Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.106 n.7-8 Pretoria Jul./Aug. 2010
RESEARCH LETTER
Mesoporous ethanesilica materials with bimodal and trimodal pore-size distributions synthesised in the presence of cobalt ions
Neil J. CovilleI; Alufelwi M. TshavhungweI,II
IMolecular Sciences Institute, School of Chemistry, University of the Witwatersrand, South Africa
IIDepartment of Science and Technology, Pretoria, South Africa
ABSTRACT
Mesoporous organosilica materials containing ethane groups in their framework were formed with two and three pore sizes (i.e. bimodal and trimodal pores) when synthesised by the sol-gel method in the presence of cobalt ions. The compounds 1,2-bistrimethoxysilylethane and tetraethylorthosilicate were used as silicon sources and the reactions were done in the presence of a surfactant, which served as a template. Diffuse reflectance infrared Fourier transform spectroscopy revealed that organic functional groups were incorporated into the ethanesilica. Powder X-ray diffraction and nitrogen adsorption data indicated that the mesophase and textural properties (surface area, pore volume, pore diameter) of the materials were dependent on the ageing temperature, the amount/ratio of silica precursors and cobalt ion incorporation. Secondary mesopores were drastically reduced by changing the ratio of silicon precursors.
Keywords: cobalt; ethanesilica; mesoporous materials; porosity; sol-gel synthesis
INTRODUCTION
Owing to their high surface area and large pore volume, porous materials are frequently used as adsorbents and industrial catalyst supports. These materials are classified by the International Union of Pure and Applied Chemistry (IUPAC) according to the following predominant pore sizes: microporous (pore diameters less than 2 nm), mesoporous (pore diameters between 2 nm and 20 nm) and macroporous (pore diameters greater than 20 nm).1 Despite the natural abundance of silica, much research has focused on the synthesis of ordered mesoporous silicas, such as MCM41 and SBA15, with pore walls containing Si-O-Si repeat units and pores greater than 2 nm.2,3 In order to extend the variety of silica materials, the templating method has been used to synthesise hybrid silica materials, in which an organic spacer is inserted into the silica framework (i.e. between two silicon atoms).4,5,6,7,8 This insertion is achieved by replacing a pure silica precursor (e.g. tetraethylorthosilicate, TEOS) with a bridged organosilica precursor, in which an organic group is directly bonded to two silicon atoms. The building block to make these ordered materials is a bridged silsesquioxane, with the general structural formula (R'O)3Si-R-Si(R'O)3, where R and R' are alkyl/aryl groups. The products produced from the hybrid silica materials contain the 'O-Si-R-Si-O' unit and are often called periodic mesoporous organosilicas (PMOs).
PMOs have gained much attention because various organic functionalities (e.g. the R group in the 'O-Si-R-Si-O' unit) can be integrated within the stable inorganic framework.4,5,6,7,8 The PMOs possess both the structural properties (i.e. high surface area, high hydrothermal stability, large pore volume and ordered pores with tunable, uniformly sized pore sizes) and the chemical properties of ordered mesoporous materials, determined by both the silica and the organic bridging group. They have many potential applications that exploit these properties; in catalysis, adsorption, separation, sensing and nanotechnology.
The introduction of larger secondary pores in mesoporous materials, to form hierarchical porous materials with bimodal or multimodal pore-size distributions (PSDs), improves the activity of mesoporous catalysts.9 A common strategy to synthesise hierarchical mesoporous materials is to use two or more templates of different sizes. Although hierarchical mesoporous silicas with bimodal PSDs have been reported,10,11 there are very few reports in the literature on the synthesis of this type of hierarchical mesoporous organosilicas.12 In this paper, we report on the synthesis of a hierarchical ordered mesoporous ethanesilica by co-condensation of 1,2-bistrimethoxysilylethane (BTME) with TEOS in the presence of cobalt ions and hexadecyltrimethylammonium bromide (CTAB) as the surfactant. Large-size mesopores in trimodal mesoporous organosilica materials were controlled by varying the ratio of silica precursors.
The development of new nanoporous materials requires reliable characterisation methods. Two of the three most commonly used methods of characterising nanoporous silica materials, X-ray diffraction (XRD) and physical adsorption, were used to characterise the hybrid silica materials. Low temperature nitrogen adsorption isotherms are commonly used to evaluate the pore structure parameters (i.e. surface area, pore volume and PSDs) of mesoporous materials.13,14 The traditional method for analysing PSDs in the mesopore range is the Barrett-Joyner-Halenda (BJH) method, which is based on the Kelvin equation and thus has a thermodynamic origin.15 We also applied the recently developed density functional theory (DFT) method16,17,18 for PSD analysis from nitrogen adsorption and desorption isotherms to analyse the pore sizes of hybrid mesoporous silica materials. PSDs obtained from the DFT method were compared to those obtained by the BJH method. In contrast to a previous study,6 in which we used only one silica precursor (BTME), two silica precursors (TEOS and BTME) were used in the current study. This work is a continuation of our study of co-supported catalysts.6
MATERIALS AND METHODS
Chemicals
The 1,2-bistrimethoxysilylethane (BTME), hexadecyltrimethylammonium bromide (CTAB) and cobalt nitrate hexahydrate [Co(NO3)2.6H2O] compounds were obtained from Aldrich (Johannesburg, South Africa), while tetraethylorthosilicate (TEOS), ammonium hydroxide (NH4OH) and hydrochloric acid (HCl) were obtained from Fluka (Johannesburg, South Africa). All chemicals were used as received.
Synthesis
Cobalt ion incorporated ethanesilica materials were synthesised by adding 0.05 g cobalt nitrate to a mixture of CTAB (0.68 g, 1.86 mmol), water (28 mL) and 28% ammonium hydroxide (8 mL, 0.0576 mol). The mixture was stirred for 30 min. BTME (1.8 mL, 7.14 mmol) and TEOS (in increasing amounts: 0.8 mL, 3.2 mL and 6.4 mL) were simultaneously added dropwise to the stirred mixture so that the initial mole ratio was BTME:TEOS:CTAB:NH4OH:H2O:Co(NO3)2 = 1:x:0.293:14:244:0.054 (where x = 0.5, 2.0 and 4.0; see Table 1 for material nomenclature). Stirring was continued for a further 30 min, followed by ageing (at varying temperatures: 27 ºC, 55 ºC and 80 ºC) for 4 days. The solid was suction-filtered in a Büchner funnel, washed with an excess of water and dried in air overnight. This material was termed the 'as-synthesised' material. A portion of the sample (1.5 g) was stirred in a solution of concentrated HCl (5 mL) in methanol (200 mL) at 50 ºC for 2 days. The resulting solid was suction-filtered, washed with water and, finally, with methanol (200 mL) and then dried in air. This material was termed the 'solvent-extracted' material.
Characterisation
Powder X-ray diffraction measurements were made on a Philips PW1710 diffractometer (Westborough, United States of America) with Cu K-α radiation (λ = 1.5443 Å). Powdered disc samples were analysed at a scan speed of 5º s-1 in the 1.5º 2θ - 10º 2θ range at a generator voltage of 40 kV and a generator current of 20 mA. Nitrogen adsorption and desorption isotherms were measured at -196 ºC using a Micromeritics ASAP2010 instrument (Norcross, USA). Prior to adsorption measurements, all samples were degassed for 12 h at 120 ºC. The Brunauer-Emmett-Teller (BET) surface area was calculated from the adsorption isotherm in a relative pressure (P/Po) range from 0.001 to 0.20. The total pore volume was evaluated from the amount of nitrogen adsorbed at a relative pressure of 0.98. The average pore diameter was evaluated by the BET method (4V/A). The PSD was obtained from analysis of the adsorption and desorption branches of the isotherm using DFT-Plus software (Micromeritics) and the BJH method.
The use of DFT was reported in detail by Ravikovitch and Neimark18. The DFT approach is based on the established principles of statistical mechanics and necessarily assumes a model solid structure and pore topology. The different sized pores are assumed to be all of the same regular shape (e.g. cylinders or slits) and, generally, each pore is assumed to behave independently. According to DFT, the solid-fluid and fluid-fluid interactions control the pore filling, which may take the form of micropore filling or capillary condensation. DFT was demonstrated to be a reliable method for the characterisation of ordered siliceous materials and zeolites.16,17 The DFT method provides pore size, porosity and surface area information over the complete range of micropores and mesopores. In contrast, the BJH method is limited to determining pore sizes greater than 1.7 nm.18
Diffuse reflectance spectra were recorded using a Bruker Tensor 27 Spectrometer (Ettlingen, Germany) equipped with a Thermo Spectra-Tech Collector II diffuse accessory (Shelton, USA) and a high temperature chamber. Spectra were acquired at a nominal resolution of 4 cm-1 after signal averaging of four scans. Dispersions of the solid material in KBr (5% w/w) also were prepared.
RESULTS AND DISCUSSION
Synthesis
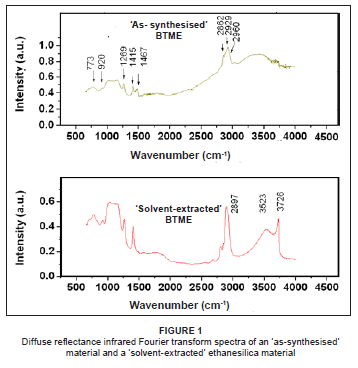
Addition of a mixture of BTME and TEOS to a basic solution of CTAB, in the presence of cobalt nitrate, formed grey/black powders. Similar periodic mesoporous organosilica materials, prepared without TEOS, have previously been reported.6 Diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) results confirmed the formation of organosilica materials and showed that the surfactant was removed by solvent extraction. Typical DRIFTS spectra for an 'as-synthesised' and a 'solvent-extracted' ethanesilica material are shown in Figure 1. The presence of CTAB was clearly seen in the 'as-synthesised' materials (with a shoulder at approximately 2970 cm-1 and a peak at 1467 cm-1). After removal of the surfactant by the extraction process, the peaks associated with CTAB had disappeared, while those resulting from the alkyl C-H stretch (2897 cm-1) and alkyl C-H deformations (1410 cm-1) of the BTME ethane group were still to be seen.8
Powder X-ray diffraction
All samples exhibited a prominent low angle peak in the diffraction pattern at approximately 2θ = 2º (Figure 2), which indicates the presence of uniformly sized pores for ordered materials.2 The most intense peak was arbitrarily assigned as the d100 peak and lattice parameters (d100-values) are listed in Table 1. There was a shift to lower d-values for samples prepared at a given temperature as the amount of TEOS in the reaction mixture increased (Table 1). The low intensity peaks observed between 2.5º and 3.5º in the XRD patterns of materials aged at lower temperatures (Figure 2a and 2b) are better resolved when compared to the corresponding materials aged at 80 ºC (Figure 2c). The absence of low intensity peaks in materials aged at 80 ºC indicates loss of long-range ordering. A similar XRD pattern was obtained for a corresponding ethanesilica prepared in the presence of cobalt ions but without adding TEOS.6 XRD patterns of materials aged at 55 ºC (A55 and B55) displayed a broad secondary feature in the 2θ range 2.5º - 3.5º. XRD patterns with a similar feature were also observed for other ethanesilica materials.4 Two resolved low intensity peaks were observed for C55, which can be indexed as the d110 and d200 peaks of a hexagonal mesophase.12
Nitrogen sorption
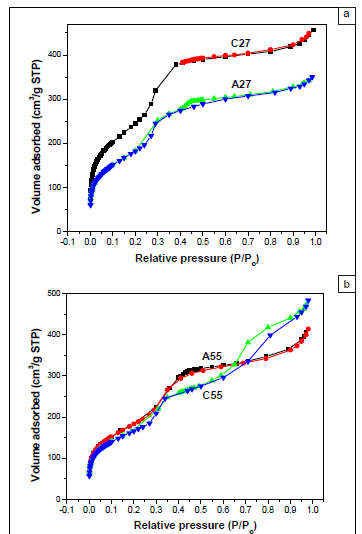
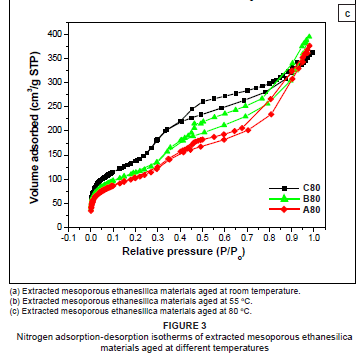
As shown in Figure 3, nitrogen adsorption-desorption isotherms of extracted ethanesilica materials aged at various temperatures (room temperature, 55 ºC and 80 ºC) exhibited Type IV characteristics associated with mesoporous materials, with relatively small microporous content.13 PMOs aged at room temperature (Figure 3a) exhibited Type IV isotherms with little or no hysteresis loops, indicating that the adsorption process was completely reversible, whereas those aged at 80 ºC (Figure 3c) had Type H3 hysteresis loops. Well-defined reversible Type IV isotherms with no hysteresis loops have been reported for MCM-41.14
A sharp inflection, characteristic of capillary condensation within uniform framework-confined mesopores (primary mesopores) with an average pore width of about 3 nm, was observed at relative pressures of between 0.3 and 0.4 for all the materials. A second inflection was also observed at a higher relative pressure (P/Po > 0.7) for the material containing 20% TEOS and aged at 80 ºC (A80), indicating the presence of a significant amount of secondary mesopores. Primary mesopores are formed when the surfactant is removed from the 'as-synthesised' material and their pore diameters depend on the surfactant used. Secondary mesopores arise from interparticle capillary condensation or from the structure collapse of portions of the mesoporous silica structure during hydrothermal treatment.10 The proportion of mesopores is expected to increase at higher ageing temperatures. In this study, the second capillary condensation step became less significant as the relative amount of TEOS in the reaction mixture increased. As such, the second capillary condensation step was negligible for materials synthesised at room temperature, indicating that the materials prepared at this temperature were dominated by monosize primary mesopores.
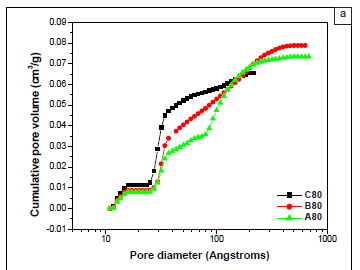
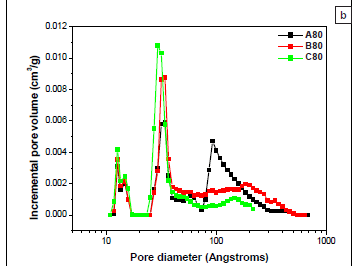
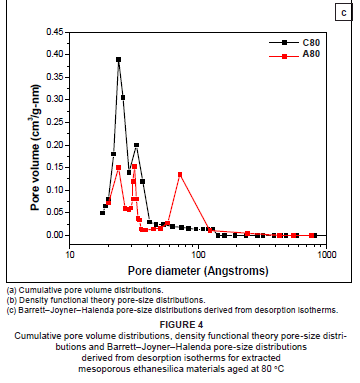
DFT pore-volume distributions (Figure 4a) and the corresponding DFT PSDs (Figure 4b) reveal that hybrid mesoporous material A80 had three pore types (i.e. a trimodal pore-size distribution). These pore types originated from capillary condensation in micropores, primary mesopores and secondary mesopores with pore diameters of approximately 1.3 nm, 3.2 nm and 9.3 nm, respectively. Sample A80 thus had a bimodal distribution of pores in the mesopore range. Figure 4c clearly shows that secondary mesopores were drastically reduced as the amount of TEOS in the reaction mixture increased. The PSD for sample C80 was bimodal, with one micropore (1.3 nm) and one mesopore (2.9 nm) (Figure 4c). These results show that large secondary mesopores can be introduced or removed in hybrid ethanesilicas by varying the ratio of precursors in the reaction mixture. Sizes of primary mesopores depend on the structure of surfactant used as a template. As expected for mesoporous materials synthesised from CTAB, sizes of primary mesopores in samples A80, B80 and C80 were similar (approximately 3 nm).
PSDs derived from adsorption isotherms by the DFT method (Figure 4b) and from desorption isotherms by the BJH method (Figure 4c) all showed that samples A80 and C80 had trimodal and bimodal distributions of pores, respectively. However, all the pores were in the mesoporous range and none were in the microporous range in BJH PSDs, whereas DFT PSDs had both mesopores and micropores. It is worth reiterating here that the BJH method is limited to determining pore sizes greater than 1.7 nm. Values of pore diameters of the first mesopore obtained from PSDs derived from DFT calculations were exactly the same as those obtained from the second mesopore peak of PSDs derived from BJH desorption isotherms (Table 1), suggesting that this peak corresponds to primary mesopores. In agreement with the trend obtained from DFT results, the intensity of the large size mesopore peak decreased significantly with increasing amounts of TEOS. The pore diameter of secondary mesopores obtained by the DFT method (7.2 nm) in sample A80 was lower than that obtained by the BJH method (9.2 nm). Previous reports have found that, compared with new methods that rely on more localised descriptions such as DFT, thermodynamically based methods, such as the BJH method, overestimate the relative pressure at desorption and therefore underestimate the calculated pore diameters by about 1 nm.16 Therefore, it is highly important to consider the method used to determine pore sizes and pore types when comparing the hierarchies of nanoporous materials reported in the literature.
The increase in average pore diameter from 4.40 nm to 6.48 nm for samples C80 and A80, respectively, is consistent with the increase in secondary mesopores as the amount of TEOS is decreased (Table 1). For samples prepared at a given temperature, the surface area was found to increase when increasing amounts of TEOS were added. Surface areas of materials aged at lower temperatures were higher than those of the corresponding materials aged at higher temperatures.
CONCLUSION
Ethanesilica materials with a bimodal and trimodal PSD in the nanopore range were synthesised by co-condensation of BTME and TEOS in the presence of a surfactant and cobalt ions. The observed Type IV isotherms for the samples had hysteresis loops, which became less pronounced as the ageing temperature was lowered. Large size secondary mesopores were drastically reduced by increasing the amount of TEOS beyond that, at which trimodal porosity was observed. The surface area was found to increase when the ageing temperature was reduced or when the amount of TEOS was increased. PSDs derived from DFT analysis show fewer peaks in the mesopore region than those obtained from a desorption isotherm determined by the BJH method. However, the total number of peaks was the same for both methods. Materials with more dominant X-ray diffraction structural features and larger X-ray diffraction lattice parameters, higher surface areas, lower pore volumes and average pore diameters were obtained when low ageing temperatures were used.
ACKNOWLEDGEMENTS
We wish to thank the University of the Witwatersrand, the South African National Research Foundation and the National Department of Science and Technology for financial support.
REFERENCES
1. Sing KSW, Everett DH, Haul RAW, et al. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Recommendations 1984. Pure Appl Chem. 1985;57:603-619. [ Links ]
2. Beck JS, Vartuli JC, Roth WJ, et al. A new family of mesoporous molecular sieves prepared with liquid crystal templates. J Am Chem Soc. 1992;114:10834-10843. [ Links ]
3. Zhao D, Huo Q, Feng J, Chmelka BF, Stucky GD. Nonionic triblock and star diblock copolymer and oligomeric surfactant syntheses of highly ordered, hydrothermally stable, mesoporous silica structures. J Am Chem Soc. 1998;120:6024-6036. [ Links ]
4. Melde J, Holland BT, Blanford CF, Stein A. Mesoporous sieves with unified hybrid inorganic/organic frameworks. Chem Mater. 1999;11:3302-3308. [ Links ]
5. Hatton B, Landskron K, Whitnall W, Perovic D, Ozin GA. Past, present and future of periodic mesoporous organosilicas - The PMOs. Acc Chem.Res. 2005;38:305-312. [ Links ]
6. Tshavhungwe AM, Coville NJ. In situ and post reaction cobalt incorporation into periodic mesoporous ethanesilica materials. J Sol Gel Sci Tech. 2004;31:161-164. [ Links ]
7. Tshavhungwe AM. Synthesis and characterisation of periodic mesoporous organosilica materials. PhD thesis, Johannesburg, University of the Witwatersrand, 2005. [ Links ]
8. Wahab MA, Kim I, Ha C. Hybrid periodic mesoporous organosilica materials from 1,2-bis(triethoxysilyl)ethane and (3-cyanopropyl)triethoxysilane. Micropor Mesopor Mater. 2004;69:19-27. [ Links ]
9. Yuan Z, Su B. Insights into hierachicallly meso-macroporous structured materials. J Mater Chem. 2006;16:663-677. [ Links ]
10. Wang X, Dou T, Xiao Y. Synthesisis of double-mesopore silica using aqueous ammonia as catalyst. Chem Commun. 1998;1035-1 036. [ Links ]
11. Kuang D, Brezesinski T, Smarsly B. Hierachical porous silica materials with a trimodal pore system using surfactant templates. J Am Chem Soc. 2004;126:10534-10535. [ Links ]
12. Nakanishi K, Kobayashi Y, Amatani T, Hirao K, Kodaira T. Spontaneous formation of hierachical macro-mesoporous ethane-silica monolith. Chem Mater. 2004;16:3652-3658. [ Links ]
13. Gregg SJ, Sing KSW. Adsorption, surface area and porosity. London: Academic Press; 1982. [ Links ]
14. Sing KSW. Adsorption methods for the characterization of porous materials. Adv Colloid Interface Sci. 1998;76-77:3-11. [ Links ]
15. Barrett EP, Joyner LG, Halenda PP. The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J Am Chem Soc. 1951;73:373-380. [ Links ]
16. Ustinov EA, Do DD, Jaroniec M. Adsorption of argon and nitrogen in cylindrical pores of MCM41 materials: Application of density functional theory. Appl Surf Sci. 2005;252(4):1013-1028. [ Links ]
17. Ravikovitch PI, Haller GL, Neimark AV. Density functional theory model for calculating pore size distributions: Pore structure of nanoporous catalysts. Adv Colloid Interface Sci. 1998;76-77:203-226. [ Links ]
18. Ravikovitch PI, Neimark AV. Characterization of nanoporous materials from adsorption and desorption isotherms. Colloid Surface Physicochem Eng Aspect. 2001;187-188:11-21. [ Links ]
 Correspondence to:
Correspondence to:
Alufelwi Tshavhungwe
Postal address: Department of Science and Technology
Private Bag X894
Pretoria 0001, South Africa
email: Alufelwi.Tshavhungwe@dst.gov.za
Received: 16 Nov. 2009
Accepted: 03 June 2010
Published: 26 July 2010
This article is available at: http://www.sajs.co.za