd-Ni and palladium-copper (Pd-Cu). Nickel surface coatings are known to improve the overall mechanical strength and corrosion resistance of intermetallides. In addition, surface coatings of nickel are known to increase the catalytic activity of AB5-type intermetallides towards hydrogen exchange reactions.21 Likewise, Pd-Cu mixed-metal coatings have the potential to enhance resistance to thermal degradation.22
This study details approaches for the preparation of new composite materials, based on AB5-type intermetallides that are surface-modified using Pd-Ni and Pd-Cu mixed-metal coatings deposited by electroless plating, for the separation, purification and storage of hydrogen from impure feed streams. The materials were evaluated in terms of morphological, elemental and hydrogen sorption properties. Importantly, the materials should exhibit kinetics of hydrogen charging superior to that of intermetallides that are surface-modified using palladium only.
EXPERIMENTAL METHODS
A rare earth mischmetal-nickel-based AB5-type intermetallide La0.40Ce0.48(Nd,Pr)0.16Ni3.34Co0.64Al0.63Mn0.58, produced in collaboration with the Guangzhou Research Institute for Non-Ferrous Metals, was employed as the core material for surface modification. The intermetallide was prepared from high-purity metals by a modified arc-melting technique under an inert gas atmosphere. The melt was cast into sheets using a high-pressure rolle r system fitted within an oven. The sheets were further annealed at 1295 ºC for 2 h under an inert atmosphere. Finally, the intermetallide was pulverised, by ball-milling, in a sodium hypophosphite (NaH2PO2) solution and sieved to 200 mesh (74 µm). The material was allowed constant exposure to air throughout the experimental studies. The particle size distribution was determined, using field emission scanning electron microscopy (FESEM), as 10 µm - 70 µm, with all particles possessing irregular shape.
Surface modification of the AB5-type intermetallide was conducted using electroless plating of palladium, nickel, and copper mantles either by single-layer, mixed-bath (co-deposition), or sequential deposition, in a hypophosphite-based electroless plating bath. The intermetallide was first sensitised and activated in a palladium-tin (Pd-Sn) colloidal solution.23 The activated intermetallides were subsequently suspended in the palladium, nickel and copper plating baths. An equivalent volume of NaH2PO2 solution (10 g/L) was added separately. The plating time and stirring rate were fixed at 30 min and 300 rpm, respectively. The concentrations of components in the palladium-based, nickel-based and copper-based plating solutions, as well as the volume ratio of the solutions to be mixed into the plating bath, were selected to provide a 5.0 x 10-4 mol/L to 1.5 x 10-3 mol/L concentration of Pd2+, a 1.0 x 10-3 mol/L to 2.5 x 10-3 mol/L concentration of Ni2+ and a 1.0 x 10-2 mol/L to 4.0 x 10-2 mol/L concentration of Cu2+ in the bath. Descriptions of the formulations for the hypophosphite-based electroless plating baths are given in Table 1.24,25,26
X-ray diffraction (XRD) studies of the unmodified and surface-modified AB5-type intermetallides were performed using a Bruker Advance powder diffractometer (Madison, USA; 40 mA, 40 keV, λCu-K = 0.15406 nm) at the Materials Research Group, iThemba Labs, in Cape Town, South Africa. Synchrotron radiation XRD (SR-XRD) studies of the unmodified and surface-modified AB5-type intermetallides were performed using the BM01B diffractometer (λ = 0.05 nm) coupled to the Swiss-Norwegian beamline of the third-generation synchrotron radiation accelerator (6.0 GeV) at the European Synchrotron Radiation Facility in Grenoble, France. Small quantities of the samples were introduced into quartz glass capillary tubes (0.7 mm diameter), which were then sealed to the gas distribution system using carbon ferrules. The capillary tubes were connected to a goniometer head and oscillated, under vacuum, around their axes to minimise orientation effects.27 The sample materials were then irradiated using X-rays from the synchrotron storage ring. After collection, the SR-XRD data were further refined by profile analysis with General Structure Analysis System software.28
All prepared materials were characterised using FESEM (Zeiss Ultra 55 Limited Edition, Gottingen, Germany; 3.0 mm; 4.0 kV) for morphological studies of palladium particle size and shape on the surface of the AB5-type intermetallides. Estimations of the intermetallide surface composition were conducted using atomic absorption spectroscopy (AAS; Philips PU9100, Eindhoven, Germany).
The influence of micro-encapsulation of the AB5-type intermetallide on the kinetics of hydrogen charging was studied by a comparison of hydrogen absorption under mild conditions (at a temperature of about 20 ºC and a hydrogen pressure of about 500 kPa), after exposure to air for approximately 2 weeks and evacuation before hydrogen charging (10-4 kPa, 20 ºC). Evacuation prevented the possibility of the presence of residual oxygen on the surface of the alloy. Hydrogen charging was conducted for 24 h. The experimental results were processed by application of formal kinetic analysis, using the Avrami-Erofeev equation29:
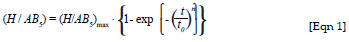
where (H/AB5) is the actual hydrogen concentration in the intermetallide; (H/AB5)max is the maximum hydrogen concentration in the intermetallide (equal to 5, according to results of the pressure-concentration-temperature study)30; t is time; t0 is the characteristic time of hydrogen charging (reciprocal rate constant k); and the index of power, n, can be interpreted as a value indirectly connected to the reaction mechanism.
RESULTS AND DISCUSSION
The surface morphologies of the unmodified and surface-modified AB5-type intermetallides are shown in Figure 1. The unmodified intermetallide was composed of particles which, generally, exhibited irregular shape and relatively smooth surfaces, and which were free of porous structure (Figure 1a).
The palladium coatings on the AB5-type intermetallides were found to be discontinuous in nature (Figure 1b). The observation of discontinuous palladium coatings on the surface of AB5-type intermetallides was shared by Willey et al.13, Doyle et al. 16, Pratt et al. 18 and Harris et al.19 The deposited palladium particles were somewhat spherical in appearance and exhibited an average particle size of 135 nm. They also exhibited a coarse, granular appearance that may be credited to their amorphous nature after impregnation of phosphorus into the palladium layer. Agglomeration of the palladium particles was not observed on the intermetallide surface.
Good dispersion of the particles was observed after sequential and co-deposition of Pd-Ni, similar to that observed after pure palladium deposition (Figures 1c and 1d). The Pd-Ni coatings were also found to be discontinuous in nature, while very little agglomeration of the particles was observed on the intermetallide surface. Surface particles were fairly spherical in shape after Pd-Ni sequential deposition, while Pd-Ni co-deposition resulted in particles with a very rough appearance. Average particle sizes of 92 nm and 105 nm were determined after Pd-Ni sequential and co-deposition, respectively.
Similarly, Pd-Cu sequential-deposited and co-deposited coatings were found to be discontinuous in nature and well dispersed (Figures 1e and 1f). In both cases, the surface particles were observed to have fairly rough shapes, with particles exhibiting a nodular appearance. Average particle sizes of 291 nm and 109 nm were determined for the intermetallides that were surface-modified using Pd-Cu sequential- and co-deposition, respectively. The larger particle size of the sequentially deposited Pd-Cu layer, compared to the co-deposited layer, may be credited to the dominant surface presence of the copper particles, as confirmed by the reddish-brown appearance of the surface-modified intermetallide particles.
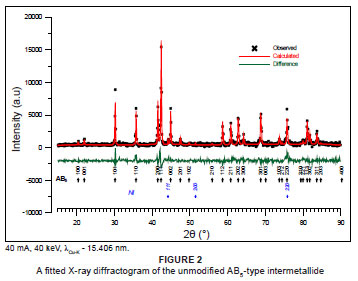
XRD studies of the unmodified AB5-type intermetallide (Figure 2) have shown that the material contains a major CaCu5-type intermetallic phase (95 net wt. %) with lattice parameters a = 0.5003 nm, c = 0.4052 nm and a minor AB5-type intermetallic phase (4.7 net wt. %) with similar lattice parameters a = 0.5023 nm, c = 0.4079 nm. The lattice parameters corresponded well with those reported previously (a = 0.495 nm - 0.505 nm, c = 0.402 nm - 0.405 nm) for a similar intermetallide La0.8(1-x)Ce0.8x(PrNd)0.2B5, where B5 is Ni3.55Co0.75Mn0.4Al0.3, x = 0 - 1.31 It was most likely that the presence of the second AB5-type intermetallide phase was a direct result of inhomogeneity in the B-component of the parent AB5-type intermetallide. Similarly, Willey et al.13 detected the presence of 10 net wt. % of a second phase, ascribed as A2B7. Trace quantities (0.7 net wt. %) of a mixed rare-earth-nickel oxide (RE2NiO4) perovskite-type oxide phase, having K2NiF4-type structure,32 a= 0.38572 nm; c=1.2660 nm, were detected and most probably originated from the surface oxidation of the material. This phase is present in unmodified sample only and disappears after surface treatment of the alloy.
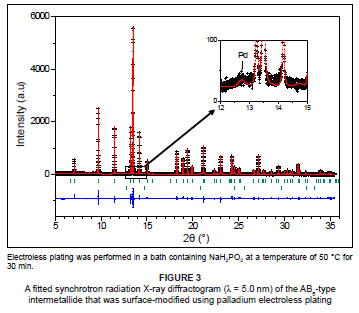
The SR-XRD diffractogram of the AB5-type intermetallide, surface-modified by palladium coatings derived from NaH2PO2-based electroless plating baths, showed that the material contained relatively unchanged lattice constants in terms of the major and minor AB5-type intermetallic phases (Figure 3). A palladium phase was detected (±1.0 net wt. %) at 2θ = 12.8º. The measured XRD patterns did not show the presence of crystalline palladium, which allows for the deduction of the amorphous nature of the palladium coating, and is due to the inclusion of phosphorus into the metal coating.
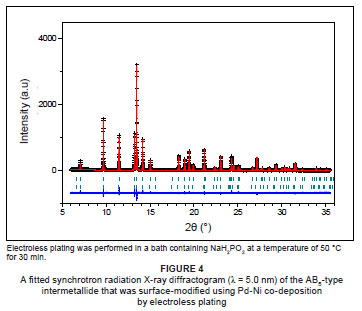
The SR-XRD diffractogram of the AB5-type intermetallide that was surface-modified by Pd-Ni co-deposition is shown in Figure 4. No clear peaks from either of the deposited nickel and palladium particles were observed. This observation may be due to the amorphous state of the particles or the lack of detectable quantities for SR-XRD analysis. Similarly to palladium-deposition, the material also contained relatively unchanged lattice constants in terms of the major and minor AB5-type intermetallic phases. Identical observations were made for the intermetallide that was surface-modified using Pd-Cu co-deposition. In addition, the absence of peaks corresponding to the deposited nickel and palladium particles may be a result of the relatively low surface loadings of these two metals on the surface of the AB5-type intermetallides.
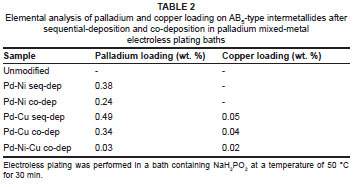
Elemental analysis was conducted using AAS to ascertain the quantities of palladium, nickel, and copper deposited on the surface of the encapsulated intermetallide materials. A comparative table (Table 2) presents the surface elemental properties of the sample materials. Deposited nickel could not be resolved from the nickel surface matrix of the AB5-type intermetallide. The AAS analyses showed that fairly low palladium and copper loadings were deposited onto the surface of the AB5-type intermetallide after electroless plating.
Measurements of the influence of Pd-Ni and Pd-Cu mixed-metal coatings on the kinetics of hydrogen charging of surface-modified AB5-type intermetallides were undertaken after exposure to air, and without thermal activation (Figure 5). Results of the fit of the experimental data by the Avrami-Erofeev equation are presented in Table 3; the sequence corresponded to the increase in the calculated rate constant (k).
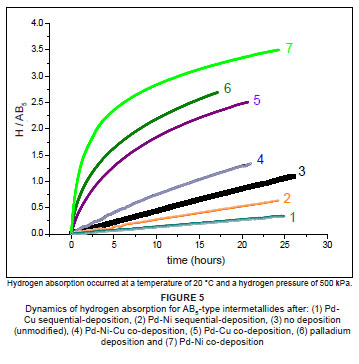
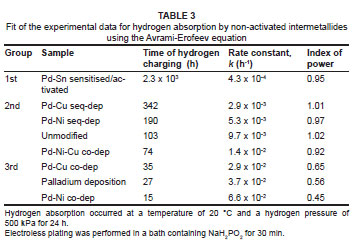
Sample materials could be divided into two groups, in terms of their hydrogen absorption behaviour. The first group included the unmodified intermetallide, the intermetallide modified by sequential deposition of palladium and nickel (or copper), as well as the intermetallide modified by co-deposition of palladium, nickel and copper. These materials were characterised by slow hydrogen absorption, reaching an estimated characteristic time of hydrogen absorption, t0, of between 74 h and 340 h and a hydrogen sorption capacity corresponding to H/AB5 = 0.35-1.35. The poor kinetic performances of the surface-modified intermetallides belonging to this group may have originated from the formation of dense nickel (or copper) layers on the surface, which acted as inhibitors to the transport of hydrogen, and limited the catalytic ability of the palladium component of the deposited layer.
The second group included the sample surface-modified by palladium and co-deposited palladium and nickel (or copper). These materials were characterised by significantly faster kinetics of hydrogen charging and absorbed more than one-half of the maximum quantity of hydrogen within 24 h (H/AB5 = 2.5 - 3.5), with a characteristic time of hydrogen charging 3-fold to 7-fold lower than that of the unmodified intermetallide. Fitted n values were close to 0.5, which allowed for the assumption that hydrogen charging of these materials had a mechanism different from that of the sample materials in the first group (n = ±1.0).
The best kinetics of hydrogen charging were observed for the AB5-type intermetallide, surface modified by co-deposition of palladium and nickel. It was deduced that nickel particles in Pd-Ni co-deposition facilitated the creation of an interfacial 'bridge' between nickel surface clusters of the intermetallide and deposited an amorphous palladium layer. This interface may have increased the rate of transport of the hydrogen atoms, after dissociation of the hydrogen molecules on the palladium particles, towards the bulk material. Similarly, the reverse process of passing the hydrogen atoms from the bulk to the modified surface, followed by the associative hydrogen molecules desorption, can also be promoted. The improvement in the kinetics of hydrogen charging could also be credited to a synergistic effect between the palladium and nickel atoms in the catalytic mantle, which promoted the absorption and transport of hydrogen atoms.
An attempt was made to study the long-term sorption stability of the surface-modified intermetallides. Measurements of the kinetics of hydrogen charging by the sample materials were undertaken after ±3 months and ±11 months exposure of the samples to air, and without thermal activation. The results with respect to the absorption of hydrogen were collected and are presented in Figure 6.
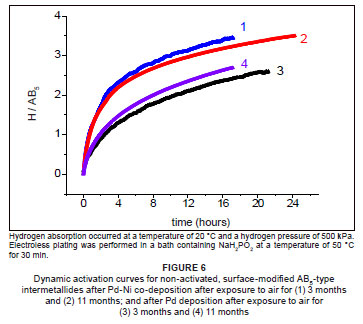
Rate constants for both the samples did not deteriorate very much, illustrating the long-term sorption stability of the materials, compared to the unmodified intermetallide. In addition, the sample prepared by co-deposition of Pd-Ni retained its superior kinetics of hydrogen charging, compared to that prepared by deposition of palladium only. This observation further illustrates that the addition of nickel to the palladium-based surface layer further enhanced the sorption stability of the intermetallide material, compared to that prepared by deposition of palladium only. We have thus confirmed our hypothesis that mixed-metal surface layers on AB5-type intermetallides offer enhanced sorption stabilities.
CONCLUSION
It was proposed that the use of mixed-metal coatings for the surface micro-encapsulation of AB5-type intermetallides, instead of with pure palladium only, had the potential to further improve the hydrogen absorption properties and sorption stability of the intermetallide materials. Pd-Ni co-deposited coatings, prepared by electroless plating, were found to promote faster hydrogen charging performances of the air-exposed, non-activated intermetallide materials, compared to those coated with palladium only. Improvements in the kinetics of hydrogen charging were credited to a synergistic effect between the palladium and nickel atoms in the catalytic mantle and to the formation of an 'interfacial bridge' for hydrogen diffusion by the nickel atoms in the deposited layer.
The developed composite materials may find application in rapid hydrogen separation, purification and storage from contaminated hydrogen feed streams.
ACKNOWLEDGEMENT
This work was supported by the South Africa-Norway Programme of Research Co-operation (2007-2010), Project #180344.
REFERENCES
1. Sandrock G. A panoramic overview of hydrogen storage alloys from a gas reaction point of view. J Alloys Compd. 1999;293-295:877-888. [ Links ]
2. Dantzer P. Properties of intermetallic compounds suitable for hydrogen storage applications. Mater Sci Eng A. 2002;A329-331:313-320. [ Links ]
3. Mani N, Ramaprabhu S. Effect of substitutional elements on hydrogen absorption properties in Mm-based AB5 alloys. J Alloys Compd. 2004;363:275-291. [ Links ]
4. Mungole MN, Balasubramaniam R. Effect of hydrogen cycling on the hydrogen storage properties of MnNi4.2Al0.8. Int J Hydrogen Energy. 2000;25:55-60. [ Links ]
5. Schlapbach L, Seiler A, Siegmann HC, Waldkirch TV, Zurcher P, Brundle CR. Self restoring of the active surface in LaNi5. Int J Hydrogen Energy. 1979;4:21-28. [ Links ]
6. Lototsky MV. The effect of improvement of hydrogen-sorption properties in multiphase polymetallic compositions. In: Saetre TO, editor. Hydrogen power, theoretical and engineering solutions. Paper presented at: Proceedings of the Hydrogen Power, Theoretical and Engineering Solutions International Symposium II 1997; 1997 Aug 18-22; Grimstad, Norway. Dordrecht: Kluwer Academic Publishers; 1998. p. 343-352. [ Links ]
7. Da Silva EP. Industrial prototype of a hydrogen compressor based on metallic hydride technology. Int J Hydrogen Energy. 1993;18:307-311. [ Links ]
8. Wang X-L, Iwata K, Suda S. Hydrogen purification using fluorinated LaNi4.7Al0.3. J Alloys Compd. 1995;231:860-864. [ Links ]
9. Sun Y-M, Suda S. Studies on the fluorination method for improving surface properties and characteristics of AB5- types of hydrides. J Alloys Compd. 2002;330-332:627-631. [ Links ]
10. Xiao L, Wang Y, Liu Y, Song D, Jiao L, Yuan H. Influence of surface treatments on the microstructure and electrochemical properties of La0.7Mg0.3Ni2.4Co0.6 hydrogen storage alloy. Int J Hydrogen Energy. 2008;33:3925-3929. [ Links ]
11. Uchida H. Surface processes of H2 on rare earth based hydrogen storage alloys with various surface modifications. Int J Hydrogen Energy. 1999;24:861-869. [ Links ]
12. Chuang HJ, Chan SLI. Effect of Ni encapsulation on the properties of Ti-Zr-based hydrogen storage alloys. J Alloys Compd. 2001;314:224-231. [ Links ]
13. Willey DB, Harris IR, Pratt AS. The improvement of the hydrogenation properties of nickel metal hydride battery alloy by surface modification with platinum group metals (PGMs). J Alloys Compd. 1999;293-295:613-620. [ Links ]
14. Bratanich TI, Bulanov VN, Skorokhod VV, Klimenko VP. Reversible hydriding of LaNi5-xAlx-Pd composites in the presence of carbon monoxide. Powder Metall Met Ceram. 2000;39:575-583. [ Links ]
15. Geng M. Electrochemical characterization of MmNi5- based alloy powder coated with palladium and nickel-palladium. J Alloys Compd.1994:215:151-153. [ Links ]
16. Doyle ML, Harris IR, Pratt AS, Willey DB. Hydrogen storage materials, United States Patent 6165643. 2000. [ Links ]
17. Züchner H, Opara L, Barlag L. Hydrogen diffusion in palladium f.c.c. based alloys. J Alloys Compd. 2002;330-332:434-437. [ Links ]
18. Pratt AS, Willey DB, Harris IR. High performance metal hydride alloy for rechargeable battery technology. Platinum Met Rev. 1999;43:50-58. [ Links ]
19. Harris IR, Willey DB, Pederzollic D, Pratt AS, Swift J, Walton A. Low temperature hydrogenation properties of platinum group metal treated nickel metal hydride electrode alloy. J Alloys Compd. 2002;330-332:806-809. [ Links ]
20. Lianos L, Debauge Y, Massardier J, Jugnet Y, Bertolini JC. 1,3-butadiene hydrogenation of Pd50Cu50 single crystals. Catal Lett. 1997;44:211-216. [ Links ]
21. Slys I, Scherbakova L, Rogozinskaya A, Schur D, Rogozinskii A. Development of methods of deposition of discontinuous nickel coatings on powders of AB5 type alloys. Paper presented at: NATO Advanced Research Workshop on Hydrogen Materials Science and Chemistry of Carbon Nanomaterials 2003. Proceedings of the NATO Advanced Research Workshop on Hydrogen Materials Science and Chemistry of Carbon Nanomaterials; 2003 September 14-20; Sudak, Ukraine. New York: Springer; 2003. p. 131-136. [ Links ]
22. Lewis FA. The palladium/hydrogen system. London: Academic Press, 1967; p. 1-12, p. 48-49, p. 94-117. [ Links ]
23. Williams M, Pineda-Vargas CA, Khataibe EV, Bladergroen BJ, Nechaev AN, Linkov VM. Surface functionalization of porous ZrO2-TiO2 membranes using γ-aminopropyltriethoxysilane in palladium electroless plating. Appl Surf Sci. 2008;254:3211-3219. [ Links ]
24. Changrong X, Xiaoxia G, Fanqing L, Dingkun P, Guangyao M. Preparation of asymmetric Ni/ceramic composite membrane by electroless plating. Colloids Surf A Physicochem Eng Asp. 2001;179:229-235. [ Links ]
25. Yeung KL, Christiansen SC, Varma AJ. Palladium composite membranes by electroless plating technique: Relationships between plating kinetics, film microstructure and membrane. J Memb Sci. 1999;159:107-122. [ Links ]
26. Cheng DH, Xu WY, Zhang ZY, Yiao ZH. Electroless copper plating using hypophosphite as reducing agent. Met Finish. 1997;Jan:34-37. [ Links ]
27. Maehlen JP, Yartys VA, Denys RV, Fichtner M, Frommen Ch, Bulychev BM, Pattison P, Emerich H, Filinchuk YE, Chernyshov D. Thermal decomposition of AlH3 studied by in situ synchrotron x-ray diffraction and thermal desorption spectroscopy. J Alloys Compd. 2007;446-447:280-289. [ Links ]
28. Von Dreele RB, Larson AC. General structure analysis system; Regents of the University of California, 2001. [ Links ]
29. Førde T, Maehlen JP, Yartys VA, Lototsky MV, Uchida H. Influence of intrinsic hydrogenation/dehydrogenation kinetics on the dynamic behaviour of metal hydrides: A semi-empirical model and its verification. Int J Hydrogen Energy. 2007;32:1041-1049. [ Links ]
30. Williams M, Nechaev AN, Lototsky MV, Yartys VA, Solberg JK, Denys RV, Pineda CA, Li Q, Linkov VM. Influence of aminosilane surface functionalization of rare earth hydride-forming on palladium treatment by electroless deposition and hydrogen sorption kinetics of composite materials. Mat Chem Phys. 2009;115:136-141. [ Links ]
31. Yuan X, Liu H-S, Ma Z-F, Xu N. Characteristics of LaNi5-based hydrogen storage alloys modified by partial substituting La for Ce. J Alloys Compd. 2003;359:300-306. [ Links ]
32. Huot J, Swainson IP, Schulz R. Neutron diffraction study of lixiviated nanocrystalline Mg-Li compound. J Alloys Compd. 1999;292:292-295. [ Links ]
 Correspondence to:
Correspondence to:
Mario Williams
Postal address: South African Institute for Advanced Materials Chemistry Department of Chemistry University of the Western Cape
Private Bag X17
Bellville 7535, South Africa
email: 2438658@uwc.ac.za
Received: 12 June 2009
Accepted: 06 July 2010
Published: 05 Oct 2010
This article is available at: http://www.sajs.co.za
© 2010. The Authors. Licensee: OpenJournals Publishing. This work is licensed under the Creative Commons Attribution License.














