Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.106 n.1-2 Pretoria Jan./Feb. 2010
RESEARCH LETTER
Apoptosis-promoting effects of Sutherlandia frutescens extracts on normal human lymphocytes in vitro
Vanessa C. Korb; Devapregasan Moodley; Anil A. Chuturgoon
Discipline of Medical Biochemistry, Nelson R Mandela School of Medicine, University of KwaZulu-Natal, Durban, South Africa
ABSTRACT
Sutherlandia frutescens (SF), an indigenous medicinal plant to South Africa, is traditionally used to treat a diverse range of illnesses. More specifically, the immune-enhancing potential of SF has been recognised to the extent that SF extracts have been recommended as an adjuvant in HIV/AIDS treatment by the South African Ministry of Health, despite a lack of knowledge of its mechanism of action or potential immune toxicity. As yet, unsubstantiated data support the notion of immunostimulatory effects of SF extracts in HIV-infected patients. This was suggested by post-treatment recovery of CD4+ cells brought about by the reduction of the impact of virus-induced apoptosis. This study investigated the apoptotic effects of SF extracts on normal human lymphocytes in vitro. Initially, an acute cytotoxic profile of SF extract was formulated, from which an IC50 of 7.5 mg/mL was calculated and administered for 3 h, 6 h and 12 h to cell populations. At 12 h, SF caused a significant increase in apoptosis in the total lymphocyte population and CD4+ cells as evidenced by increased phosphatidylserine (PS) translocation, caspase-3/7 activity, and decreased ATP content. After 12 h, the SF extract initiated lymphocyte activation in both total lymphocyte and CD4+ subpopulations, indicated by a doubling of the number of cells expressing the CD69 activation marker. The apoptosis observed may thus be the result of activation-induced lymphocyte cell death (AICD). Our results are in conflict with preliminary clinical evidence which has suggested SF extracts are possibly beneficial in the treatment of HIV infection. More extensive evaluations of the effects of SF extracts on the immune system in such subjects are urgently needed.
Keywords: Sutherlandia frutescens; immune cells; apoptosis; mitochondrial depolarisation; caspases
INTRODUCTION
Sutherlandia frutescens (SF), an indigenous member of the Leguminosae family, commonly known as 'cancer bush', is a multipurpose medicinal plant endemic to South Africa.1 There is currently much interest in its proposed anti-oxidant, anti-inflammatory and anticarcinogenic potential. Leaf extracts of SF have traditionally been used by people, such as the Khoi and Nama, to treat a diverse range of ailments.1-3 More recently, SF extracts have been suggested as a treatment for internal cancers, and a possible immune enhancer in HIV/AIDS.1,4 Despite limited knowledge of the pharmacological properties, efficacy and toxicity of SF extracts of the plant are currently recommended by the South African Ministry of Health as an adjuvant to existing antiretroviral therapies.5 The evidence supporting its proposed chemotherapeutic and immunostimulatory effects is highly preliminary and the mechanisms of action are still largely undetermined.6
The therapeutic effects of SF have generally not been attributed to a single active ingredient, but rather to synergism between a number of phytocompounds, resulting in a multifaceted mechanism of action. The biologically active ingredients of SF are reported to include L-canavanine, gamma-aminobutyric acid (GABA) and D-pinitol. The plant is also rich in a number of amino acids, and contains trace amounts of saponins, tannins, flavonoids and parabens.3,7 Both GABA and D-pinitol have been proposed to improve wasting conditions in HIV/AIDS and cancer patients by the inhibition of the action of pro-inflammatory cytokines, specifically TNF-α and IL-1β.7,8,9 L-canavanine is a non-protein amino acid structurally analogous to L-arginine,10,11 and can displace L-arginine in growing polypeptide chains, leading to aberrant protein synthesis and function.12 L-canavanine has been documented as possessing antiviral effects against influenza and HIV,13 possibly by interfering with viral protein synthesis and function. D-pinitol has insulin-like properties and is useful in stimulating cellular metabolism.8
HIV is characterised by the progressive depletion of CD4+ T-lymphocytes, resulting eventually in the onset of AIDS. Loss of CD4+ cells causes impairment of cell-mediated immune responses, which leads to opportunistic infections. A number of pathophysiological mechanisms have been suggested to contribute to the extensive CD4+ cell lymphocytopenia, but non-selective enhanced apoptosis is regarded as the primary cause.14 Continuous HIV replication causes chronic activation of the immune system, which enhances apoptotic activity in CD4+ cells. Studies in animal models have shown viral replication to be dependent on, and more effective in activated, rather than quiescent lymphocytes.15,16 Chronic immune stimulation and activation leads to a reduced ability to maintain an adequate immune response.
Administration of SF preparations to HIV patients has led to improved CD4+ cell counts and reduced viral loads; weight gain and increased energy levels were also reported, although the mode of action has not been identified.4,17 These effects may have been due to SF-induced reduction of apoptotic activity in circulating lymphocytes, specifically CD4+ cells. The effects of SF on apoptosis in normal circulating lymphocytes have, surprisingly, not been evaluated to date, which prompted the present study of the cytotoxic and apoptotic effects of SF extracts on normal human lymphocytes.
There has been an increasing trend in many African countries towards the use of traditional medicines as an alternative to allopathic treatments, providing a less expensive and more culturally accepted treatment approach. Much research into the mechanisms, efficacy and potential toxicity of these remedies is required.18,19,20
MATERIALS AND METHODS
This study received ethical approval from the Biomedical Research Ethics Administration Office of the University of KwaZulu-Natal (Reference number: BE063/08). Informed consent was obtained from the volunteers (all of whom were male) before blood samples were taken.
Extraction of peripheral blood mononuclear cells
Buffy coats containing peripheral blood mononuclear cells (PBMC) were extracted from heparinised whole blood by differential centrifugation. Briefly, 5 mL whole blood isolated from healthy adult male donors, was carefully layered onto equivolume Histopaque-1077 (Sigma-Aldrich, South Africa) in 15 mL polypropylene tubes and centrifuged at 400 g for 30 min at room temperature. Following this, buffy coats were aspirated into new polypropylene tubes and washed twice in phosphate buffered saline (PBS) (400 g, 20 min). Cell density was determined by trypan blue exclusion and manual cell counting on a haemocytometer adjusted to a density of 1 × 106 cells/mL.
Extraction of Sutherlandia frutescens active compounds
Ethanol extracts of SF were prepared as follows:
Commercially available tablets (Phyto-Nova Sutherlandia SU1TM Immune Booster; 9.0 g), each containing approximately 300 mg active ingredient, were crushed and extracted in 30% ethanol for 4 h at room temperature. Thereafter, insoluble material was removed by centrifugation (400 g, 5 min, room temperature) and the supernatant containing active ingredients was filter-sterilised (0.45 µm). This SF stock solution was subsequently serially diluted (10.0, 7.5, 5.0, 2.5, 1.0, 0.5, 0.1, 0.05 and 0.01 mg active ingredients/mL) in complete culture medium (CCM; RPMI-1640 suspension medium supplemented with 10% (v/v) foetal calf serum, 1% (v/v) L-glutamine and 2% (v/v) penicillin-streptomycin-fungizone solution) for treatments.
Treatment protocol
PBMCs were seeded into a microtitre plate (3.0 × 105 cells/well) and treated in triplicate with serial dilutions of SF extract and the corresponding ethanol vehicle control at 37 ºC for 3 h, 6 h and 12 h, and longer in certain cases as indicated.
MTT cell viability assay
After each incubation period, PBMCs were washed twice in PBS and each sample supplemented with a 1:5 ratio of filter-sterilised (0.45 µm) MTT salt solution (5 mg/mL in PBS) and CCM. The plate was incubated at 37 ºC for 4 h. Following incubation, the cells were pelleted (400 g, 10 min, room temperature), resuspended in dimethyl sulphoxide (DMSO) (100 µL) and incubated at 37 ºC for a further 1 h. Optical density of the formazan product was measured by an enzyme-linked immunosorbent assay (ELISA) plate reader (Bio-tek µQuant) at 595/655 nm. Results were expressed as mean percentages of the control response.
IC50 calculation from dose-response curves
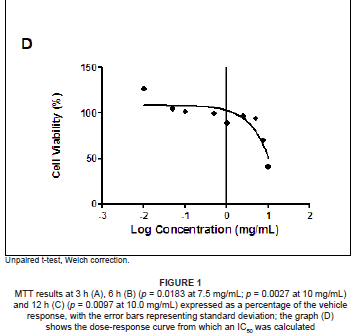
Based on MTT cell viability results, a dose-response curve was plotted for each time period (using GraphPad Prism v5.0 software, GraphPad Software Inc., La Jolla, USA) to ascertain the concentration of SF extract that produced a 50% inhibition (IC50) of live cells by linear extrapolation. The IC50 value (7.5 mg/mL active ingredients) and corresponding vehicle (1.0125% ethanol in CCM) were used for PBMC treatments in subsequent assays.
Intracellular ATP quantification
Quantities of 1 × 105 of SF extract treated and untreated PBMCs were seeded into a white luminometry plate to which the ATP CellTitre Glo (Promega, Madison, USA) reagent (10 µL) was added and allowed to react in the dark for 10 min at room temperature. Following incubation, the luminescent signal proportional to the cellular ATP content was detected with a microplate luminometer (Turner Biosystems, Sunnyvale, USA). Results were expressed as mean relative light units (RLU).
Apoptosis studies
Detection of phosphatidylserine externalisation
An Annexin-V FITC apoptosis detection kit (Roche, Johannesburg, South Africa) was used to detect externalised phosphatidylserine (PS) on PBMCs. Briefly, 5 × 105 SF-extract treated and untreated cells were transferred to polystyrene cytometry tubes and stained with 5 µL of both the Annexin-V FITC and propidium iodide (PI) components by incubation in the dark for 15 min at room temperature. Thereafter, the samples were supplemented with 400 µL of Annexin-V Binding Buffer (1×). Labelled PBMCs were detected by flow cytometry (FACS Calibur; BD Biosciences, Johannesburg, South Africa). Data was collected for 50 000 events per sample and analysed with FlowJo 7.1 software (Tree Star Inc., Ashland, USA).
Assessment of PBMC mitochondrial membrane potential
The JC-1 Mitoscreen assay (BD Biosciences) was utilised to assess PBMC mitochondrial membrane potential. Manufacturer's guidelines were followed. Briefly, 5 × 105 PBMCs (SF-extract treated and untreated) were transferred to polystyrene cytometry tubes and incubated (15 min at room temperature) with 150 µL of JC-1 dye. Thereafter, PBMCs were washed twice in JC-1 wash buffer (1×) and re-suspended in 200 µL flow cytometry sheath fluid. Labelled PBMCs were enumerated by fluorescence-activated cell sorting (FACS). Data was analysed with FlowJo 7.1 software and expressed as a percentage of cells containing depolarised mitochondria.
Luminometric evaluation of caspase-3/7 activity
A quantity of 1 × 105 PBMCs (treated with SF extract and untreated) was aliquotted into a white luminometer plate, following which 10 µL of the Caspase-Glo® 3/7 reagent (Promega) was added and allowed to react. Plates were agitated at low speed for 30 min. Following incubation, the luminescent signal produced by mono-oxygenation of amino-luciferin was measured with a microplate luminometer. The results were expressed as RLU.
Analysis of lymphocyte surface markers - CD69 and CD4
Anti-CD4 (APC) and anti-CD69 (FITC) monoclonal antibodies (BD Biosciences) were used to identify CD4+ lymphocyte subsets and activated lymphocytes, respectively. In each case, 5 × 105 PBMCs (treated with SF extract and untreated) were transferred into polystyrene cytometry tubes and supplemented with 2 µL of each antibody and thereafter incubated in the dark. Labelled lymphocytes were enumerated by FACS. The anti-CD4 antibody (2 µL) was included in both the JC-1 Mitoscreen and Annexin-V assays in order to specifically analyse the effects of SF on apoptosis in the CD4+ T-cell subset.
Statistical analysis
Statistical analyses were performed using GraphPad Prism v5.0 software.
RESULTS
Cell viability at high concentrations of SF extract
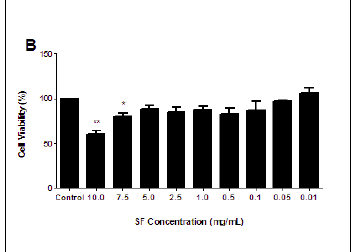
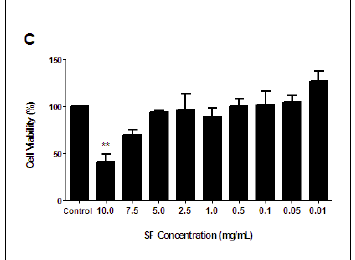
The MTT assay measures cell viability based on the NADH/NADPH reductase system characteristic of metabolically active cells. There was no significant decrease in the cell viabilities of the SF extract treated PBMCs at 3 h (ranging from 87.04% to 114.55%), and this treatment could not yield a 50% inhibitory concentration (Figure 1A). Exposure to SF extract over 6 h had a more marked effect on the metabolic activities of the PBMCs (Figure 1B), with significant cell death of 39.35% at 10.0 mg/mL (p = 0.0027) and 19.98% at 7.5 mg/mL (p = 0.0183). After 12 h, the SF extract increased cell mortality further so that a concentration of 10.0 mg/mL decreased cell viability by more than half (58.9%, p = 0.0097; Figure 1C). An IC50 of 7.5 mg/mL SF extract was calculated and thereafter used in subsequent apoptotic assays (Figure 1D).
SF extract significantly decreased the ATP content of PBMC at 12 h
The CellTitre-Glo assay measures cell viability as a function of cellular ATP content. At 3 h, there was no significant difference between the ATP concentrations of SF-extract treated and untreated cells (4.725 × 106 ± 2.508 × 105 RLU vs 4.192 × 106 ± 5.506 × 105 RLU). In contrast, PBMCs treated with SF extract over 6 h showed a significant (p = 0.0108) 1.2-fold decrease in the ATP concentration (6.645 × 106 ± 3.360 × 105 RLU vs 8.624 × 106 ± 5.012 × 105 RLU). At 12 h, there was a further significant (p = 0.0059) 1.8-fold reduction in the measured ATP concentration in the SF extract treated PBMCs, as compared to the controls (4.569 × 106 ± 4.570 × 104 RLU vs 8.331 × 106 ± 5.017 × 105 RLU). The above results are represented in Figure 2.

SF extract significantly increased PS translocation at 12 h
Externalisation of PS is an early characteristic of apoptosis, which is determined by labelling of cells with Annexin-V. After 3 h, SF extract did not significantly alter the percentage of lymphocytes undergoing apoptosis (4.72% vs 5.1%), but the CD4+ subset of cells showed a twofold increase in apoptosis (3.18% vs 6.24%). After 6 h, the SF extract substantially increased (1.4-fold) lymphocyte apoptosis (5.96% vs 8.28%) and CD4+ cell apoptosis (1.5-fold; 4.74% vs 6.89%). After 12 h, the SF extract induced a 2.3-fold and threefold increase in apoptosis in both the treated lymphocyte (11.3% vs 25.5%) and CD4+ (5.11% vs 15.4%) populations as compared to untreated controls. The above results are represented in Table 1A.
SF extract reduced mitochondrial depolarisation at 3 h and 6 h, but not at 12 h
The JC-1 Mitoscreen assay was performed to assess mitochondrial membrane potential (ΔΨm). At 3 h, and most notably 6 h, SF extract treatment decreased the percentage of PBMCs with depolarised mitochondria by 9.8% and 22.7%, respectively. This effect was mirrored to a lesser extent in the CD4+ cells, with decreases of only 2.71% at 3 h and 7.23% at 6 h. At 12 h, SF extract treatment had no significant effect on the percentage of PBMCs containing depolarised mitochondria (36.7% vs 36.2%). SF extract slightly increased mitochondrial depolarisation (12% vs 13.1%) in the CD4+ cells. The above results are represented in Table 1B.
SF extract reduced capase-3/7 activity at 3 h and 6 h, but increased activity at 12 h
The Caspase-Glo assay quantifies the activity of caspases 3 and 7 as an indicator of apoptosis. At 3 h and 6 h, SF extract treatments reduced (non-significantly) the caspase activity in the PBMCs (1.950 × 105 ± 4.936 × 104 RLU vs 1.414 × 105 ± 1.718 × 104 RLU, 2.349 ×105 ± 1.192 × 104 RLU vs 2.113 × 105 ± 3.820 × 103 RLU, respectively) At 12 h, however, SF extract induced a twofold increase in caspase 3 and 7 activity in the cells (3.249 × 105 ± 6.871 × 104 RLU vs 7.215 × 105 ± 1.556 × 105 RLU). The above results are represented in Figure 3.

SF extract induced lymphocyte activation at 12 h
The lymphocyte activation marker, CD69, is characterised as the earliest marker, transiently induced after lymphocyte activation. After 3 h and 6 h of SF extract treatment, there was a clear increase in activation of cells in both the total lymphocyte population and CD4+ subset, the most apparent increase being 0.38% (0.84% vs 1.22%) at 3 h in the CD4+ cells. At 12 h, the SF extract induced a twofold increase in activation in both the PBMC and CD4+ lymphocytes (2.13% vs 4.49% and 1.31% vs 2.41%, respectively). The above results are represented in Table 1C.
DISCUSSION
SF extracts are reputedly a broad-spectrum 'tonic', effective in the treatment of a wide range of ailments. Recently, they have been proposed as an immune enhancer and supplement to conventional HIV/AIDS therapies.4,17 Because apoptosis is the principal mechanism of HIV pathogenesis, the therapeutic effects of SF extracts in HIV/AIDS may occur by influencing lymphocyte apoptotic responses.14
Apoptosis is a tightly regulated process of cell death characterised by specific morphological changes and biochemical events within a cell.21 It can be initiated by a diverse range of internal or external stimuli and does not elicit inflammatory responses.22 Apoptosis is important in normal tissue homeostasis and immunoregulatory processes.23 It has, however, been implicated as a significant contributor to the pathophysiology of various diseases, either by excessive activity, or suppressed induction, such as in tumourigenesis. Thus, the apoptotic pathway provides an obvious target of pharmacological manipulation.21, 24, 25
An early characteristic of apoptosis is the externalisation of PS (death signal) on the cell membrane, which provokes phagocytosis of apoptotic cells by macrophages without initiating an inflammatory response. SF-extract therapy stimulated translocation of PS in Jurkat cells.26 In this study, SF extracts caused a time-dependent increase in apoptosis in total lymphocytes which was more pronounced in the CD4+ sub-population of these cells.
Several biochemical and functional alterations of mitochondria are associated with the apoptotic process. Depending on the death pathway, the mitochondria can act as an executioner or facilitator of apoptosis. Mitochondrial depolarisation is a critical event in apoptosis which leads to the uncoupling of ATP production, cessation of the electron transport chain (ETC), and inhibition of the Krebs cycle. Depolarisation is caused by the induction of permeability transition (PT) pores in the inner mitochondrial membrane, leading to loss of membrane potential and selective permeability.27, 28, 29, 30
Mitochondrial dysfunction results in aberrant metabolic activity of the ETC. In this study, the SF extracts decreased cellular metabolic activity as observed from the MTT results. This may be due to a decrease in the rate of the Krebs cycle resulting in a decreased production of reducing equivalents (NADH and FADH2) required for the reduction of the MTT salt to its formazan product by succinate dehydrogenase. SF extracts decreased cell viabilities in a time-dependent manner so that over all time periods there was a trend in the metabolic effects of treated cells - lesser doses increased cell viability, contrastingly high doses (7.5 mg/mL and 10.0 mg/mL) decreased viability. The increased metabolic rate at the lower SF extract-doses may be attributed to D-pinitol, which has insulin-like properties.17
A predominant function of the mitochondria is aerobic respiration in order to generate the majority of the cell's ATP fuel by chemiosmosis.31 The mitochondrial proton gradient couples the ETC to produce ATP.32 Thus, the observed time-dependent aberrations in the mitochondrial NADH reductase system supports the corresponding time-dependent decrease in total cellular ATP concentration. This demonstrates that SF extract may exert a negative effect on ATP generation by the mitochondrial respiratory chain, thereby depleting ATP concentrations.
SF-extract-induced loss of cell viability and ATP content would be associated with a collapse of the electrochemical gradient across the outer mitochondrial membrane. This study shows that at shorter time periods, SF extract conserves this potential, as demonstrated by a reduced percentage of lymphocytes containing depolarised mitochondria. The SF extract may confer mitochondrial membrane protection, but this does not translate to uncompromised mitochondrial functioning as shown by decreases in cell viabilities and consequent ATP production. At 12 h, the percentage of depolarised mitochondria was the same in SF-extract treated and untreated cells, while treated cells showed decreases in ATP production and cell viability at this time period despite the unchanged mitochondrial polarity. The effects of SF extract may not necessarily have been confined to manipulations of the mitochondrial membrane potential, but may have also affected other transmembrane proteins, including uncoupling proteins which interfere with oxidative phosphorylation.28,29
The caspases mediate a series of morphological changes in a cell undergoing apoptosis. The SF extract reduced caspase-3 and caspase-7 activities after 3 h of treatment. Together with the low levels of apoptosis observed, it seems that SF extract did not stimulate the apoptotic process in PBMCs after such short exposure times. The observation that SF extract reduced caspase activity may be a possible mechanism whereby the PBMCs are protected from cellular death and thus support the protection conferred on the mitochondrial membrane. Caspase activity in SF extract-treated cells was also reduced at 6 h, despite an increase in observed apoptosis. Externalisation of PS occurs earlier in apoptosis than terminal caspase activation. Our data indicates that lymphocyte apoptosis is detected only after 6 h of exposure to SF extract and may represent the initiation phase of apoptosis. In contrast, caspase-3 and caspase-7 activities were increased at 12 h, indicating that SF extract stimulated apoptosis at this time period; this was consistent with the externalisation of PS, an effect observed in both PBMCs and CD4+ cells. The apoptotic response may not be induced by mitochondrion-mediated signals when the cells are treated with SF extract but instead may be a result of death receptor ligation or immune activation.
The increases in lymphocyte apoptosis induced by SF extract observed in this in vitro study are consistent with published data on apoptotic studies in cancerous cell lines.7,17,26 They are in conflict with the claim of improved CD4+ counts in HIV patients,4,17 especially as the apoptotic effect in whole lymphocyte populations was mirrored in the CD4+ lymphocyte subset.
The overall apoptosis-promoting effects of SF extract were complicated by contradictory observations at different time points and concentrations. For example, SF extract had a protective effect on mitochondrial membranes, which would tend to decrease apoptosis. The activation marker CD69 is the earliest marker transiently induced after lymphocyte activation and its expression rapidly declines in the absence of lymphocyte activating stimul.33 Ligand cross-linking of the CD69 protein results in cellular activation processes mediated by protein kinase C leading to lymphocyte proliferation and the induction of specific genes.34 SF-extract treatment over 3 h and 6 h did not induce lymphocyte activation as demonstrated by unchanging levels of CD69+ lymphocytes. In contrast, at 12 h, SF-extract treatment was associated with a doubling of the number of activated lymphocytes indicating that longer periods of SF extract exposure may have led to lymphocyte activation.
Lymphocytes may be activated either by classical antigen processing and presentation, or in a mitogenic manner by biochemical agents. The low molecular weight of the active ingredients in SF extracts7,17 may act as mitogens to induce lymphocyte activation.
The response of lymphocytes to an antigen is varied. Some can survive as memory cells for extended periods of time while others have a limited lifespan in order to control the reaction and prevent lymphoid enlargement in response to moderate antigenic stimulation.35 These end-cells undergo apoptosis via activation-induced lymphocyte cell death (AICD) once an effective cell-mediated and antibody immune response has been mounted.35 The apoptosis observed in the lymphocytes following SF-extract treatment may thus occur via AICD as a consequence of SF-extract-induced lymphocyte activation. CD4+ lymphocytes are further sensitised to HIV-induced apoptosis, once activated, and the intensity positively correlates to the degree of immune activation.36 SF-extract-induced activation of lymphocytes and, more specifically, of CD4+ cells, may be indicative of a detrimental effect of SF administration in HIV-infected patients.
CONCLUSION
Despite limited preliminary clinical evidence of the beneficial effects of SF extract in HIV management, it is evident that caution must be exercised in its use as a therapeutic agent. The toxic effects of SF extract, particularly in the immune system, need to be further investigated.
ACKNOWLEDGEMENTS
Miss V. Korb acknowledges a prestigious Masters scholarship from the National Research Foundation, South Africa.
REFERENCES
1. Van Wyk BE, Van Oudshoorn B, Gericke N. Medicinal plants of South Africa. Pretoria: Briza Publications; 1997. [ Links ]
2. Rood B. Uit die Veldapteek. Cape Town: Tafelberg Publishers; 1994. [ Links ]
3. Van Wyk BE, Gericke N. People's plants: A guide to useful plants of southern Africa. Pretoria: Briza Publications; 2000. [ Links ]
4. Chaffey N, Stokes T. In Brief: AIDS herbal therapy. Trends Plant Sci. 2002;7(11):57-59. [ Links ]
5. The Indigenous Knowledge Systems Division, MRC. A toxicity study of Sutherlandia leaf powder (Sutherandia microphylla) consumption. Final Report 2002 [document on the internet]. Medical Research Council and National Research Foundation of South Africa; April 2002 [cited 2009 July 19]. Available from: SA Health Info. [ Links ]
6. Fernandes AC, Cromarty AD, Albrecht C, van Rensburg CE. The antioxidant potential of Sutherlandia frutescens. J Ethnopharmacol. 2004;95(1):1-5. [ Links ]
7. Tai J, Cheung S, Chan E, Hasman D. In vitro culture studies of Sutherlandia frutescens on human tumor cell lines. J Ethnopharmacol. 2004;93(1):9-19. [ Links ]
8. Ostlund R, Sherman W, inventors; Washington University, assignee. Pinitol and derivatives thereof for the treatment of metabolic disorders. United States patent 5827896. 1998 Oct 27. [ Links ]
9. Mombereau C, Kaupmann K, Froestl W, Sansig G, van der Putten H, Cryan JF. Genetic and pharmacological evidence of a role for GABA(B) receptors in the modulation of anxiety- and antidepressant-like behavior. Neuropsychopharmacol. 2004; 29(6):1050-1062. [ Links ]
10. Allende CC, Allende JE. Purification and substrate specificity of arginyl-ribonucleic acid synthetase from rat liver. J Biol Chem. 1964;239(4):1102-1106. [ Links ]
11. Mitra SK, Mehler AH. The arginyl transfer ribonucleic acid synthetase of Escherichia coli. J Biol Chem. 1967;242(23):5490-5494. [ Links ]
12. Rosenthal GA, Dahlman DL. Studies of L-canavanine incorporation into insectan lysozyme. J Biol Chem. 1991;266(24):15684-15687. [ Links ]
13. Green M, inventor; The Regents of the University of California, assignee. Method of treating viral infections with amino acid analogs. United States patent 5110600. 1992 May 5. [ Links ]
14. Schnittman SM, Greenhouse JJ, Psallidopoulos MC, et al. Increasing viral burden in CD4+ T cells from patients with human immunodeficiency virus (HIV) infection reflects rapidly progressive immunosuppression and clinical disease. Ann Intern Med. 1990;113(6):438-443. [ Links ]
15. Goletti D, Weissman D, Jackson RW, et al. Effect of Mycobacterium tuberculosis on HIV replication: Role of immune activation. J Immunol. 1996;157(3):1271-1278. [ Links ]
16. Stanley SK, Ostrowski MA, Justement JS, et al. Effect of immunization with a common recall antigen on viral expression in patients infected with human immunodeficiency virus type 1. N Engl J Med. 1996;334(19):1222-1230. [ Links ]
17. Van Wyk BE, Albrecht C. A review of the taxonomy, ethnobotany, chemistry and pharmacology of Sutherlandia frutescens (Fabaceae). J Ethnopharmacol. 2008;119(3):620-629. [ Links ]
18. Cowan MM. Plant products as antimicrobial agents. Clin Microbiol Rev. 1999;12(4):564-582. [ Links ]
19. World Health Organisation. Traditional medicine: Growing needs and potential. In: WHO Policy Perspectives on Medicines. Geneva: World Health Organisation; 2002. p. 1-6. [ Links ]
20. De Smet PA. The role of plant-derived drugs and herbal medicines in healthcare. Drugs. 1997;54(6):801-840. [ Links ]
21. Margolis RL, Chuang DM, Post RM. Programmed cell death: Implications for neuropsychiatric disorders. Biol Psychiatry. 1994;35(12):946-956. [ Links ]
22. Schultz RM. Cell cycle, programmed cell death and cancer. In: Delvin TM, editor. Textbook of biochemistry with clinical correlations. 6th ed. United Kingdom: Wiley-Liss, 2006; p. 1013-1036. [ Links ]
23. Majno G, Joris I. Apoptosis, oncosis, and necrosis: An overview of cell death. Am J Pathol. 1995;146(1):3-15. [ Links ]
24. Olivetti G, Abbi R, Quaini F, et al. Apoptosis in the failing human heart. N Engl J Med. 1997;336(6):1131-1141. [ Links ]
25. Goodman Y, Bruce AJ, Cheng B, Mattson MP. Estrogens attenuate and corticosterone exacerbates excitotoxicity, oxidative injury, and amyloid beta-peptide toxicity in hippocampal neurons. J Neurochem. 1996;66(5):1836-1844. [ Links ]
26. Chinkwo KA. Sutherlandia frutescens extracts can induce apoptosis in cultured carcinoma cells. J Ethnopharmacol. 2005;98(1-2):163-170. [ Links ]
27. Vayssiere JL, Petit PX, Risler Y, Mignotte B. Commitment to apoptosis is associated with changes in mitochondrial biogenesis and activity in cell lines conditionally immortalized with simian virus 40. Proc Natl Acad Sci USA. 1994;91(24):11752-11756. [ Links ]
28. Bernardi P, Vassanelli S, Veronese P, Colonna R, Szabo I, Zoratti M. Modulation of the mitochondrial permeability transition pore: Effect of protons and divalent cations. J Biol Chem. 1992;267(5):2934-2939. [ Links ]
29. Petronilli V, Nicolli A, Costantini P, Colonna R, Bernardi P. Regulation of the permeability transition pore, a voltage-dependent mitochondrial channel inhibited by cyclosporin A. Biochim Biophys Acta. 1994;1187(2):255-259. [ Links ]
30. Harris MH, Thompson CB. The role of the Bcl-2 family in the regulation of outer mitochondrial membrane permeability. Cell Death Differ. 2000;7(12):1182-1191. [ Links ]
31. Mitchell P. Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. Biol Rev Camb Philos Soc. 1966;41(3):445-502. [ Links ]
32. Becker WM, Kleinsmith LJ, Hardin J. Chemotrophic energy metabolism: Aerobic respiration. In: The world of the cell. 5th ed. San Francisco: Pearson's Education, Inc. publishing as Benjamin Cummings, 2003; p. 398-444. [ Links ]
33. Cambiaggi C, Scupoli MT, Cestari T, et al. Constitutive expression of CD69 in interspecies T-cell hybrids and locus assignment to human chromosome 12. Immunogenetics. 1992;36(2):117-120. [ Links ]
34. Ziegler SF, Ramsdell F, Hjerrild KA, et al. Molecular characterization of the early activation antigen CD69: A type II membrane glycoprotein related to a family of natural killer cell activation antigens. Eur J Immunol. 1993;23(7):1643-1648. [ Links ]
35. Delves P, Martin S, Burton D, Roitt I. Roitt's Essential Immunology. 11th ed. Massachusetts: Blackwell Publishing Inc.; 2006. [ Links ]
36. Finkel TH, Tudor-Williams G, Banda NK, et al. Apoptosis occurs predominantly in bystander cells and not in productively infected cells of HIV- and SIV-infected lymph nodes. Nat Med. 1995;1(2):129-134. [ Links ]
 Correspondence to:
Correspondence to:
Anil Chuturgoon
Discipline of Medical Biochemistry, Nelson R Mandela School of Medicine
University of KwaZulu-Natal, Private Bag 7
Congella 4013, Durban, South Africa
email: chutur@ukzn.ac.za
Received: 16 Sept. 2009
Accepted: 08 Dec. 2009
Published: 11 Mar. 2010
This article is available at: http://www.sajs.co.za