Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Science
versión On-line ISSN 1996-7489
versión impresa ISSN 0038-2353
S. Afr. j. sci. vol.105 no.3-4 Pretoria mar./abr. 2009
RESEARCH ARTICLES
Pyrethroid resistance in a major African malaria vector Anopheles arabiensis from Mamfene, northern KwaZulu-Natal, South Africa
J.C. MouatchoI, II; G. MunhengaI, II, III; K. HargreavesIV; B.D. BrookeI, V; M. CoetzeeI, V, VI; L.L. KoekemoerI, V, *
IVector Control Reference Unit, National Institute for Communicable Diseases, NHLS, Private Bag X4, Sandringham 2131, South Africa
IISchool of Animal, Plant and Environmental Sciences, University of the Witwatersrand, Private Bag 3, WITS 2050, South Africa
IIIDepartment of Biological Sciences, University of Zimbabwe, P.O. Box MP167, Mount Pleasant, Harare, Zimbabwe
IVMalaria Control Programme, KwaZulu-Natal Department of Health, Jozini, South Africa
VDivision of Virology and Communicable Diseases Surveillance, School of Pathology of the National Health Laboratory Service and the University of the Witwatersrand, Johannesburg, South Africa
VINRF Research Chair in Medical Entomology and Vector Control, School of Pathology, University of the Witwatersrand, Johannesburg, South Africa
ABSTRACT
A population of Anopheles arabiensis, a major malaria vector in South Africa, was collected during 2005 from inside sprayed houses in Mamfene, northern KwaZulu-Natal, South Africa, using window exit traps. None of these specimens (n = 300 females) was found to be infected with Plasmodium falciparum. Insecticide susceptibility assays on 2–3 day old F1 progeny using WHO susceptibility kits revealed 100% susceptibility to bendiocarb, resistance to deltamethrin (95.91%) was suspected, while resistance to permethrin (78.05%) was confirmed. The knockdown resistant (kdr) genotype was not found in the surviving mosquitoes. Biochemical analysis using enzyme assays showed elevated levels of monooxygenase that correlated with the permethrin bioassay data. While elevated levels of non-specific esterase were found in some families (11/12 for α- and 6/12 for β-esterases), the data did not show any correlation with the permethrin bioassay. Analysis of permethrin and bendiocarb tolerant lines, selected in the laboratory to characterise biochemical resistance profiles, showed increased levels of non-specific esterase and monooxygenase activity in the case of the permethrin-selected cohorts, and elevated glutathione S-transferases and general esterases in that of the bendiocarb-selected line. Synergist assays, using piperonyl butoxide, confirmed the involvement of monooxygenase and glutathione S-transferase in pyrethroid and bendiocarb resistance. This study underlines the importance of routine surveillance for insecticide susceptibility in wild anopheline populations.
Key words: Anopheles arabiensis, pyrethroid resistance, metabolic resistance, KwaZulu-Natal, South Africa
Introduction
Malaria vector control in southern Africa relies mainly on indoor residual house spraying (IRS) with insecticides. Synthetic pyrethroids and DDT (dichloro-diphenyl-trichloroethane) are used in IRS applications, while permethrin-treated bed-nets are used for personal protection in some households. DDT was introduced for malaria control in 1946 in South Africa and remained in use until 1996 when the choice of insecticide was changed to the pyrethroid deltamethrin.1 Following a major upsurge of malaria cases in 1999/2000 and the detection of pyrethroid resistance in the major malaria vector Anopheles funestus,2 DDT was re-introduced in 2001 and a marked reduction in malaria morbidity and mortality was recorded.3,4 Deltamethrin is currently used in modern-style houses with painted walls while DDT is used in traditional houses.
Anopheles arabiensis is a major malaria vector in southern Africa, second only to Anopheles funestus. Historically, it has been considered to be susceptible to all insecticides.5 Standard bioassay susceptibility studies in three provinces of South Africa (KwaZulu-Natal (KZN), Limpopo and Mpumalanga) in 1996 showed full susceptibility to DDT, deltamethrin and fenitrothion in all provinces, while resistance to propoxur was reported in Limpopo province.6 Biochemical analyses on these samples provided little information on the mechanism causing the propoxur resistance. The first record of DDT resistance in An. arabiensis from South Africa was reported in 2003 in KZN.3 Bioassay data from this study also showed reduced susceptibility to deltamethrin. Biochemical analyses on these samples showed increased levels of non-specific esterases (EST) and glutathione S-transferases (GST), enzymes that are commonly involved in metabolic resistance. However, there was a lack of correlation between elevated enzyme levels and the bioassay data.
This study presents the first report of permethrin resistance in An. arabiensis in South Africa.
Materials and methods
Field material
Mosquitoes were collected from Mamfene, northern KwaZulu-Natal (KZN), South Africa (27°23'S, 32°12'E) between February and September 2005, using window exit traps in permethrin-sprayed houses.7 Mosquitoes identified morphologically as members of the An. gambiae complex8 were transported to the Vector Control Reference Unit (VCRU), National Institute for Communicable Diseases, Johannesburg, South Africa, for further investigation. Live females were induced to lay eggs under standard insectary conditions (25°C, 75–80% relative humidity and 12 h light:dark with dusk/dawn transition lighting) and each egg batch was treated separately as a 'family' cohort.
Larvae from each family were reared through to adults, and the wild parental specimens preserved on silica gel for further analysis using a polymerase chain reaction (PCR)9 and Plasmodium falciparum sporozoite detection using an enzyme-linked immunosorbent assay (ELISA).10 Samples of F1 progeny (1–4 days old) from each family were subjected to standard WHO insecticide susceptibility tests.11 Additional samples of unexposed males and females (1–4 days old) from various families were frozen at –70°C for biochemical analysis.12 The remaining progeny were used to analyse the pyrethroid-resistant mechanism involved.
Plasmodium falciparum analysis
The heads and thoraces of wild female mosquitoes were separated from the rest of the body and tested for the presence of malaria parasites using ELISA.10 Negative controls consisted of unfed specimens of An. arabiensis, while synthetic peptide standardised against P. falciparum was included on each plate as a positive control. Results were analysed using a microtitre plate reader at a wavelength of 405 nm.
Insecticide susceptibility tests
Susceptibility tests were done using WHO insecticide-impregnated papers and test kits. Three-day-old F1 adults (n = 20–25) were exposed to deltamethrin (0.05%), bendiocarb (0.1%) and permethrin (0.75%). A laboratory colony of An. arabiensis (KGB), known to be susceptible to all insecticides, was used as a control to ensure the reliability of impregnated papers. Negative controls consisted of mosquitoes exposed to untreated papers. Knockdown was recorded after 1 h and a 10% sucrose solution was made available to survivors. Final mortality was recorded 24 h post-exposure as defined by WHO criteria for determining susceptibility/resistance to diagnostic insecticide concentrations.11
Knockdown resistance (kdr) mutation
Polymerase chain reaction genotyping to detect the West African Leu-Phe kdr allele was performed on samples of An. arabiensis resistant to permethrin.13 Briefly, DNA was extracted from individual mosquitoes and re-suspended in TE buffer14 as detailed in the PCR conditions previously described.15
Selection for resistance
Permethrin selected line: Anopheles arabiensis progeny not used for laboratory analysis were pooled and used to colonise a strain named Kwag. This new colony was allowed to stabilise for a year before selection experiments were initiated. Newly emerged male and female adults were separated to ensure that mating did not take place prior to insecticide exposure. Adults were placed in cages with a 10% sugar solution. Two-day-old adults were exposed to 0.75% permethrin for 20 min according to the standard WHO bioassay procedure.11 Knockdown after 20 min and final mortality 24 h post-exposure were recorded. Surviving males and females were placed into a cage and left for four days to mate. Females were offered blood meals three times per week. Eggs from each generation were reared through to adults and these were subjected to subsequent selection pressure, using 0.75% permethrin, for each generation. Exposure time was increased to 1 h once survival above 50% was achieved. Sub-samples from the third, fifth and seventh generations were stored at –70°C for biochemical analysis. The pyrethroid selected line was named Kwag-Perm. Observed differences in susceptibility between the baseline and selected generations were analysed by comparing LT50 values calculated from time– mortality regression analysis.
Carbamate selected line: Samples of wild An. arabiensis were collected from Mamfene during a field study in 2003.3 A colony was established during this time and was named MBN. This strain had low levels of carbamate resistance and selections against 0.1% bendiocarb were initiated as described above. The selected line was named MBN-Carb.
Synergist assays
Synergist assays were performed using piperonyl butoxide (PBO), an inhibitor of monooxygenase, diethyl maleate (DEM), an inhibitor of GSTs, and triphenyl phosphate (TPP), an inhibitor of esterases, to synergise the bendiocarb and permethrin resistance phenotypes in laboratory An. arabiensis from KZN.16–18 Three cohorts (groups) of adults (n = 75) were used from each resistant strain. One group was exposed to 4% PBO, 8% DEM or TPP (1% and 6%) for 1 h, and then immediately exposed to either 0.75% permethrin for 1 h or 0.1% bendiocarb for 40 min. Beyond 40 min, selected An. arabiensis were still fully susceptible to 0.1% bendiocarb. The second cohort/group was exposed to either permethrin or bendiocarb only, while the third cohort was exposed to PBO, or DEM or TPP. Following an average of five replicates, final mortality 24 h post-exposure was compared between synergised and unsynergised samples using ANOVA.
Biochemical analysis
Assays designed to quantify relative levels of monooxygenase, non-specific esterases and GST activities in individual mosquitoes were performed.12 In addition, the presence in individual mosquitoes of an altered acetylcholinesterase associated with carbamate/organophosphate resistance was assayed.12 Propoxur was used as a reference carbamate insecticide for the acetylcholinesterase assay. Mixed samples of female F1 progeny from familial material initially stored at –70°C were assayed concurrently with female samples taken from the KGB-susceptible laboratory strain of An. arabiensis. Similar assays were used to quantify exposure levels/activities in the selected lines compared with their respective baselines (Kwag and MBN). Statistical analysis was conducted using a two-sample Student's t-test assuming equal variances.
Results
Field-collected samples
A total of 461 mosquitoes were collected and morphologically identified as members of the An. gambiae complex.8 These were further identified to species level9 as 425 An. arabiensis, six An. merus and six An. quadriannulatus. The remaining 24 specimens could not be identified despite three attempts at amplification. This may have been the result of poor DNA preservation and subsequent DNA degradation. Wild-caught females (n = 300) were tested for the presence of P. falciparum circumsporozoites. None were found to be infected.
A minimum of 10 and a maximum of 45 mosquitoes per family were exposed to each of three insecticides. The mortality rates were 95.91% and 100% against deltamethrin and bendiocarb, respectively. Exposure to permethrin, however, showed evidence of resistance in 12/22 families (54%), with an average final mortality of 78.06% across all families (Table 1). Mortalities of control samples (KGB) were 100% for all three insecticides. All specimens from the resistant families genotyped for the kdr allele showed the susceptible genotype (data not shown).
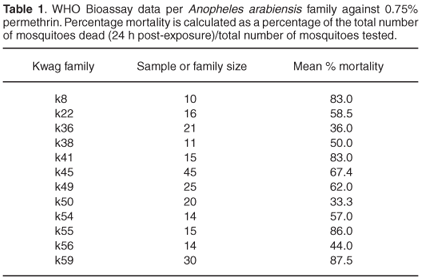
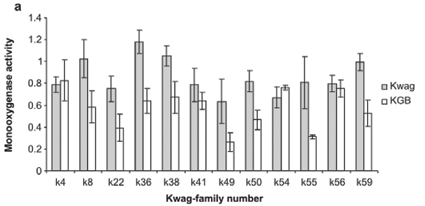
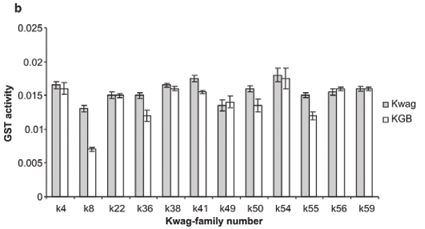
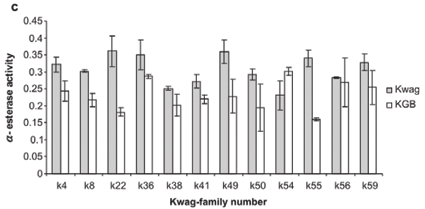
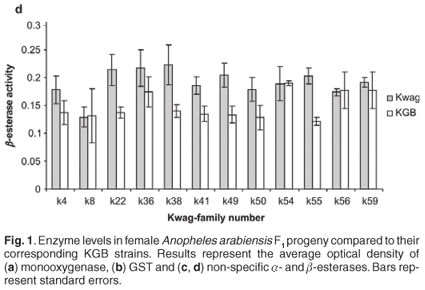
Average monooxygenase levels in An. arabiensis F1 female progeny were significantly higher in 8/12 permethrin-resistant families compared with the standard susceptible strain KGB (P < 0.05) (Fig. 1A). Those families (k8, k22, k36, k38, k49, k50, k55, k59) with high levels of monooxygenase showed a significant correlation with permethrin bioassay mortality data (r > 0.9, P < 0.05). The mean levels of GST activity were not significantly higher than those of the control samples (P = 0.14, Fig. 1B). There was no correlation between the mean levels of GST activity and the bioassay results (r < 0.5, P > 0.05). The levels of non-specific esterases using α- and β-naphthyl acetate as substrates were significantly higher than those of the control (P < 0.05, Figs 1C and 1D). No correlations were observed between esterase levels and bioassays across families. None of the resistant family showed significant (r < 0.5, P > 0.05) inhibition of acetylcholinesterase when challenged with propoxur.
Selection for resistance
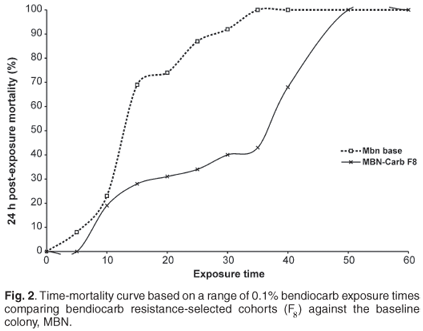
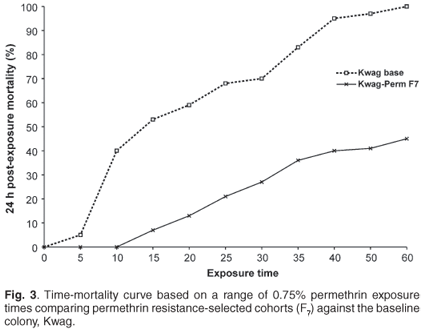
The permethrin selection process led to a rapid increase in resistance over successive generations. Mortalities in permethrin-resistant lines decreased from 70% in F0 to 27% in the F7 cohort following an exposure period of 30 min. Susceptibility to bendiocarb steadily decreased over successive generations, and mortality ranged from 71% in F0 to 32% in the F8 cohorts following exposure to bendiocarb for 20 min. Exposure of F8 cohorts to the WHO discriminating dose with bendiocarb for 60 min resulted in 100% mortality, indicating no resistance according to the WHO definition.
Based on time mortality curves (Figs 2 and 3), there were significant differences (P < 0.05) in the susceptibility levels between the selected and non-selected cohorts. The predicted exposure time causing 50% mortality (LT50) increased from 20 min to 58 min for the permethrin-selected cohorts, while the LT50 for the carbamate-selected line increased from 16 to 30 min.
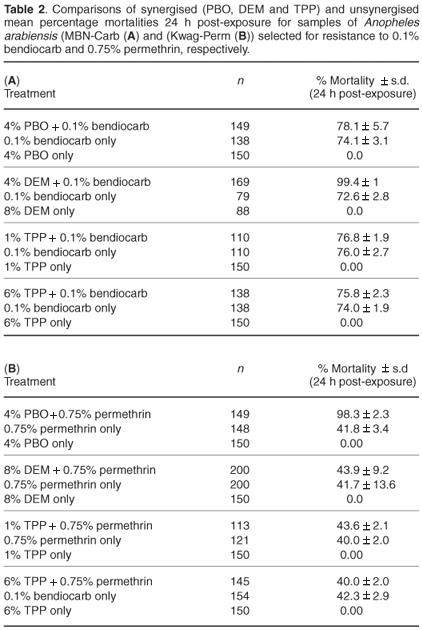
Synergist assays
Table 2 details the synergist results on carbamate- and pyrethroid-selected lines. The differences in mortality 24 h post- exposure between synergised and unsynergised samples were statistically significant for both the bendiocarb-selected cohorts (P < 0.05 using 8% DEM) and the permethrin-selected cohorts (P < 0.05 using 4% PBO). There were no significant differences between synergised and unsynergised samples for both selected lines using 1% TPP (P > 0.05 for Kwag-Perm and P > 0.05 for MBN-Carb) and 6% TPP (P > 0.05 for Kwag-Perm and P > 0.05 for MBN-Carb).
Biochemical analysis
Table 3 summarises the results of biochemical assays on the carbamate-selected line (MBN-Carb); there was no significant increase in monooxygenase levels. Levels of non-specific esterases using α- and β-naphthyl acetate as substrates showed a highly significant difference between the selected and non-selected cohorts (P < 0.05 assuming unequal variance). The mean level of GST activity was significantly elevated in the selected cohort (P < 0.05). Altered acetylcholinesterase assays gave no suggestion of reduced propoxur inhibition in the F8 generation, with a mean inhibition of 88% compared to the baseline samples which showed mean inhibition of 92% (P > 0.05).
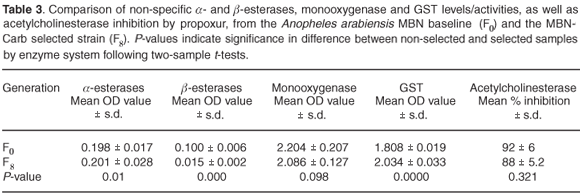
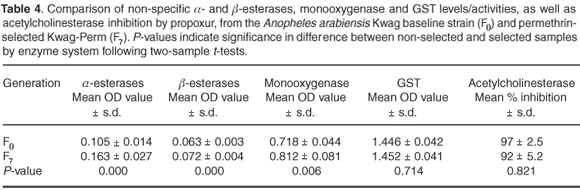
Table 4 shows metabolic enzyme levels of Kwag baseline samples compared to a sample of the F7 generation of the permethrin-selected strain. A significantly higher level of monooxygenase was detected in the selected (F7) strain compared with the non-selected parent colony (F0) (P < 0.05). Levels of non-specific esterases using both α- and β-naphthyl-acetate as substrates showed significant differences between the selected and non-selected cohorts (P < 0.05). Mean GST activity showed no statistically-significant differences in expression between the baseline cohorts and those selected for permethrin resistance (P > 0.05). Inhibition of acetylcholinesterase when challenged with propoxur was not significantly different between the selected and non-selected strains (P > 0.05).
Discussion
The mosquito collections conducted for this study during 2005 revealed an abundance of An. arabiensis in Mamfene, KwaZulu-Natal. Although specimens were collected inside exit window traps, none of the An. arabiensis specimens was infected with P. falciparum. This was not surprising as malaria transmission in the area was very low during this period (Feb–May, average 130 cases per month, Aug–Sept, average 50 cases). The low index of malaria transmission was attributed to decreased sporozoite prevalence as a result of indoor residual spraying and drug interventions.3
Anopheles arabiensis showed reduced susceptibility to deltamethrin based on the WHO11 recommendation which states that a 98–100% mortality rate is indicative of susceptibility, one of 80–98% requires further investigation, and one of <80% confirms resistance. While these results were similar to those obtained in the same area in 2003,3 the new results showed resistance to permethrin at levels varying between 33–83%. Pyrethroid toxicity is highly dependent on stereochemical structure.19 Permethrin and deltamethrin are Class I and Class II pyrethroid insecticides, respectively, which could explain the variation observed in susceptibility.
Increased levels of monooxygenase were correlated with resistance to permethrin across families reared from wild-caught material. The selected line showed a rapid increase in survival from the F0 to the F7 generation, illustrating the potential rate of rapid resistance development in the wild parent population. Currently, field populations may not be under stringent pyrethroid-selection pressure, which may explain the lag in the development of resistance in these circumstances. The Kwag-Perm colony also showed elevated monooxygenase levels confirmed by synergist assays using PBO, a synergist that specifically inhibits monooxygenase activity. These data strongly suggest that pyrethroid resistance in An. arabiensis from South Africa is mediated by monooxygenase detoxification, because synergism with PBO was complete.
Synergism with DEM and TPP, agents that specifically inhibit GST and esterase activity respectively, had no effect on the pyrethroid-resistance phenotype, suggesting that GSTs and the elevated esterases observed played no role in pyrethroid resistance.
Although no carbamate resistance was observed in the field populations, the bendiocarb-selected line provided insight into the possibility of An. arabiensis developing bendiocarb resistance with the extensive use of this insecticide. Selection for carbamate resistance was unsuccessful, indicating that this population did not possess carbamate-resistance genes. The MBN-Carb strain showed significant increases in GST and non-specific esterases, and the involvement of GST in bendiocarb tolerance was supported by synergist studies using DEM. Although there was an up-regulation of non-specific esterases, their involvement in tolerance was discounted using TPP, an inhibitor of general esterases.
The data presented in this paper were collected during the 2005 malaria season. An additional survey was carried out in the same area at the beginning of 2008 to ascertain if pyrethroid resistance was still present; the results showed 84% mortality with permethrin. This resistance, however, had still not resulted in an increase in malaria cases in the area (Department of Health, unpubl. data). This study underlines the importance of routine surveillance of insecticide susceptibility in wild anopheline populations. Insecticide resistance can develop rapidly, but is not necessarily associated with control programme failure and an increase in malaria transmission. Understanding these mechanisms allows for better resistance management by vector control managers.
We thank Prof. R.A. Wirtz (CDC Entomology Branch, Atlanta, GA) for supplying P. falciparum-positive controls and monoclonal antibody P. falciparum 2A10 for indirect ELISAs used in this study. This investigation received financial support from the Multilateral Initiative on Malaria (MIM) project A40036 through the UNICEF/UNDP/World Bank/WHO Special Programme for Research and Training in Tropical Diseases (TDR) to L.L.K. This work was also partially funded by grants from the South African Medical Research Council and the National Health Laboratory Service Research Trust to L.L.K.
1. Hargreaves K., Hunt R.H., Brooke B.D., Mthembu J., Weeto M.M., Awolola T.S. and Coetzee M. (2003). Anopheles arabiensis and An. quadriannulatus resistance to DDT in South Africa. Med. Vet. Entomol. 17, 417–422. [ Links ]
2. Hargreaves K., Koekemoer L.L., Hunt R.H., Brooke B.D., Mthembu J. and Coetzee M. (2000). Anopheles funestus resistant to pyrethroid insecticides in South Africa. Med. Vet. Entomol. 14, 181–189. [ Links ]
3. Maharaj R., Mthembu D.J. and Sharp B.L. (2005). Impact of DDT re-introduction on malaria transmission in KwaZulu-Natal. S. Afr. Med. J. 95, 871–874. [ Links ]
4. Coetzee M. (2006). Malaria and dengue vector biology and control in southern and eastern Africa. In Bridging Laboratory and Field Research for Genetic Control of Disease Vectors, eds B.G.J. Knols and C. Louis, ch. 9, vol. 11, pp. 101–109. Wageningen Ur Frontis Series, Springer, Dordrecht. [ Links ]
5. Coetzee M., Horne D.W.K., Brooke B.D. and Hunt R.H. (1999). Insecticide resistance in the African malaria vector mosquitoes: historical review and the implications for future malaria control in the southern African sub-region. S. Afr. J. Sci. 45, 215–218. [ Links ]
6. Gericke A., Govere J.M. and Durrheim D.N. (2002). Insecticide susceptibility in the South African malaria mosquito Anopheles arabiensis (Diptera: Culicidae). S. Afr. J. Sci. 98, 205–208. [ Links ]
7. Muirhead-Thomson R.C. (1947). Studies on Anopheles gambiae and A. melas in and around Lagos. Bull. Entomol. Res. 38, 527–558. [ Links ]
8. Gillies M.T. and Coetzee M. (1987). A supplement to the Anophelinae of Africa South of the Sahara. S. Afr. Inst. Med. Res. Publication no. 55. [ Links ]
9. Scott J.A., Brogdon W.G. and Collins F.H. (1993). Identification of single specimens of Anopheles gambiae complex by the polymerase chain reaction. Am. J. Trop. Med. Hyg. 49, 520–529. [ Links ]
10. Wirtz R.A., Zavala F., Charoenvit Y., Campbell G.H., Burkot T.R., Schneider I., Esser K. M., Beaudoin R.L. and André R.G. (1987). Comparative testing of Plasmodium falciparum sporozoite monoclonal antibodies for ELISA development. Bull. WHO. 65, 39–45. [ Links ]
11. WHO (1998). Test Procedures for Insecticide Resistance Monitoring in Malaria Vectors, Bio-efficacy and Persistence of Insecticides on Treated Surfaces. Document WHO/CDS/CPC/MAL/98. 12. World Health Organization, Geneva. [ Links ]
12. Penilla R.P., Rodríguez A., Hemingway J., Torres J.L., Arredondo-Jiménez J.I. and Rodríguez M.H. (1998). Resistance management strategies in malaria vector mosquito control. Baseline data for a large-scale field trial against Anopheles albimanus in Mexico. Med. Vet. Entomol. 12, 217–233. [ Links ]
13. Martinez-Torres D., Chandre F., Williamson M.S., Darriet F., Berge J.B., Devonshire A.L., Guillet P., Pasteur N. and Pauron D. (1998). Molecular characterization of pyrethroid knockdown resistance (kdr) in the major vector Anopheles gambiae s.s. Insect Mol. Biol. 7, 179–184. [ Links ]
14. Collins F.H., Mendez A.M., Rasmussen M.O., Mehaffey C.F., Besansky N.J. and Finnerty V. (1987). A ribosomal RNA gene probe differentiates member species of the Anopheles gambiae complex. Am. J. Trop. Med. Hyg. 37, 37–41. [ Links ]
15. Matambo T.S., Abdalla H., Brooke B.D., Koekemoer L.L., Mnzava A., Hunt R.H. and Coetzee M. (2007). Insecticide resistance in the malaria mosquito Anopheles arabiensis and association with the kdr mutation. Med. Vet. Entomol. 21, 97–102. [ Links ]
16. Brogdon W.G. and Barber A.M. (1990). Microplate assay of glutathione s-transferase activity for resistance detection in single-mosquito triturates. Comp. Biochem. Physiol. 96B, 339–342. [ Links ]
17. Brogdon W.G., McAllister J.C. and Vulule J. (1997). Heme peroxidase activity measured in single mosquitoes identifies individuals expressing an elevated oxidase for insecticide resistance. J. Am. Mosq. Control Assoc. 13, 233–237. [ Links ]
18. Chareonviriyaphap T., Rongnoparut P., Chantarumporn P. and Bangs M.J. (2003). Biochemical detection of pyrethroid resistance mechanisms in Anopheles minimus in Thailand. J. Vector Control 28, 108–116. [ Links ]
19. Milam C.D., Farris J.L. and Wilhide J.D. (2000). Evaluating mosquito control pesticides for effect on target and non-target organisms. Arch. Environ. Contam. Toxicol. 39, 324–328. [ Links ]
Received 20 October 2008. Accepted 17 March 2009.
* Author for correspondence E-mail: lizettek@nicd.ac.za