Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Science
versão On-line ISSN 1996-7489
versão impressa ISSN 0038-2353
S. Afr. j. sci. vol.106 no.5-6 Pretoria Mai./Jun. 2010
RESEARCH ARTICLE
Synthesis of zeolite-P from coal fly ash derivative and its utilisation in mine-water remediation
Viswanath R.K. VadapalliI; Wilson M. GitariII; Annabelle EllendtI; Leslie F. PetrikI; Gillian BalfourI
IEnvironmental and Nano Sciences Group, Department of Chemistry, University of the Western Cape, South Africa
IIDepartment of Ecology and Resources Management, University of Venda, South Africa
ABSTRACT
Solid residues resulting from the active treatment of acid mine drainage with coal fly ash were successfully converted to zeolite-P under mild hydrothermal treatment conditions. Scanning electron microscopy showed that the zeolite-P product was highly crystalline. The product had a high cation exchange capacity (178.7 meq / 100 g) and surface area (69.1 m2/g) and has potential application in waste-water treatment. A mineralogical analysis of the final product identified zeolite-P, as well as mullite and quartz phases, which indicated incomplete dissolution of the fly ash feedstock during the ageing step. Further optimisation of the synthesis conditions would be required to attain complete utilisation of the feedstock. The zeolite-P was tested for decontamination potential of circumneutral mine water. High removal efficiency was observed in the first treatment, but varied for different contaminants. The synthesised zeolite-P exhibited a high efficiency for the removal of heavy metal cations, such as aluminium, iron, manganese, zinc, copper and nickel, from contaminated mine water, even with repeated use. For potassium, calcium, strontium and barium, the removal was only efficient in the first treatment and decreased rapidly with subsequent treatments, indicating preferential adsorption of the other metals. A continuous release of sodium was observed during decontamination experiments, which decreased with subsequent treatments, confirming that sodium was the main exchangeable charge-balancing cation present in the zeolite-P product.
Keywords: acid mine drainage; circumneutral mine waters; fly ash; low temperature synthesis; solid residues; zeolite-P
INTRODUCTION
South Africa's leading power-producing company, Eskom, generated nearly 36.7 million tons of fly ash in 2009 from coal combustion, of which only 5.7% was utilised beneficially, mainly in construction industries. The bulk of fly ash is stored in ash dams and landfills, as per the information provided on Eskom's website.1 The ash dams are costly to manage, lead to loss of usable land, impact negatively on the environment, cause air and water pollution and mar landscapes.2 Highly acidic acid mine drainage is another problematic waste stream, resulting inter alia from coal mining, for which the adverse effects are well known.3,4,5
The alkalinity imparted by the high calcium oxide content, together with high surface area and low particle size, makes South African fly ash a suitable neutralising agent for the remediation of acid mine drainage. It has been shown that fly ash can be used successfully to treat acid mine drainage.6 This process offers the opportunity to simultaneously utilise two harmful waste products: acid mine drainage and fly ash, for environmental remediation. The treatment of acid mine drainage with fly ash results in much cleaner process water, but generates a solid residue waste that requires disposal. The solid residue may be suitable as feedstock material for zeolite synthesis because of the high silica and aluminium content. Thus it may be possible to beneficiate the solid residue.
Studies by Querol et al.7 confirm that zeolites have a high commercial value with many industrial applications, such as catalysts for transformation of hydrocarbon feedstocks, sorption of pollutants from waste waters, encapsulation of radioactive wastes and gas separations and as a substitute for phosphates in detergents. Various authors have studied the synthesis of zeolites using fly ash.7,8,9 More recent studies have shown that zeolite-Y (Faujasite) can successfully be synthesised from solid residue at a high temperature (600 ºC).10,11,12,13 Owing to the associated costs resulting from high energy consumption at the high reaction temperature, synthesis of zeolites at high temperatures may not be a feasible option at an industrial scale. Fly ash has been converted successfully into zeolitic phases using a low-temperature (about 100 ºC) method.14,15 However, no studies have been carried out so far on synthesising zeolites using solid residue at low temperature. Therefore, this paper describes the synthesis of zeolitic product from solid residues resulting from neutralisation of acid mine drainage with fly ash, using mild hydrothermal conditions. A two-step process, described by Hollman et al.15, for the synthesis of zeolites was adapted for this study.
Many authors have investigated fly ash derived zeolites for waste- or mine-water remediation.16,17,18 Somerset et al.13 used zeolites synthesised from solid residues at high temperature (zeolite-Y) to remove selected elements from mine water. In general, circumneutral mine waters are partially neutralised acidic mine waters due to interaction with surrounding dolomitic bedrocks. They have a pH of between 6 and 8, high concentrations of calcium, magnesium, potassium and sulphate and significant concentrations of heavy metals. In this study, a zeolite synthesised from solid residues, resulting from neutralisation of acid mine drainage with fly ash using a low-temperature method, was evaluated for decontamination of circumneutral mine waters. This study also evaluated the reusability of the zeolites to treat these mine waters with an objective of optimum utilisation and minimum disposal. The main objectives of this study were, (1) to investigate the conversion of the solid residue into a zeolitic phase at low temperature and (2) to apply the synthesised zeolite in mine-water remediation.
MATERIALS AND METHODS
Sample preparation and starting material
Fresh fly ash from a power station in Mpumalanga Province, South Africa was collected from the precipitators and stored in polyethylene (HDPE) buckets away from direct sunlight. Acid mine drainage was obtained from one of the coal mines in Mpumalanga Province and stored in HDPE containers sealed with airtight lids. All the acid mine drainage samples were refrigerated at 4 ºC. Solid residues were produced from the treatment of acid mine drainage with fly ash at an acid mine drainage: fly ash mass ratio of 3:1. The required volume of acid mine drainage was measured into a beaker and the pH and electrical conductivity were recorded at ambient temperature. A predetermined volume of acid mine drainage was mixed with a predetermined mass of fly ash to give an L:S ratio of 3:1. The mixture was then agitated with an overhead stirrer. The pH and electrical conductivity of the reaction mixture were measured at regular intervals and once the solution reached a neutral pH, the reaction was stopped. Some of the alkalinity in the fly ash thus remained available for further neutralisation. The pH and electrical conductivity was measured using a HANNA HI 991301 portable pH/EC/TDS/Temp probe (Hanna Instruments, Cape Town, South Africa). Prior to measurements, the probe was calibrated using pH 4 and pH 7 buffer solutions (SAAR190444KN and SAAR190555KN, Merck Chemicals, Johannesburg, South Africa) and accuracy of measurements were ascertained by the use of a pH 10 buffer solution (Merck Chemicals). Calibration for electrical conductivity was done by using a 12.88 mS/cm conductivity standard solution (Hanna Instruments). Then the reaction mixture was filtered with a 0.45-µm pore nucleopore membrane (Macherey Nagel, Johannesburg, South Africa) and the filtrate stored in a refrigerator at 4 ºC until analysis. The recovered solid residue was dried in an oven at 100 ºC for 12 h. The dried solid residue was stored in an airtight HDPE container. The composition of acid mine drainage used in this study to prepare solid residues and process water quality after neutralisation with the fly ash is presented in another study.6
Low-temperature synthesis
Low-temperature zeolite synthesis involves two steps, namely ageing or pretreatment and mild-temperature thermal treatment under reflux at approximately 100 ºC. In the ageing step, 20 g of solid residue (Table 1) was mixed with a sodium hydroxide (NaOH) solution (2 mol) that was prepared by dissolving 20 g of NaOH (analytical grade from Merck Chemicals) pellets in 125 mL of ultra-pure water. The resulting synthesis slurry had a molar composition (normalised to silica) of 1.0 Si: 3.3 Na: 3.3 OH: 0.63 Al: 46 H2O. The slurry was aged at temperatures between 40 ºC and 45 ºC by stirring the mixture for 24 h. After this ageing, 125 mL of ultra-pure water was then added to the slurry. The slurry then underwent a thermal treatment by heating at a temperature of approximately 100 ºC under reflux system for 96 h. At the end of the thermal treatment, the synthesised solid product was washed with ultra-pure water until the rinsing water reached a pH of about 9. The solid product was then separated from the solution by centrifugation and dried overnight at 100 ºC.
X-ray fluorescence spectroscopy
The major oxides and trace elements in solid residue feedstock were measured by X-ray fluorescence (XRF) spectroscopy, using a Philips 1404 XRF wavelength dispersive spectrometer (PANalytical, Johannesburg, South Africa), with six crystals and a rhodium X-ray tube target. To prevent interaction of the X-rays with air particles, samples were analysed within a vacuum.
X-ray diffraction analysis
X-ray diffraction (XRD) analysis of solid residue feedstock and zeolitic products was performed using a Philips analytical graphite monochromator (Bruker AXS D8 advance diffractometer) with CuKα radiation and measured in the range of 5º - 70º, degree 2θ at 0.1º intervals and a counting speed of 0.05º/s. The mineral phases present in the sample were identified by comparison with the Joint committee on powder diffraction standards - International centre for diffraction (JCPDS-ICD) reference library that was supplied with the XRD instrument.
Surface area and pore volume determination
The physical analysis of the synthesised zeolitic products was carried out by the application of gravimetric nitrogen Brunauer-Emmett-Teller (N2 BET) surface analysis technique. The sample to be analysed (0.35 g - 0.5 g) was out gassed at 110 ºC on the Flow Prep 060 (Micromeritics Gemini, Johannesburg, South Africa) using helium gas.
Cation exchange capacity determination
Approximately 5 g of zeolite sample was weighed out and placed in a 100-mL polyethylene bottle, to which 25 mL of 1 mol ammonium acetate (NH4C2H3O2) solution was added. The mixture was shaken in a mechanical wrist shaker for 1 h. The supernatant was then filtered directly into a 100-mL volumetric flask through filter paper. Then, 20 mL of ethanol was added to the bottle containing the solids. The bottle was shaken and, after allowing the solid material to settle, the supernatant was filtered into the same 100-mL volumetric flask used earlier. The washing, shaking and filtering procedure was repeated twice. The solution was made up to 100 mL with deionised water. The concentrations of exchangeable cations (Na+, Mg2+, Ca2+ and K+) in the final solution were determined with inductively coupled plasma-mass spectroscopy (ICP-MS) (PerkinElmer, Johannesburg, South Africa) The sum of the equivalents of the exchangeable cations was assumed to be equivalent to the cation exchange capacity (CEC).
Fourier transform infrared spectroscopy
Samples of the solids were taken at different times during the thermal treatment and their respective Fourier transform infrared (FTIR) spectra were compared. The dried samples were ground with potassium bromide to make up a mixture containing 0.05% mass of the sample. A small pellet of each sample was pressed and scanned on a PerkinElmer FTIR spectrometer (PerkinElmer).
Scanning electron microscopy and scanning electron microscopy - energy dispersive spectroscopy analysis
Scanning electron microscopy (SEM) was used to observe the size and shape of crystals in the zeolitic material synthesised. The energy dispersive X-ray spectroscopy (EDS) analyser (Hitachi X-650 scanning electron microanalyser) that was coupled to the SEM was used to identify the chemical composition of a specific area of a sample. Before analysis, the samples were dried, ground to a fine powder, spread on a carbon tape mounted on an aluminium stub and coated with graphite to make them conductive.
Decontamination experiments
Circumneutral mine water (Table 2) was used for these experiments, in which 1 g of the adsorbent (zeolite-P) was mixed with 100 mL of the circumneutral mine water in a beaker. The mixture was stirred for 1 h with an overhead stirrer. At the end of the test, pH and electrical conductivity were recorded. The mixture was then filtered through a 0.45-µm pore nucleopore membrane to remove the adsorbent and the filtrate water sample was sent for major and trace element analysis by ICP-MS. The adsorbent was collected and again mixed with a new aliquot of 100 mL of the respective circumneutral mine water and the experiment repeated as described above. This procedure was repeated four times consecutively.
RESULTS AND DISCUSSION
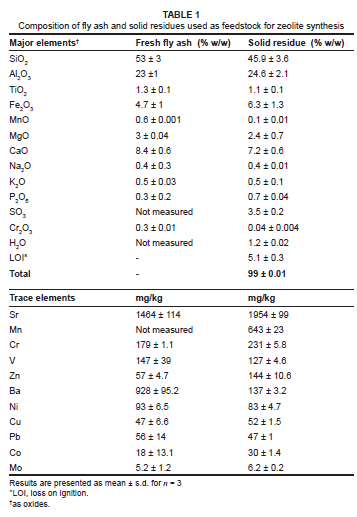
Chemical characterisation of fly ash and solid residues
Table 1 shows the XRF analysis of fly ash and solid residue. The results indicate both the fly ash and solid residue are aluminosilicate materials. Various authors have investigated the influence of the silica : aluminium ratio of the fly ash on the type of zeolites obtained.7,14,19 Somerset et al.10 have indicated that XRF can be used as a predicting tool for the conversion of zeolite using solid residue by analysing the silica and aluminium content. The silica : aluminium ratio of the feedstock (solid residue) in this study was 1.6 and, according to Rayalu et al.20, the solid residue has a sufficient quantity of silicate and aluminium for successful conversion into a zeolite. From Table 1 it is also interesting to see that the solid residue had significant levels of toxic metals incorporated into its structure. Gitari6 has studied the toxic element removal from acid mine drainage using fly ash and he concluded that the primary mechanisms that governed the removal of toxic elements were precipitation, co-precipitation and adsorption of toxic elements at the surface of the fly ash particles and incorporation into the precipitating amorphous aluminium and iron hydroxides and oxyhydroxides. This finding explains the presence of toxic elements in solid residue feedstock.
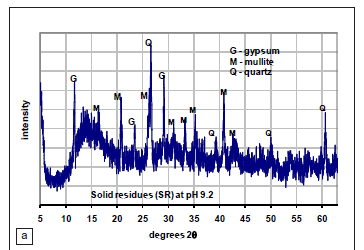
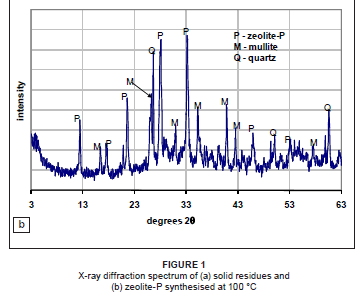
X-ray diffraction spectra
Figure 1 presents the XRD spectrum of the solid residue feedstock and the synthesised product after 24 h of ageing and 96 h of thermal treatment at 100 ºC. The spectra show partial conversion of the solid residues into zeolite-P. Major peaks identified in the synthesised product include mullite and zeolite-P. This indicates that there was incomplete dissolution of mullite in the feedstock under the conditions employed in the synthesis. In addition, the broad hump between 18 and 32 degrees two theta (Figure 1a) indicates that the amorphous glassy phase was still present (Figure 1b), indicating incomplete dissolution. Several authors7,21,22 have noted that increasing the synthesis temperature would increase nucleation and growth of crystals, as well as mullite digestion, leading to an increased silica : aluminium ratio in the solution. This increased ratio, in turn, influences the gel composition and subsequently the zeolite formation. The XRD results indicate that the synthesis temperature employed was not optimum for complete conversion into zeolite-P. This study employed 2 mol NaOH at the ageing step. Woolard et al.23 observed that ageing fly ash with NaOH concentrations greater than 6 mol completely dissolved the mullite in the feedstock.
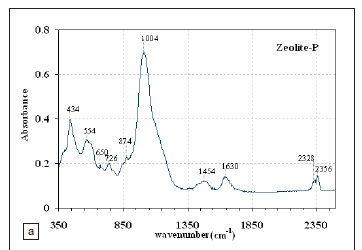
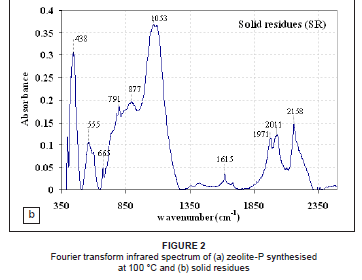
Fourier transform infrared spectra
Figures 2a and 2b present the FTIR spectrum of solid residue feedstock and zeolite-P synthesised at 100 ºC. The bands at 432 cm-1 - 438 cm-1 observed for both solid residue and zeolite-P are attributed to the characteristic aluminosilicate matrix present in both materials.24 In the case of zeolite-P, the bands at 400 cm-1 - 420 cm-1 are also related to the pore opening or motion of the tetrahedral rings which form the pore opening of the zeolites. Bands in the region 420 cm-1 - 500 cm-1 are mostly attributed to internal tetrahedron vibrations of Si-O and Al-O of the solid residues and zeolite-P. The shifting of the T-O band (T = silica or aluminium) at 1053 cm-1, associated with stretching vibrations to lower frequencies (1004 cm-1), indicates zeolitisation of the solid residue feedstock. Musyoka25 observed a shift from 1053 cm-1 to 1004 cm-1 and increased absorbance at 1004 cm-1 as the fly-ash/NaOH slurry was increasingly converted to zeolite-P, with increasing hydrothermal treatment time at a temperature of 100 ºC. The band at 1004 cm-1 is observed to be sharper, indicating increased crystallisation of the zeolitic product and is also observed to have a higher absorbance than in the solid residue feedstock. The bands at 434 cm-1 - 554 cm-1 are clearly resolved in the zeolitic product compared to the solid residues, indicating increased crystallisation of the product.
Scanning electron micrographs and scanning electron microscopy - energy dispersive spectra
Figures 3a and Figures 3b show the SEM of the solid residue feedstock and zeolite-P synthesised at 100 ºC. There is a clear transformation of the spherical particles characteristic of the fly ash used to produce the solid residues (Figures 3a) to a highly crystalline product (Figures 3b). The angular plate-like particles appearing in the solid residues (Figures 3a) are the new mineral phases formed as a result of the reaction of fly ash with acid mine drainage. X-ray diffraction results of the solid residue feedstock indicate the presence of gypsum as one of the new mineral phases formed in the neutralisation process.
The chemical composition by EDS analysis indicates that the zeolite product was rich in aluminium, silica and oxygen, characteristic of a typical zeolite. The presence of iron indicates its possible incorporation into the zeolite crystal lattice framework from the starting material (solid residue). The percentage of different charge-balancing cations determined by EDS show higher levels of calcium than sodium, but some of the calcium could represent the amount of calcium in the unconverted solid residue matrix.
Cation exchange capacity
The exchangeable cations identified in the synthesised product include Na+, Ca2+, Mg2+ and K+. The concentrations of the exchangeable cations were (meq / 100 g): Na+: 134.8, Ca2+: 44.5 and K+: 0.43. Since NaOH was applied as the activator and a source of exchangeable Na+ cations, the Ca2+ and K+ ions identified as exchangeable cations must have originated from the solid residue feedstock. The fly ash used to produce the solid residues was observed to have significant concentrations of calcium and potassium (Table 1). The CEC of the synthesised zeolite-P was taken to be the sum of the concentration of the exchangeable cations (178.7 meq / 100 g). The discrepancy between the relatively high mass percentage of calcium detected by EDS, compared to the low value for the exchangeable cations of potassium and calcium, probably indicates that the majority of the calcium was not available for exchange, more than likely because it was entrapped in the amorphous component of the product matrix. Musyoka25 synthesised a highly crystalline zeolite-P with a CEC of 411 meq / 100 g from coal fly ash at a hydrothermal treatment temperature of 140 ºC. The fact that the zeolite-P prepared at 100 ºC from the solid residues had a low CEC value indicates poor conversion of the feedstock into a zeolitic product and further optimisation of the hydrothermal reaction conditions would be required to produce purer zeolite-P.
Surface area
The synthesised zeolite-P had a surface area of 69.1 m2/g, an average pore volume of 0.004 cm3/g and an average pore size of 4.5 nm. The surface area of the South African fly ash generally varies from 1 m2/g to 2 m2/g. Conversion of the coal fly ash derived solid residue into zeolite-P significantly increased the surface area. Zeolites with similar surface areas were obtained by Woolard et al.23 and Chang and Shih26 on conversion of coal fly ash to zeolites using hydrothermal treatment. However, in another study using solid residue as feedstock material and a high-temperature fusion method, zeolite-Y with a higher surface area (327.3 m2/g) was synthesised.27 The difference in the type and surface area of the synthesised zeolite obtained using the same feedstock material can be attributed to the milder synthesis conditions applied. The XRD spectra (Figure 1) of the zeolitic product indicate that it was a combination of a zeolite-P phase, and unconverted mullite and quartz phases originating from the solid residue feedstock. This highlights the importance of further optimisation of synthesis conditions for complete zeolitisation of the solid residue feedstock.
Mine-water remediation
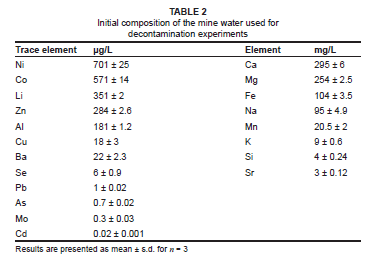
The initial composition of the mine water, prior to the contact with adsorbent, is presented in Table 2. The initial pH of the mine water was 6.2 and the electrical conductivity was 3.8 mS/cm. The concentrations of calcium, magnesium, sodium and iron were in the range of 100 mg/L - 300 mg/L. This indicates that the bedrock where this mine water was generated had a high content of dolomite, which would have contributed to the partial neutralisation of the initial acid mine drainage generated as a result of pyrite oxidation. The high content of the calcium and magnesium resulted from the dissolution of a dolomitic rock.
The pH and electrical conductivity of the waters recovered after the four consecutive decontamination experiments with zeolite-P is shown in Figure 4. A rapid increase in pH to 8.3 was observed in the mine water upon contact with zeolite-P for the first treatment and, thereafter, a gradual decrease to 7.5 was observed after the next three treatments.
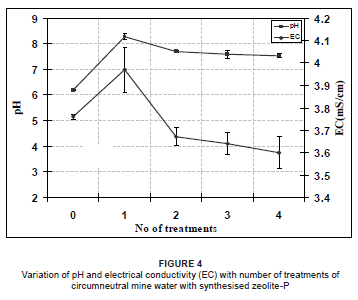
The initial rapid increase in pH was probably a result of the dissolution of the unconverted fly ash matrix. The solid residue feedstock was activated using a NaOH solution and, although the zeolite-P product was rinsed with ultra-pure water until the supernatant reached a pH of 9-10, there were still OH- ions trapped in the micropores of the zeolite-P that were bound to diffuse out when the adsorbent made contact with the aqueous media. The released OH- rapidly increased the pH; on complete diffusion of the OH-, the pH of the solution then stabilised as was observed by the small changes in pH for the second, third and fourth treatments (pH varied from 7.7-7.5).
Electrical conductivity followed the same trend as pH, with a rapid increase to 4 mS/cm for the first treatment and a gradual decrease to 3.6 mS/cm after the next three treatments. It should be noted that the final electrical conductivity was below the initial value of the circumneutral mine water. The initial rapid increase in electrical conductivity was attributed to the initial release of OH- that had remained trapped in the micropores of the zeolite-P even after washing the product with ultra-pure water. Dissolution of unconverted fly ash in the zeolitic product could also have contributed to the rapid increase in electrical conductivity. The subsequent decrease in electrical conductivity after the second, third and fourth treatments was attributed to the removal of the contaminants from the circumneutral mine water on contact with the adsorbent.
The variation of the percentage removal of selected species in the circumneutral mine water with the number of treatments is presented in Figures 5a-5f. There are several processes through which cationic species can be removed by contacting with an adsorbent, including ion exchange, adsorption and precipitation of metal hydroxides.18
Sodium was released into the solution in high concentrations in the first treatment and the rate of release decreased with subsequent treatments (Figure 5a). The Na+ cation accounted for the high CEC in the synthesised zeolite-P and would have been exchanged for other cations in solution, which would explain the continuous release of Na+ after contacting the circumneutral mine water with the adsorbent. The subsequent decrease in the release of Na+ with the number of treatments reflects the exhaustion of the exchange sites in the adsorbent by uptake of the contaminant species. The K+ cations were removed in the first and second treatments but were exchanged out of the zeolite in the third and fourth treatments (Figure 5a) indicating, again, the exhaustion of the exchange sites and probably the competitive adsorption with other species for the exchange sites available in the zeolite.
The Ca2+ and Sr2+ cations showed similar removal trends. They registered high removal rates for the first and second treatments but started to leach at the third and fourth treatments (Figure 5c). The Ba2+ cation was completely removed after the second treatment. However, similar to Ca2+ and Sr2+, it started to leach out during the third and fourth treatments (Figure 5b). Among the cations Ca2+, Sr2+, Mg2+ and Ba2+, zeolite-P showed a strong preference for Sr2+. The subsequent release of Ca2+, Sr2+ and Ba2+ during the third and fourth treatments indicates competitive adsorption of other metal ions or exhaustion of the exchange sites. The increased release of Ba2+ during the fourth treatment was probably as a result of the dissolution of unconverted solid residues in the zeolitic product. The Mg2+ cation showed marginal release (-7% to -13% removal) through the four consecutive treatments, meaning that it was not adsorbed but remained largely in solution.
The heavy metal cations Fe2+/Fe3+, Al3+ and Mn2+ showed the highest removal rates: iron (99.1% - 99.9%), aluminium (68% - 77%), manganese (8% - 64%) (Figure 5d). These heavy metal cations are sensitive to pH changes and may have precipitated out at the right pH conditions. The pH of the supernatant was above 10 for all treatments (Figure 4). At a pH > 6 both Fe2+/Fe3+ were precipitated as iron oxyhydroxide,28 while at a pH > 4 Al3+ was precipitated as aluminium oxyhydroxide. The high removal rates were ascribed to the precipitation of these species as metal-oxyhydroxides at a circumneutral pH (pH 7.5-8.3) generated on contacting the circumneutral mine water with the adsorbent (Figure 4). The effect of pH was best exhibited by the trends in Mn2+. The Mn2+ cation precipitated out of solution at a pH of 8-9 due to oxidation, with the highest removal rate (64.3%) being observed at a pH of 8.28. The removal rate was observed to decrease as the pH decreased to 7 with subsequent treatments.
The Zn2+, Cu2+ and Ni2+ cations exhibited fairly high removal rates (58% - 69.2% for zinc, 43% - 68.6% for nickel and 54% - 88.9% for copper) (Figure 5e). The Cu2+ removal trend resembled that of Al3+, indicating that Cu2+ could have adsorbed on the precipitated aluminium oxyhydroxide. The zinc and nickel removal rates seemed to decrease gradually with subsequent treatments. The removal trends of zinc and nickel decreased with a decrease in pH, which perhaps indicates removal through precipitation. However, it was beyond the scope of this paper to distinguish which proportions of metal contaminants were removed through ion-exchange, adsorption or precipitation.
A rapid increase in selenium removal was observed during the second treatment (from 13.98% for the first treatment to 61% for the second treatment) (Figure 5f), but selenium was released into the solution during the third and fourth treatments. At a circumneutral pH, selenium exists as an oxyanion and the high removal rates could have been through co-precipitation with Fe- and Al- oxyhydroxides.
Because the experiments were not carried out in a closed system, the adsorption data, such as removal of Cu2+, is tentative.
CONCLUSION
Solid residue resulting from active treatment of acid mine drainage with fly ash was successively converted to zeolite-P under mild hydrothermal treatment conditions. Mineralogical analysis of the final zeolitic product showed the presence of mullite and quartz, which indicated incomplete dissolution of the solid residue feedstock. The SEM, CEC and BET analyses showed the synthesised zeolite-P product was crystalline, had a high CEC and an enhanced surface area compared to the feedstock material. The results indicated that conversion of the solid residue feedstock into zeolite-P resulted in a coal fly ash beneficiated, high-capacity adsorbent that can subsequently be applied in environmental remediation of mine water.
The synthesised zeolite-P was tested for decontamination of circumneutral mine water to show its applicability for environmental remediation. Its reusability was also assessed by reusing the same portion of the adsorbent for up to four consecutive treatments. The synthesised zeolite-P was observed to be highly efficient in the removal of the heavy metal cations Al3+, Fe3+, Mn2+, Zn2+, Cu2+, and Ni2+, even with repeated use. For K+, Ca2+, Sr2+ and Ba2+ the removal was efficient in the first treatment but decreased rapidly with subsequent treatments, indicating competitive adsorption or exhaustion of exchange sites by uptake of contaminant species. A rapid and continuous release of Na+ was observed that decreased with subsequent treatments, indicating that Na+ was the main exchangeable charge balancing cation incorporated in the zeolite during synthesis. However, it should be noted that the maximum removal efficiency for most of the contaminants was achieved in the first treatment and this varied for different contaminants.
ACKNOWLEDGEMENTS
The authors are grateful to the Water Research Commission, the South African National Research Foundation, Coaltech 2020 and the Claude Leon Foundation for their financial support.
REFERENCES
1. Eskom. Annual report [homepage on the Internet]. c2009 [cited 2010 May 18]. Available from: http://eskom.co.za/annreport09 [ Links ]
2. Carlson CL, Adriano DC. Environmental impacts of coal combustion residues. J Environ Qual. 1993;22:227-247. [ Links ]
3. Baird C. Environmental chemistry. New York: WH Freeman & Co; 1995. [ Links ]
4. Bell FG, Halbich TFJ, Bullock SET. The effects of acid mine drainage from an old mine in the Witbank coalfield, South Africa. Quart J Eng Geol Hydrogeol. 2002;35(3):265-278. [ Links ]
5. Fajt J, Kabrna M, Tichy R, Ledvina R. Environmental risks associated with aeration of a freshwater sediment exposed to mine drainage water. Env Geol. 2002;41(5):563-570. [ Links ]
6. Gitari W. Evaluation of leachate chemistry and contaminant attenuation by fly ash blended acid mine drainage/fly ash solid residues. PhD thesis, Cape Town, University of the Western Cape, 2006. [ Links ]
7. Querol X, Plana F, Alastuey A, Lopez-Soler A. Synthesis of Na-zeolites from fly ash. Fuel. 1997;76(8):793-799. [ Links ]
8. Querol X, Umana JC, Plana F, et al. Synthesis of zeolites from fly ash at pilot plant scale: Examples of potential applications. Fuel. 2001;80(6):857-865. [ Links ]
9. Ojha K, Pradhan NC, Samanta AN. Zeolite from fly ash: Synthesis and characterization. Bull Mater Sci. 2004;27(6):555-564. [ Links ]
10. Somerset VS, Petrik LF, White RA, Klink MJ, Key D, Iwuoha E. The use of X-ray fluorescence (XRF) analysis in predicting the alkaline hydrothermal conversion of fly ash precipitates into zeolites. Talanta. 2004;64:109-114. [ Links ]
11. Somerset V, Petrik LF, Etchebers O, White R, Key D, Iwuoha E. Acid mine drainage transformation of fly ash into zeolitic crystalline phases. Fresenius Environ Bull. 2005;14(10):1074-1076. [ Links ]
12. Somerset VS, Petrik LF, White RA, Klink MJ, Key D, Iwuoha EI. Alkaline hydrothermal zeolites synthesized from high SiO2 and Al2O3 co-disposal fly ash filtrates. Fuel. 2005;84(18):2324-2329. [ Links ]
13. Somerset V, Petrik LF, Iwuoha EI. Alkaline hydrothermal conversion of fly ash filtrates into zeolites 2: Utilization in wastewater treatment. J Environ Sci Health Part A. 2005;40(8):1627-1636. [ Links ]
14. Inada M, Eguchi Y, Enomoto N, Hojo J. Synthesis of zeolite from coal fly ashes with different silica-alumina composition. Fuel. 2005;84:299-304. [ Links ]
15. Hollman GG, Steenbruggen G, Janssen-Jurkovičová M. A two-step process for the synthesis of zeolites from coal fly ash. Fuel. 1999;78:1225-1230. [ Links ]
16. Moreno N, Querol X, Pereira CF, Jurkovicova MJ. Utilization of zeolites synthesized from coal fly ash for the purification of acid mine waters. Environ Sci Technol. 2001;35:3526-3534. [ Links ]
17. Fungaro DA, De Carvalho Izidoro J. Remediation of acid mine drainage using zeolites synthesized from coal fly ash. Quim Nova. 2006;29(4):735-740. [ Links ]
18. Rios CA, Williams CD, Roberts CL. Removal of heavy metals from acid mine drainage (acid mine drainage) using coal fly ash, natural clinker and synthetic zeolites. J Hazard Mater. 2008;156(1-3):23-35. [ Links ]
19. Inada M, Tsujimoto H, Eguchi Y, Enomoto N, Hojo J. Microwave-assisted zeolite synthesis from coal fly ash in hydrothermal process. Fuel. 2005;84(12-13):1482-1486. [ Links ]
20. Rayalu S, Meshram SU, Hasan MZ. Highly crystalline faujasitic zeolites from fly ash. J Hazard Mater. 2000;77(1-3):123-131. [ Links ]
21. Feijen EJP, Martens JA, Jacobs PA. Zeolites and related microporous materials: State of the art. Stud Surf Sci Catal. 1994;84(Part A):3-21. [ Links ]
22. Catalfamo P, Corigliano F, Primerano P, Pasquale SD. Study of the pre-crystallisation stage of hydrothermally treated amorphous aluminosilicates through the composition of the aqueous phase. J Chem Soc. 1993;89(1):171-175. [ Links ]
23. Woolard CD, Petrus K, Van der Horst M. The use of a modified fly ash as an adsorbent for lead. Water SA. 2000;26(4):531-535. [ Links ]
24. Fernández-Jiménez A, Palomo A. Mid-infrared spectroscopic studies of alkali-activated fly ash structure. Microp Mesop Mater. 2005;86:207-214. [ Links ]
25. Musyoka NM. Hydrothermal synthesis and optimisation of zeolite Na-P1 from South African coal fly ash. MSc thesis, Cape Town, University of the Western Cape, 2009. [ Links ]
26. Chang HL, Shih WH. A general method for the conversion of fly ash into zeolites as ion exchangers for cesium. Ind Eng Chem Res. 1998;37:71-78. [ Links ]
27. Petrik LF, Hendricks N, Ellendt A, Burgers C. Water research commission report No. 1546/1/07. Johannesburg: Water Research Commission; 2007. [ Links ]
28. Gitari WM, Petrik LF, Etchebers O, Key DL, Iwuoha E, Okujeni C. Utilization of fly ash for treatment of coal mines waste water: Solubility controls on major inorganic contaminants. Fuel. 2008;87:2450-2462. [ Links ]
 Correspondence to:
Correspondence to:
Leslie Petrik
Postal address:
Environmental and Nano Sciences Group, University of the Western Cape
Private Bag X17, Bellville
7535, South Africa
email: lpetrik@uwc.ac.za
Received: 22 Aug. 2008
Accepted: 19 Feb. 2010
Published: 09 June 2010
This article is available at: http://www.sajs.co.za