Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.105 n.1-2 Pretoria Jan./Feb. 2009
REVIEW ARTICLES
When to stay, when to go: trade-offs for southern African arid-zone birds in times of drought
W.R.J. DeanI, *; P. BarnardI, II; M.D. AndersonIII
IDST/NRF Centre of Excellence at the Percy FitzPatrick Institute of African Ornithology, University of Cape Town, Rondebosch, 7701 South Africa
IIBirds and Environmental Change Partnership, Climate Change and Bio-Adaptation Division, South African National Biodiversity Institute, Private Bag X7, Claremont, South Africa
IIIBirdLife South Africa, P.O. Box 515, Randburg 2125, Gauteng, South Africa
ABSTRACT
Arid environments remind one of the punctuated equilibrium theory of evolution: they experience long periods of stasis and low productivity, interrupted with episodic rainfall which spurs reproduction and movement. Birds, as highly dispersive organisms, are among the most dramatic indicators of these fluctuations. Here we review birds' two main strategies, residency and nomadism, and the trade-offs faced by individuals in uncertain times. In general, wet years stimulate higher densities of nests (i.e. smaller territories), larger clutch sizes, unseasonal breeding, and at some times of year, higher breeding success. Rainfall above a certain threshold triggers breeding in resident species and an influx of nomadic species which breed and then move on. The environmental cues which trigger nomadism are sometimes poorly understood, but include distant thunderstorms for aquatic species, and perhaps for insectivores. Environmental cues that draw nomadic granivores to areas that have had recent rain are not known.
Key words: avian-nomadism, arid environments, Karoo-Namib Desert, Kalahari, breeding
Introduction
Prolonged spells of drought pose dilemmas for most organisms, even those adapted to hyper-aridity. For birds, reproduction, feeding, dispersal and moult are all activities which may need careful timing in relation to fluctuations in ecological productivity. In arid and semi-arid ecosystems, rainfall often triggers events in the life cycle which may be suppressed for months or even years during dry periods.1,2,3,4 In times of very low productivity or harsh conditions, birds, like many other animals, can move to escape local conditions and improve their chances of feeding or reproduction.5 However, some resident species may not move, but simply adjust their activities to 'ride out' difficult periods.
Semi-arid ecosystems are characterised by wet or dry states that are patchy in time and space. This patchiness is particularly true of southern Africa, where much of the region is semi-arid6 and the environment experiences extremes in weather, from periods of intense and prolonged drought to exceptionally high rainfall events.7 Rainfall in arid and semi-arid ecosystems in the southern hemisphere, including southern Africa, is greatly affected by El Niño Southern Oscillations (ENSO), leading to large variability in rainfall and prolonged droughts.6 Similarly, extensive wet periods (La Niña events) have concomitant effects on ecosystem functioning.8 The effects of El Niño and La Niña have been well studied in a few organisms, particularly birds, where changes in species richness and density9 and reproduction10 are correlated with the wet–dry cycles of ENSO years.
Birds, other animals and plants respond to increased rainfall in similar ways, and both birds and plants tend to increase reproductive effort with more rain.11–14 The response by biota to extended dry periods in semi-arid environments, however, differs markedly between and within invertebrate and vertebrate phyla, and markedly between most animals and plants. Plants cope with droughts by dying after depositing dormant propagules (ephemerals) or becoming dormant and restricting their internal water use by discarding leaves or leaves and stems.13,15 Although death and propagule dormancy are options taken by plants and some invertebrate taxa, viz. brine shrimp (Artemia), these options are not available to vertebrates, leaving escape and water conservation as the alternatives.5,16
Drought-induced dormancy in vegetation therefore has effects on animals. Animals in general, and birds in particular, cope with droughts and changes in vegetation by using behavioural and physiological tactics, including opportunistic movement away (in birds), shifts in habitat5 and deferred hatching or dormancy in eggs (in locusts).2,3,4 It is not known in detail whether birds make dietary shifts during extended droughts, but many species are opportunistic in their foraging and feeding in the Karoo17 and it is very likely that such shifts do occur.
Our questions in this review are: (1) what is the influence of variability in rainfall on avian populations, including breeding and movements? and (2) how resilient are bird populations to extended dry periods? While we do not explicitly review climate change per se, our conclusions should be of value in predicting species-specific responses and levels of vulnerability to changing rainfall patterns in Africa.
The influence of rainfall on population dynamics
Exceptionally high rainfall may stimulate rare breeding events. For example, banded stilts (Cladorhynchus leucocephalus) in arid Australia arrive and breed in very large colonies following exceptionally high rainfall at ephemeral pans that may have been dry for decades.1,18 Variability in rainfall has strong effects on clutch sizes and population dynamics in birds, as observed in unrelated species in different arid parts of the world, such as Galapagos ground finches on the Galápagos Islands,10 galahs (Cacatua roseicapilla) in Australia19 and larks,20 thrushes and helmet-shrikes in arid southern Africa.12,21 Droughts have different effects on bird populations, often stimulating movements into better-watered areas, but also, conversely and seemingly inexplicably, stimulating movements into dry areas.22 Crowned hornbills (Tockus alboterminatus) wander from their forest habitat in the non-breeding season,23 some years reaching far into the semi-arid Karoo.24,25 These movements are thought to be related to unusual aridity or cold in their coastal habitats,23 although these are probably always more mesic than the areas to which the birds disperse.
The movements of nomadic birds in arid and semi-arid ecosystems throughout the world have been reviewed by Dean.5 We know that some birds have evolved to cope with stochastic weather events in their habitat. What is less clear, however, is what resident bird species do when faced with increasing aridity in their environment. For those species that move, the benefits must outweigh the costs.26 Similarly, for those species that are resident, the benefits of being resident must outweigh the cost of moving. For example, in partially-migratory rock kestrels (Falco rupicolus), individual males used one or the other of these two tactics.27 Males departed for non-breeding areas later than did females, and returned to their territories earlier than did females. Not all males migrated, and those that remained successfully retained their territories and had a higher probability of breeding in the next breeding season. However, there was a cost: males that stayed on their territories during the winter faced increased competition for food. Males that migrated probably had higher survival, but risked losing their territories. Male rock kestrels in the Karoo thus faced a trade-off between increased chances of breeding and the risk of mortality.27
Trade-offs are necessary where rainfall is highly unpredictable. Unlike the short breeding season in the temperate northern hemisphere, the concept of extended 'equally good months' for breeding (EGMs)28 often applies to bird populations in drier parts of the southern hemisphere, where weather patterns are less predictable and suitable or unsuitable conditions for breeding may arise at any time of the year. EGMs are calculated as the months in which there are breeding records equal to or above the average number of breeding records per month for the species. Africa and Australia both have a mean EGM 1.9 months a year longer than anywhere else,29 presumably caused by out-of-season rainfall, or rainfall in areas that have been dry for a long period. Even within South Africa, areas with higher coefficients of variation in total annual rainfall,30 such as the Karoo, have more EGMs than areas with more sharply seasonal, but more predictably timed, rainfall such as the southwestern Cape (Table 1).

The most frequently measured responses by birds in arid regions to above average rainfall or isolated rainfall events are breeding out of season, breeding in large numbers, and increases in clutch size or number of young produced. For example, 'heavy rains' following a five-year drought in the western Karoo stimulated bird breeding activity, with a number of species recorded with eggs and young, and all collected specimens in breeding condition31. Similarly, 170 mm of rain between January and March 1985, following a severe drought in the central Karoo, was believed to have stimulated breeding in 27 species of birds32. However, no studies of breeding activity in either case were made during the 'severe drought' broken by the rains, so the level of increase in breeding activity was difficult to evaluate.
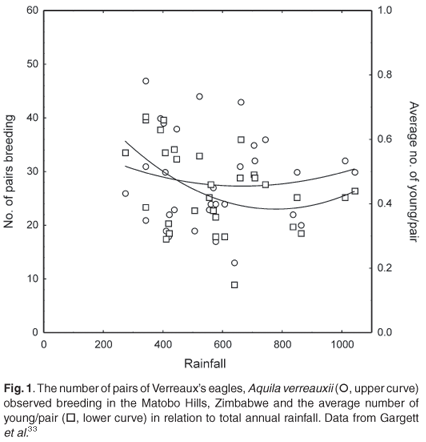
Better data on the influence of rainfall on population dynamics are provided by long-term studies. Verreaux's eagles (Aquila verreauxii) breeding in the Matobo Hills, Zimbabwe showed responses to variations in annual rainfall amounts. The number of resident pairs of Verreaux's eagles increased during the high rainfall years and correlated with increases in the number of hyraxes (Procavia capensis and Heterohyrax brucei)33. Both the number of pairs in the local population that bred and their production of young fit a negative exponential curve (Fig. 1), suggesting that the eagles benefit from both lower and higher rainfall years, but not from average rainfall years. Lower rainfall years have as many pairs breeding as higher rainfall years, with relatively higher breeding success. This suggests that breeding success is not only correlated with rainfall, but that other factors also influence the population dynamics of this species at this locality. Average rainfall years may increase competition for hyraxes, or higher rainfall years may lead to increased chick mortality.
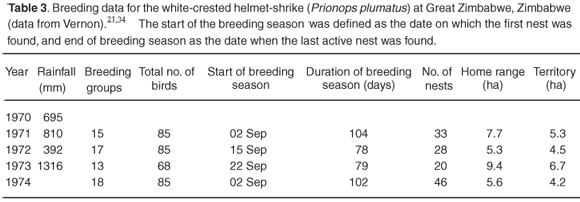
In deciduous woodland habitats that come into leaf before the rains, there is some evidence that the level of breeding activity in birds is correlated with rainfall during the previous year. A study in the Miombo woodland in Zimbabwe21 showed that rainfall during both the previous breeding season and current breeding season influences the number of nests and clutch sizes in Kurrichane thrushes (Turdus libonyanus) and white-crested helmet-shrikes (Prionops plumatus) (Tables 2 and 3). Both are resident species, generally maintaining territories all year round. The difference in breeding effort between years was not very marked in the Kurrichane thrush following the dry year of 1972, but breeding effort following the wettest year (1973) increased markedly, with an earlier start to breeding, increases in nest density per hectare (Table 2) and increases in the number of breeding attempts.21,34 With lower rainfall in the previous season there were decreases in the number of breeding groups and total number of birds, and increases in home range and territory sizes in white-crested helmet-shrikes (Table 3).21

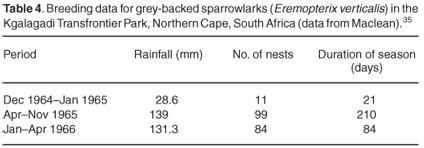
In a more arid environment, Maclean35 showed that resident and nomadic larks (Alaudidae) and resident sociable weavers (Philetarius socius) in the Kgalagadi Transfrontier Park responded quickly to rain, and that the duration of the breeding 'season' varied according to the amount of rain. Table 4 gives data for one species, the grey-backed sparrowlark (Erempoterix verticalis) and Table 5 presents data for the sociable weaver. The average annual rainfall for this area is about 180 mm per year so the amount of rain that fell in April–November 1965 and January–April 1966 was more than 70% of the average annual rainfall. The relationship between rainfall, breeding season and breeding success, however, is not linear. Maclean36 suggested that the duration of the breeding season is not directly governed by the amount of rain, but rather by the time of year in which rain falls.

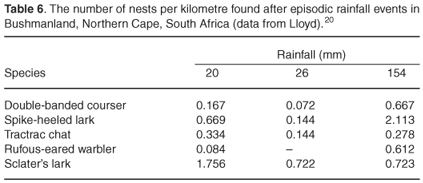
This suggestion is supported by Lloyd20 who noted that the timing and length of the breeding season in Bushmanland, Northern Cape Province depended on the integrated effect of rainfall and temperature on the growing season of the vegetation. Higher breeding success, measured as the percentage of young that left the nest, apparently depends on time of year and not the amount of rain per se (Table 5). The breeding success of sociable weavers was markedly lower during the hot summer months of January to April than the cooler, longer period from April to November36 (Table 5).
Clutch size may vary with rainfall and aridity. In sociable weavers35 there was some variation in mean clutch size per month, but specific rainfall data for those months were unavailable. However, a study of a bird community in Bushmanland, Northern Cape, showed that breeding activity of five species increased markedly after rainfall (Table 6), and more than half the species showed an increase in average clutch size compared to drier conditions.20 Two species, black-eared sparrowlarks (Eremopterix australis) and grey-backed sparrowlarks, showed a rapid response to rain and began laying larger clutches within seven days of 78 mm of rain.20
The influence of drought on bird populations
Movements in birds may be in response to rain or in response to drought. In general, drought, or extended dry periods, has the effect of reducing bird diversity and reducing the number of individuals,37 either through emigration5 or lowered survival.38 We do not know how resilient bird populations are to drought in southern Africa, because there are no intensive long-term studies of birds that have covered a full cycle of dry and wet years. There are, however, studies showing that resident birds may be quite resistant to drought. A long-term study at Tierberg, near Prince Albert in the Karoo, showed that the number of individuals of resident long-billed larks (Certhilauda subcoronata), spike-heeled larks (Chersomanes albofasciata), Karoo chats (Cercomela schlegelii) and rufous-eared warblers (Malcorus pectoralis) showed relatively small annual fluctuations regardless of rainfall39 (Table 7). This data support the idea that in some cases the benefits of remaining in an area outweigh the costs of moving.
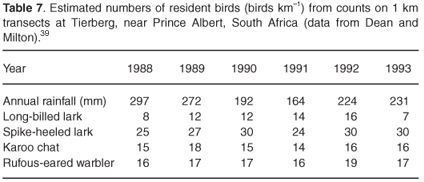
However, in the Kalahari, Botswana, resident species may show less resistance to drought, suggesting that resistance may depend to some extent on more subtle factors. For example, dry-season bird diversity, and number of individuals, were high following a higher rainfall wet season, and markedly lower following a poor rainfall season.37 Of 39 resident and nomadic species, populations of three resident species were stable and nine resident and nomadic species increased in abundance during the wet year following a dry year. But 20 species, including some nomads, decreased in abundance during that year. In the dry year following a wet year, all species had decreased markedly in number, and 12 species were no longer locally present. All nomadic species showed large increases in number in the wet year. Only one of the nomads, Temminck's courser (Cursorius temminckii), was present during the second dry year. There was no general pattern apparent in the residents or nomads, except that bird numbers were generally lower in the wet season following a dry season than in the dry season following a wet season. Both groups of birds showed fluctuations in number that cannot be entirely explained by rainfall amount. Some residents (but not all) showed some initial resilience to the dry conditions. Such resilience, however, did not last over the entire period of the study.
Studies of birds under drought conditions show that responses to changes in environmental conditions may be species-specific. In Bushmanland, Sclater's lark (Spizocorys sclateri) showed some resistance to drought, nesting at a higher density during low rainfall than during a wetter spell (Table 6). During the dry period, the larks fed on, amongst others, the large seeds of a grass (Enneapogon desvauxii), a dependable resource in dry times because the seeds are held almost below ground at the base of the plant. Because of the difficulty of extracting the seeds, this resource is used only by Sclater's larks and Stark's larks (S. starki). Stark's larks are nomadic and absent during drier periods; Sclater's larks are thus able to use the resource without competition during droughts.
In species for which the costs of moving may be high, for example, dune larks (Calendulauda erythrochlamys) on sparsely vegetated 'islands' in the Namib Desert dune sea, some individuals nevertheless moved away during a drought, with the population on some islands reduced to about one half or one third of their former size.40 There was also a reduction in foraging group size during the drought. Foraging group sizes in wetter periods were 2–6 individuals,41 whereas during the drought no groups larger than two birds were seen.40 No shifts in diet or changes in foraging methods and patterns between the wetter period and the drought were apparent, but there were seasonal differences in foraging patterns, and presumably diet, regardless of whether there was a drought or not.40 Whether the larks remained on their patch or moved away was strongly dependent on plant composition and the state of the vegetation and invertebrate populations. Safriel40 suggested that in patches where plant and invertebrate resources remained rich, foraging patterns (and presumably diet) would remain similar all year round. Dune larks can probably only be resident in the absence of any competition. The Namib Desert supports large numbers of nomadic granivores following rainfall.42 Some nomadic species overlap temporarily in habitat with dune larks, but depart as the ecosystem dries out.
Dune larks are thus able to remain on their patches provided there is no potential competition.
Decision making: when to stay and when to go?
We have shown, in this brief review, that birds use two main strategies for coping with the environment getting wetter or drier. In general, 'wet' years stimulate higher densities of nests (i.e. smaller territories), larger clutch sizes, unseasonal breeding, and, depending on the time of year, higher breeding success. Rainfall over a certain amount20,35,36,43 triggers breeding in resident species and an influx of nomadic species that breed and then move on. The amount of rainfall needed to trigger breeding varies between areas and seasons; 60 mm of rain in two days, followed by 22 mm eight days later in summer (January) stimulated breeding in the Kalahari,35 but 41 mm in Bushmanland over several days in summer (February) without any follow-up rains did not trigger breeding.20 It did, however, lead to an influx of nomadic granivores which did not breed at that time, whereas 54 mm of rain in winter (July) in the same area triggered breeding in all species.20
Nomads move in response to environmental cues, which are poorly understood for small terrestrial granivores,5,44 but better understood for larger aquatic species.45 Environmental cues for aquatic bird species may include distant thunderstorms, indicating heavy rain and thus the formation of temporary pans.45 This scenario may also hold for small terrestrial insectivores, but for small avian granivores the lag between rainfall and the response by the plants may be a week or more. It has been suggested5 that drifts of awns of grasses (used by nomadic larks and buntings for nest linings) may provide a strong visual cue that the area is suitable for nesting, but which other cues draw the birds initially to the area is not known.
Coping with an environment that is drying out may be easier for nomadic species. The environment dries out, particular food items become scarce, and provided that their young are large and able to fly, the nomads move on. What they must decide is where, and not when, to go. For resident species, however, whether to go or stay is a more difficult and apparently individual process. The examples we give suggest that in resident bird communities, there is a community-wide response to below-average rainfall, with fewer birds breeding in dry years and lower than usual production of young. But whether to breed or not, to stay or not, or to join a larger group remain individual decisions, possibly influenced by the decisions made by conspecifics.
If birds make the decision to remain in their area during a drought, they have several options to increase their chances of survival. They can increase territory size if neighbouring territories have become vacant or poorly-defended; they can shift their diet to eat a wider range of items; or they can abandon territories and join mixed-species foraging flocks. There is very little evidence of any of these tactics being particularly obvious in drought years. Furthermore, the benefits of all tactics have to be traded off against the losses. Territories that have increased in size might be too large to defend when good times arrive, territories that are abandoned need to be re-established when the drought is over, and shifts in diet may have consequences for the health or reproductive success of individual birds. But more importantly, resident species usually know their territory or home range very well.46 They know good places to forage, to nest and to avoid predators. This knowledge will increase in quality over the years.47 This fact is supported by Hanmer38 who shows that adult birds survive droughts better than immature ones, and suggests that it is due to the adults' better knowledge of parts of the habitat that gives them an advantage. Resident bird species are thus more likely to be resistant to drought and to use available resources as best they can without giving up their patch.
This review is based on several talks given at the Arid Zone Ecology Forum, Sutherland, in September 2007. We thank the Critical Ecosystem Partnership Fund for sponsoring the attendance of W.R.J.D. at the forum, and we thank the committee of the Arid Zone Ecology Forum for funding towards writing this review. We are grateful to Sue Milton for comments and suggestions on the manuscript.
1. Robinson T. and Minton C. (1989). The enigmatic banded stilt. Birds Int. 1(4), 72–85. [ Links ]
2. Lea A. (1964). Some major factors in the population dynamics of the brown locust Locustana pardalina (Walker). In Ecological Studies in Southern Africa, ed. D.H.S. Davis, pp. 269–283. W. Junk, The Hague. [ Links ]
3. Botha D.H. (1967). The viability of brown locust eggs, Locustana pardalina (Walker). S. Afr. J. Sci. 10, 445–460. [ Links ]
4. Matthee J.J. (1978). Induction of diapause in eggs of Locustana pardalina (Walker) (Acrididae) by high temperatures. J. Ent. Soc. Sth. Afr. 41, 25–30. [ Links ]
5. Dean W.R.J. (2004). Nomadic Desert Birds. Adaptations of Desert Organisms series. Springer Verlag, Berlin, Heidelberg, New York. [ Links ]
6. Tyson P.D. (1986). Climatic Change and Variability in Southern Africa. Oxford University Press, Cape Town. [ Links ]
7. Anon (1991). A history of notable weather events in South Africa: 1500–1990. Caelum (December 1991). [ Links ]
8. Holmgren M., Scheffer M., Ezcurra E., Gutierrez J.R. and Mohren G.M.J. (2001). El Niño effects on the dynamics of terrestrial ecosystems. Trends Ecol. Evol. 16, 89–94. [ Links ]
9. Jaksic F.M. and Lazo I. (1999). Response of a bird assemblage in semiarid Chile to the 1997–1998 El Niño. Wilson Bull. 111, 527–535. [ Links ]
10. Grant P.R., Grant B.R., Keller L.F. and Petren K. (2000). Effects of El Niño events on Darwin's finch productivity. Ecology 81, 2442–2457. [ Links ]
11. Siegfried W.R. and Brooke R.K. (1989). Alternative life-history styles of South African birds. In Alternative Life-history Styles of Animals, ed. M.N. Bruton, pp. 385–420. Kluwer Academic Publishers, Dordrecht. [ Links ]
12. Lepage D. and Lloyd P. (2004). Avian clutch sizes in relation to rainfall seasonality and stochasticity along an aridity gradient across South Africa. Ostrich 75, 259–268. [ Links ]
13. Milton S.J., Davies R.A.G. and Kerley G.I.H. (1999). Population level dynamics. In The Karoo: Ecological Patterns and Processes, eds W.R.J. Dean and S.J. Milton, pp. 183–207. Cambridge University Press, Cambridge. [ Links ]
14. Milton S.J., Dean W.R.J. and Leuteritz T. (2004). Opportunistic and multiple breeding attempts in plants and vertebrates of semi-deserts with unpredictable rainfall events through the year. Trans. Roy. Soc. S. Afr. 59, 43–53. [ Links ]
15. Whitford W.G. (2002). Ecology of Desert Systems. Academic Press, San Diego. [ Links ]
16. Schmidt-Nielsen K. (1983). Animal Physiology: Adaptation and Environment. Cambridge University Press, Cambridge. [ Links ]
17. Dean W.R.J. and Milton S.J. (1999). Animal foraging and food. In The Karoo: Ecological Patterns and Processes, eds W.R.J. Dean and S.J. Milton, pp. 165–177. Cambridge University Press, Cambridge. [ Links ]
18. Burbidge A.A. and Fuller P.J. (1982). Banded stilt breeding at Lake Barlee, Western Australia. Emu 82, 212–216. [ Links ]
19. Rowley I. (1990). Behavioural Ecology of the Galah Eolophus roseicapillus in the Wheatbelt of Western Australia. Surrey Beatty & Sons, Chipping Norton, NSW. [ Links ]
20. Lloyd P. (1999). Rainfall as a breeding stimulus and clutch size determinant in South African arid-zone birds. Ibis 141, 637–643. [ Links ]
21. Vernon C.J. (1978). Breeding seasons of birds in deciduous woodland at Zimbabwe, Rhodesia, from 1970 to 1974. Ostrich 49, 102–115. [ Links ]
22. Skead C.J. (1995). Life-history Notes on East Cape Bird Species (1940–1990), Vol. 1. Western District Council, Port Elizabeth. [ Links ]
23. Kemp A.C. (2005). Crowned hornbill. In Roberts Birds of Southern Africa, VIIth edn., eds P.A.R. Hockey, W.R.J. Dean and P.G. Ryan, pp. 153–154. Trustees of the John Voelcker Bird Book Fund, Cape Town. [ Links ]
24. Anon (1982). Crowned hornbills irrupt again. Bee-eater 33, 24. [ Links ]
25. Vernon C, and Every B. (1980). Another influx of crowned hornbills. Bee-eater 31, 22–23. [ Links ]
26. Lack D. (1954). The Natural Regulation of Animal Numbers. Clarendon Press, Oxford. [ Links ]
27. van Zyl A.J. (1994). Sex-related local movement in adult rock kestrels in the eastern Cape Province, South Africa. Wilson Bull. 106, 145–158. [ Links ]
28. Wyndham E. (1986). Length of birds' breeding seasons. Am. Nat. 128, 155–164. [ Links ]
29. Yom-Tov Y. (1987). The reproductive rates of Australian passerines. Aust. Wildl. Res. 14, 319–330. [ Links ]
30. Schulze R.E. (1997). Climate. In Vegetation of Southern Africa, eds R.M. Cowling, D.M. Richardson and S.M. Pierce, pp. 21–42. Cambridge University Press, Cambridge. [ Links ]
31. Winterbottom J.M. and Rowan M.K. (1962). Effect of rainfall on breeding of birds in arid areas. Ostrich 33, 77–78. [ Links ]
32. Martin R., Martin J. and Martin E. (1986). Breeding in response to rainfall in the Karoo National Park. Bokmakierie 38, 36. [ Links ]
33. Gargett V., Gargett E. and Damania D. (1995). The influence of rainfall on Black eagle breeding over 31 years in the Matobo Hills, Zimbabwe. Ostrich 66, 114–121. [ Links ]
34. Vernon C.J. (1984). Population dynamics of birds in Brachystegia woodland. In Proceedings of the 5th Pan-African Ornithological Congress, ed. J. Ledger, pp. 201– 216. SAOS, Johannesburg. [ Links ]
35. Maclean G.L. (1970). The biology of the larks (Alaudidae) of the Kalahari sandveld. Zoo. Afr. 5, 7–39. [ Links ]
36. Maclean G.L. (1973). The sociable weaver, part 3: breeding biology and moult. Ostrich 44, 219–240. [ Links ]
37. Herremans M. (2004). Effects of drought on birds in the Kalahari, Botswana. Ostrich 75, 217–227. [ Links ]
38. Hanmer D.B. (1997). Bird longevity in the eastern highlands of Zimbabwe – drought survivors. Safring News 26, 47–54. [ Links ]
39. Dean W.R.J. and Milton S.J. (2001). The density and stability of birds in shrublands and drainage line woodland in the southern Karoo, South Africa. Ostrich 72, 185–192. [ Links ]
40. Safriel U.N. (1990). Winter foraging behaviour of the Dune Lark in the Namib Desert, and the effect of prolonged drought on behaviour. Ostrich 61, 77–80. [ Links ]
41. Cox G.W. (1983). Foraging behaviour of the dune lark. Ostrich 54, 113–120. [ Links ]
42. Willoughby E.J. (1971). Biology of larks (Aves: Alaudidae) in the central Namib Desert. Zool. Afr. 6, 133–176. [ Links ]
43. Simmons R.E. (1996). Population declines, viable breeding areas and management options for flamingos in southern Africa. Cons. Biol. 10, 504–514. [ Links ]
44. Dean W.R.J. and Williams J.B. (2004). Adaptations of birds for life in deserts with particular reference to larks (Alaudidae). Trans. Roy. Soc. S. Afr. 59, 79–91. [ Links ]
45. Simmons R.E., Barnard P. and Jamieson I.G. (1999). What precipitates influxes of wetland birds to ephemeral pans in arid landscapes? Observations from Namibia. Ostrich 70, 145–148. [ Links ]
46. Hinde R.A. (1956). The biological significance of the territories of birds. Ibis 98, 340–369. [ Links ]
47. Andersson M. (1980). Nomadism and site tenacity as alternative reproductive tactics in birds. J. Anim. Ecol. 49, 175–184. [ Links ]
Received 3 June 2008. Accepted 12 February 2009.
* Author for correspondence. E-mail: lycium@telkomsa.net














