Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.104 n.11-12 Pretoria Nov./Dec. 2008
RESEARCH LETTERS
Vegetation associated with the occurrence of the Brenton blue butterfly
D.A. EdgeI, *; S.S. CilliersII; R.F. TerblancheII
IP.O. Box 2586, Knysna 6570, South Africa
IISchool of Environmental Sciences and Development, North-West University, Private Bag X6001, Potchefstroom 2520, South Africa
ABSTRACT
The Brenton blue butterfly, Orachrysops niobe (Trimen), is critically endangered and known only from one site near Knysna in the Western Cape province of South Africa, now proclaimed as the Brenton Blue Butterfly Reserve (BBBR). We have explored associations between vegetation types and the presence of O. niobe's only host plant, Indigofera erecta (Thunb.), using Braun-Blanquet vegetation classification and ordination techniques as part of a broader research project at the BBBR. Positive correlations are demonstrated between the occurrence of I. erecta and certain thicket vegetation types dominated by Pterocelastrus tricuspidatus (candlewood trees). Ordinations using soil analysis and slope data have not detected significant environmental gradients influencing vegetation types. The high degree of vegetation heterogeneity at the BBBR appears to be driven in part by various disturbance histories. Historical ecological events at the site such as fire and megaherbivore impacts, and their role in sustaining the ideal habitat for I. erecta and O. niobe, are discussed. Management techniques for the BBBR such as controlled fires or the cutting of paths through the vegetation are evaluated and an optimum future management strategy is recommended. This is the most comprehensive vegetation study ever carried out at the habitat of an endangered butterfly in South Africa, and breaks new ground by using vegetation analysis to develop a well-informed management plan for conservation of this species. It has significance for the management of small sites where many such endangered butterflies occur.
Introduction
Orachrysops niobe (Trimen)—the Brenton blue butterfly—is red-listed as 'Critically Endangered'1 and is known only from Brenton-on-Sea near Knysna. Anthropogenic influences2 led to its extinction at Nature's Valley 50 km to the east in the 1970s. The Brenton-on-Sea population was saved after government expropriated the relevant properties3 and proclaimed them as a special nature reserve in July 2003, named the BBBR.
Research into the ecological factors critical to O. niobe's survival started in 2000.2 A key research tool has been vegetation analysis, which is used increasingly to diagnose the critical habitat requirements of rare and localised Lepidoptera.4–7 Vegetation analysis requires a combination of physiognomic, floristic and environmental data to identify, classify and associate vegetation communities, using tools such as multivariate statistical analysis.8
An earlier study has provided a floristic description of the vegetation of the BBBR, and has made a preliminary diagnosis of the site's ecological processes.6 Recommendations included further research into the biology and autoecology of Indigofera erecta, the host plant of O. niobe (addressed elsewhere2), and more detailed studies of the vegetation communities and environmental factors present in sites where I. erecta occurs. The purpose of the present study is to classify and describe the vegetation communities at the BBBR, and to identify and evaluate associations between vegetation types or environmental factors and I. erecta occurrence. The ecological dynamics at the BBBR are evaluated, and appropriate management techniques to sustain the O. niobe population are proposed.
Methods
Study site
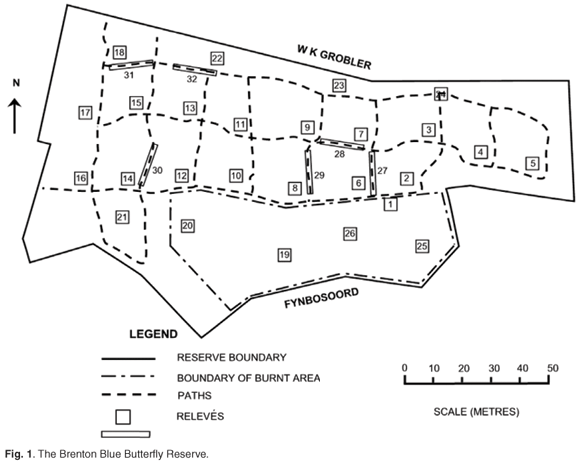
The BBBR (Fig. 1) is located at coordinates 34°04'S; 23°02'E, and between 90–115 m above mean sea level (a.m.s.l.). It is on a south-facing slope with an average inclination of 1 in 3 (18°), varying between 10° and 26°. The total area is 14 670 m2.
The local geology consists of three Pleistocene aeolianite palaeodune cordons running parallel to the coast, displaying cross-bedding and able to support steep slopes.9,10 Ages are estimated at 0.8–1.0 Myr for the landward cordon, c. 200 kyr for the middle cordon, and <130 kyr for the seaward cordon.9 The Brenton peninsula, where the BBBR is located, is the eastern-most extension of these cordons, with the aeolianites overlain in places by Holocene sands and a 0.5–2-m-deep immature sandy soil layer with low fines. The BBBR is situated about half-way up the southern slope of the secondary dune cordon inland from the coast, with the tertiary dune to the north rising to 210 m a.m.s.l. before falling away to the Knysna river estuary (see location map online at www.sajs.co.za).
According to the Köppen classification system,11 this area has a Cfa climate (warmest month >22°C with sufficient precipitation in all months, and no frost). The mean annual rainfall for Knysna is 750 mm, with a bimodal pattern—most of the rain falls in spring and autumn, with a dry period in late summer. The average daily maximum and minimum temperatures are 23.3°C and 12.5°C. The rainfall seasonality index of <10 and the coefficient of annual variation of precipitation (<25%) is among the lowest in southern Africa. Droughts are rarely severe.12 The mean annual relative humidity is high at 88%, and there is a significant incidence of coastal fog when the sea-surface temperature is lower. Prevailing winds are southeasterly during summer and southwesterly during winter. Dry northerly berg winds, especially during winter, cause a severe reduction of humidity and an increased fire hazard.
The Brenton peninsula has been mapped as Knysna Sand Fynbos (FFd10), part of the fynbos biome.13 At a finer scale, this mosaic of dune thicket, fynbos and forest has been classified as Goukamma Dune Thicket, occurring on relatively fire-protected moist south-facing slopes of palaeodunes, with rather poor calcareous aeolian soils, enriched by a build-up of humic material.14 The BBBR was described in an earlier study as a mosaic of asteraceous coastal fynbos and thicket patches dominated by P. tricuspidatus (candlewood).6 Recent management actions at the BBBR have included felling of P. tricuspidatus followed by controlled burning in September 2000 and cutting of a network of access paths in July 2001 (Fig. 1).15
Sampling and data recording
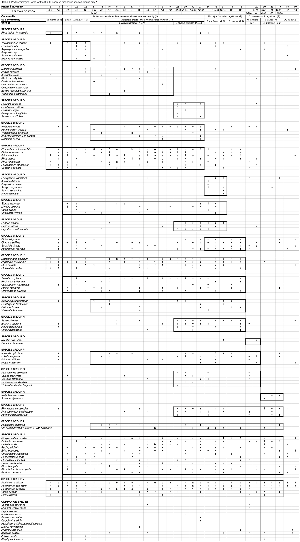
We have applied Braun-Blanquet methodology16–18 with 5 m × 5 m square relevés in the undisturbed or burnt areas, and 1.25 m × 20 m relevés for the paths. A stratified random sampling design was employed, with stratification based on the different disturbance regimes. Twenty relevés represented the undisturbed areas, six of the burnt area, and six of the paths, giving a total of 32 relevés (Fig. 1).
Plant identifications and taxonomy have been done by reference to relevant publications.19–24 Voucher specimens have been deposited at The South Cape Herbarium, the National Herbarium and the North-West University Herbarium. Midpoint values of the Braun-Blanquet scale of cover abundance were used for the ordinations.16,17
Each plant species was allocated to a life-form category (ferns, monocots, other herbs, shrubs and trees), and the height of the largest example of each species of shrub or tree and the occurrence of bare ground in each relevé was recorded. The distinction between shrubs and trees follows that of Edwards.25
The slope of each relevé was measured, and any observable disturbance (e.g. fire, cutting or soil disturbance) was recorded. Bulk soil samples were taken for relevés 1–24 and analysed using standard protocols.2,26
A census was conducted of the Pterocelastrus tricuspidatus (candlewood) trees at the BBBR between December 2001 and January 2002. Each candlewood tree's exact location, height, width of crown and number of boles was recorded, and included trees that had been felled during 2000 and which were coppicing. During the I. erecta monitoring programme,2 the distance of each I. erecta plant from the closest candlewood tree (or coppice) was recorded, as well as whether it was within the crown cover of this tree (or would have been before the tree was felled) or not .
Data analysis
Vegetation data were captured using the TURBOVEG software package for processing phytosociological data.27 Braun-Blanquet procedures8,16 were applied to arrange relevés and species into a structured phytosociological table, assisted by the MEGATAB visual editor software program.28 A detrended correspondence analysis (DCA) ordination algorithm29 was applied to the data set, to confirm phytosociological associations and to assess floristic relations between communities. A stepwise ordination approach30 was adopted to deal with the heterogeneity of the data set.
The alpha diversity of each relevé was assessed from the number of species (richness) and by computing Shannon diversity indices H' (which gives more weight to rare species).31 The communities identified in the phytosociological analysis have been statistically compared by Tukey's test for unequal sample sizes,32 using the STATISTICA 7 software program.33 Significant differences have been accepted at the P < 0.01 level.
Vegetation communities have been identified using all the above data. Names have been allocated that incorporate diagnostic and dominant species present in each community. Each vegetation community has been described with reference to the dominant plant species, the life forms occurring in the community, the patterns of species richness and diversity, any environmental factors of importance, the record of disturbance of that community, and the extent of occurrence of I. erecta within the community.
Results and discussion
Floristics
The total of 128 vascular plant species that we have recorded in the relevés at the BBBR2 is similar to findings of an earlier study.6 The intermediate disturbance hypothesis predicts that sites with an intermediate frequency of disturbance would have higher biodiversity than sites with low or high frequencies of disturbance.34,35 This hypothesis has been tested for fire frequency in fynbos.36 Higher species diversity and community heterogeneity has been found at the lowest disturbance frequencies (40 years or more between fires). Biodiversities are lower at intermediate and high disturbance frequencies (from 26 to 6 years between fires). Prior to 2000, the BBBR had not been burnt for at least 40 years2 and the 128 species recorded in the 800 m2 sample area exceed the 60–90 species per 1000 m2 recorded in the infrequently burnt fynbos plots.36 It also exceeds the average for the fynbos biome of 65.7 species per 1000 m2 reported in another study.37 The floristic data demonstrate a high degree of species turnover (29% from 1996–2003). Only 5 of the 37 apparently replaced species are geophytes or annuals—the balance are perennial herbs, shrubs and dwarf shrubs.
Vegetation types
The diagnostic species groups from a phytosociological table (see Table 1 in supplementary material online at www.sajs.co.za) has enabled three vegetation communities, three subcommunities and six variants to be identified and classified. A detrended correspondence analysis has been performed to confirm this classification (Fig. 2). Thicket subcommunities 1.1 and 1.2 are positioned either side of the ordination, with subcommunity 1.3 between them. The relevés at the top of the ordination (1, 18, 19, 20, 25, 26 – subcommunities 3.1 and 3.2) are all dominated by P. aquilinum and have all been burnt. The fynbos-dominated subcommunities 2.1 and 2.2 have some overlap with the P. tricuspidatus-dominated community 1.3.
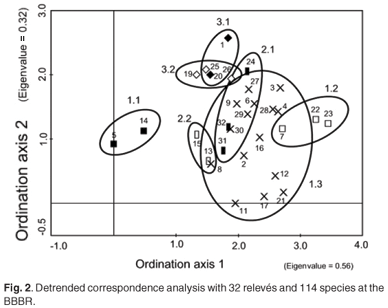
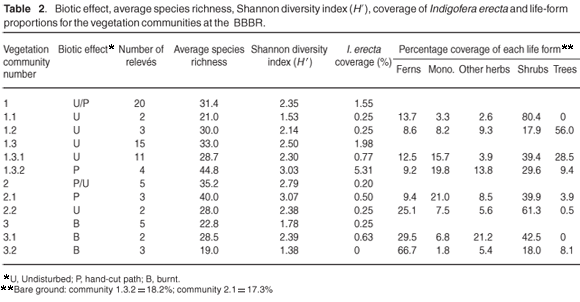
The disturbance history (biotic effect)—alpha diversity, expressed as average species-richness and Shannon diversity index, the coverage of I. erecta, and the proportions of life forms for each vegetation community—is given in Table 2. The average Shannon diversity indices for the burnt relevés are significantly lower than for the undisturbed relevés (T = –4.72; P < 0.0001); and are significantly higher for the path relevés than for the undisturbed relevés (T = 2.87; P < 0.01). There were significant differences in the coverage of I. erecta and the proportion of life forms.
Environmental factors
Data are summarised in Table 3. The soil classification ranges from sand to sandy loam. The chemical factors with the highest variability are P, K and Na. There are significant correlations between pH and Ca content (r = 0.50, P = 0.011) and between K and S-value (r = 0.60, P = 0.0022). There is also some weak evidence of relationships between the coverage of I. erecta and pH (r = 0.39, P = 0.060), and Ca content (r = 0.40, P = 0.052).

The main soil features at the BBBR are compared with published data on typical fynbos and thicket sites.38,39 The BBBR soil pH is higher (range 4.53–6.38) than fynbos sites (range 3.99–4.44) and more typical of thicket or forest sites (range 4.60–4.62). The BBBR calcium content (range 400–1208 ppm) is also much higher than fynbos sites (average 225 ppm) and similar to thicket sites (807 ppm). The range of silt + clay content and S-values can support either thicket or fynbos. Available phosphorus (average 12.8 ppm) (closely correlated with S-values but very variable) is more typical of thicket or forest sites (10–12.8 ppm) than fynbos sites (3.2–6.0 ppm). The sodium (and associated chloride) levels are unusually high at the BBBR (average 99 ppm), attributed to sea spray and sea-mist precipitation.
One of these studies38 has associated fynbos occurrence with higher soil sand contents, and thicket occurrence with higher pH, nutrients and slope, with considerable variation evident over fairly short distances. Each community appears to impact on its own soil chemistry, giving stability to communities.
The most interesting feature of the species–environmental factor ordinations done in the present study is that I. erecta consistently appears near the centroid of the ordination, indicating that most of the BBBR has suitable environmental factors for the growth of I. erecta. The actual distribution of I. erecta cannot be convincingly associated with any of these environmental factors, except tenuously with pH and Ca content. The species composition and structure of the immediate vegetation community has the greatest influence on the persistence of I. erecta.
Description of vegetation types
Ficinia ramosissima – Pterocelastrus tricuspidatus thicket community (1)
The thicket component of the mosaic prevails in this vegetation type, with dominant tree species P. tricuspidatus and diagnostic species Ficinia ramosissima, Erica speciosa, Rhus glauca, Rhus tomentosa, Peucedanum ferulaceum and Cassytha ciliolata (species group A). Three subcommunities can be distinguished:
Rhus lucida subcommunity (1.1)
R. lucida form lucida (with larger leaves than form scoparia) forms the dense thicket patches characteristic of this subcommunity (species group B). There is a markedly diminished species richness, Shannon diversity index, and abundance of herbaceous plants including I. erecta, relative to the other thicket subcommunities 1.2 and 1.3 (Table 2).
Sutera cordata – Tarchonanthus littoralis subcommunity (1.2)
T. littoralis dominates this subcommunity, with diagnostic species S. cordata and Asparagus asparagoides (species group C). This subcommunity occurs in the northern part of the BBBR, where T. littoralis-dominated thickets became established, following disturbances during road construction in the area. This subcommunity has higher species diversity than subcommunity 1.1, a preponderance of trees and shrubs, low abundance of I. erecta, and a higher than average slope (Tables 2 & 3).
Indigofera erecta – Pterocelastrus tricuspidatus subcommunity (1.3)
This subcommunity is the most prevalent at the BBBR (15 out of 32 relevés, or 47%), with species group D diagnostic (I. erecta, Hermannia involucrata, Thamnocortus cinereus, Knowltonia vesicatoria humilis and Metalasia densa). Dominant mature P. tricuspidatus trees provide dappled shade where many herb, graminoid and dwarf shrub species grow, including I. erecta. There are two variants:
Morella quercifolia – Pterocelastrus tricuspidatus variant (1.3.1)
Species group E is diagnostic for this variant (Morella quercifolia, Aristea ensifolia, Kniphofia uvaria, Rhoicissus digitata, Salvia africana-lutea, Haemanthus coccineus, Chironia melampyrifolia, Euclea racemosa bernardii and Tetragonia decumbens), but the absence of species group F is also a reliable indicator. The abundance of Indigofera erecta at 0.77% is above average but not as high as it is in variant 1.3.2 (Table 2).
Polygala fruticosa – Indigofera erecta variant (1.3.2)
Species group F is diagnostic for this variant (Polygala fruticosa, Helichrysum rutilans, Passerina rigida, Pelargonium longifolium and Senecio glastifolius). Dominant species are P. tricuspidatus, Ficinia ramosissima, Erica speciosa, Pteridium aquilinum and Helichrysum cymosum. It has the highest abundance of I. erecta (5.31%), and Shannon diversity index (Table 2), and all the relevés representing this variant are situated on hand-cut paths (Fig. 1).
Ficinia oligantha – Pteridium aquilinum fynbos community (2)
Dominant species in this community are P. aquilinum, Helichrysum cymosum, Helichrysum petiolare, Leucadendron salignum and Asparagus rubicundus. Species group G is weakly diagnostic for this community (Ficinia oligantha, Cineraria geifolia, Helichrysum foetidum and Struthiola hirsuta), which is best characterised by the low abundance of species groups E and F. The community occurs in patches within the thicket/fynbos mosaic, representative of the fynbos elements. Shrubs dominate this community (Table 1), and it has the highest species richness and Shannon diversity index, but the lowest abundance of I. erecta (0.20% coverage – Table 1). There is a lower than average soil phosphorus content (Table 2).
Pelargonium capitatum – Ficinia oligantha subcommunity (2.1)
Species group H (including Pelargonium capitatum and Selago corymbosa) is diagnostic for this subcommunity, which has some affinities with variant 1.3.2 (species group J) – also situated on hand-cut paths. The subcommunity has the highest species richness and Shannon diversity index of all the identified vegetation types at the BBBR, and is dominated by shrubs and graminoids (Table 1).
Struthiola hirsuta – Leucadendron salignum subcommunity (2.2)
The absence of species groups H, I and J best characterises this subcommunity, which is dominated by shrubs and ferns, with lower species richness and Shannon diversity index than in subcommunity 2.1 (Table 2).
Cullumia decurrens – Pteridium aquilinum community (3)
This community occurs in an area that had been burnt 18 months before sampling and is dominated by P. aquilinum, Cullumia decurrens, H. cymosum and Rhynchosia chrysoscias. The community has no diagnostic species but is characterised by the absence of L. salignum and A. rubicundus. The species richness and Shannon diversity index in this community are the poorest, with ferns and shrubs dominant and a low coverage of I. erecta (0.25%, Table 2).
Muraltia ericifolia – Cullumia decurrens subcommunity (3.1)
Manual control of bracken fern P. aquilinum has produced this subcommunity, and the shrub C. decurrens is now dominant. Diagnostic species are M. ericifolia and Geranium incanum (species group M).
Indigofera verrucosa – Chrysanthemoides monilifera subcommunity (3.2)
P. aquilinum completely dominates this subcommunity, with I. verrucosa and Solanum rigescens (species group P) diagnostic although they never occur in any abundance. The species richness and Shannon diversity are the lowest, with I. erecta completely absent from this variant (Table 2).
Vegetation associations with the occurrence of O. niobe
With earlier South African investigations of the habitat requirements of endangered butterfly species,4,7 the host plants were far more widespread than the butterflies. The presence of the correct host ant was the critical factor, and oviposition relied on a host ant-provided cue. It has been shown that the habitat favoured by the host ants is determined by the vegetation composition and structure. Studies of Maculinea butterflies in Europe have also shown the critical impact of vegetation structure in providing suitable habitat for the correct host ant.40–42
O. niobe females will oviposit only on I. erecta host plants of the correct ecotype (such as occurs at the BBBR) and do not respond to an ant-provided cue.2 O. niobe's host ant Camponotus baynei is relatively widespread in thicket and fynbos vegetation,43 and consequently O. niobe's population size is principally dependent on the abundance of I. erecta.2 Table 2 reveals that I. erecta occurs mostly in P. tricuspidatus dominated thicket patches (variant 1.3.2). It occurs only rarely in R. lucida and T. littoralis-dominated thicket patches (subcommunities 1.1 and 1.2), which are too dense and tend to exclude herbaceous species. It is also found only rarely in fynbos-dominated patches within the thicket– fynbos mosaic (community 2), and in areas that have been burnt more than two years previously (community 3).
Correlation between the occurrence of I. erecta plants and P. tricuspidatus trees
P. tricuspidatus is an important tree in Southern Afrotemperate Forest44 and is relatively common in the nearby Knysna forests. At the BBBR, it cannot attain the height it can reach in forests (up to 25 m), because of the nutrient-poor soil, consequently taking a shrub form which gives dappled shade with several boles and a height of <7 m. Mature P. tricuspidatus at the BBBR had an average height of 4.3 m and crown cover of 18.1 m2. In order to take into account the trees that had been felled, we assume that their crown cover (and hence their diameter) had been similar to the mature trees. The I. erecta plants that would have been in the shade of these trees have been included, to calculate the figures in Table 4. It was thus possible to compare the paths containing I. erecta plants that were in the shade of a P. tricuspidatus with the paths that were not. The average density of I. erecta plants in the shade of a P. tricuspidatus is 10 times greater than when no P. tricuspidatus is present (Table 4).

Our explanation is that the dappled shade under such P. tricuspidatus trees promotes the abundance of I. erecta, which has the following competitive advantages in a microclimate of this kind, because of:
-
its preference for a cooler, more mesic microclimate;2
-
its ability to photosynthesise better in dappled shade;2
-
the prostrate growth form that occupies space close to the ground.2
Disturbance regimes
Hardly any I. erecta plants persist in the relevés that were burnt in October 2000 (Table 2, subcommunity 3.2). The post-fire community is dominated by bracken fern, has far fewer trees, and has a diminished monocotyledon/herb layer (Table 2). Bracken fern regrows rapidly from its rhizome structure after its aerial fronds have been destroyed by fire,8,45 preoccupying space from slower-developing plants (including I. erecta), these having a low recruitment success after the canopy has closed.2
Cutting of paths has increased the abundance of I. erecta, particularly in the P. tricuspidatus-dominated thicket patches (Table 2, community 1.3.2). Dormant rootstocks and seeds in the soil in these areas must have been present and able to respond to a disturbance. However, cutting paths in the fynbos-dominated patches did not stimulate as much growth of I. erecta (Table 2, community 2.1).
The relatively small reserve in which O. niobe currently exists is no longer part of a natural ecosystem. We postulate that the manually cut paths may be structurally similar to the openings that used to be generated by larger megaherbivores.2 Elephants (Loxodonta africana), buffalo (Syncerus caffer) and eland (Tragelaphus oryx) were ubiquitous along the coastal belt in the eastern parts of the Western Cape up to two centuries ago, but are now absent.46 Even if these openings were only temporary, they would have provided patches of suitable habitat essential to the survival of the Brenton blue butterfly in the past. A study in the Knysna forest47 has found that the elephant's varied diet when feeding in fynbos consists of many of the species commonly found at the Brenton site, including P. aquilinum, from which they eat the rhizomes, the bark from P. tricuspidatus, and the entire plant in the case of Erica species, Leucadendron species, R. lucida and C. monilifera. Currently, the bushbucks Tragelaphus scriptus and bush pigs Potamochoercus porcus that regularly visit the site have a beneficial effect on O. niobe by generating disturbances.2
Conclusions
The BBBR has an above-average floristic diversity as a result of climatic and soil conditions (high pH, calcium and phosphorus) that are suitable for either fynbos or thicket to form, and this diversity has been promoted by the long-term absence of fire at the site.
The occurrence of O. niobe is critically dependent on the abundance of its larval host plant, I. erecta.2 There is no significant correlation between variations in I. erecta abundance in the BBBR and any environmental factor. The vegetation types hosting the greatest abundance of I. erecta are the P. tricuspidatus-dominated thicket patches. The shade in the vicinity of P. tricuspidatus trees creates the ideal microclimate for I. erecta. The vegetation types that have developed after a controlled fire at the BBBR only support a scattering of I. erecta individuals. By contrast, the vegetation types created by the cutting of paths have a far greater abundance of I. erecta, especially in the vicinity of P. tricuspidatus trees.
We conclude that fire is an inappropriate management tool to use at the BBBR. A combination of path cutting and selective removal of certain thicket species (T. littoralis and R. lucida) shows evidence of being the most beneficial and sustainable strategy for the protection of I. erecta populations and the viability of the Brenton blue butterfly. Historically, a similar disturbance pattern may have arisen from megaherbivore activities, now absent from the area.
CapeNature are thanked for allowing access to the BBBR for research. The Brenton Blue Trust and the Green Trust are thanked for financial support. North-West University assisted with sampling equipment and soil analyses. The South Cape Herbarium and the National Herbarium (Pretoria) provided valuable identification of plant species.
1. Henning G.A., Terblanche R.F and Ball J.B. (eds) (in press). South African Red Data Book: butterflies. SANBI Biodiversity Series. South African National Biodiversity Institute, Pretoria. [ Links ]
2. Edge D.A. (2005). Ecological factors influencing the survival of the Brenton Blue butterfly Orachrysops niobe (Trimen) (Lepidoptera: Lycaenidae). Ph.D. thesis, North-West University, South Africa. [ Links ]
3. Steenkamp C. and Stein R. (1999). The Brenton Blue Saga – a case study of South African biodiversity conservation, p.1. Endangered Wildlife Trust of Southern Africa, Johannesburg. [ Links ]
4. Deutschländer M.S. and Bredenkamp G.J. (1999). Importance of vegetation analysis in the conservation management of the endangered butterfly Aloeides dentatis dentatis (Swierstra) (Lepidoptera, Lycaenidae). Koedoe 42(2), 1–12. [ Links ]
5. O'Dwyer C. and Attiwill P.M. (1999). A comparative study of the habitats of the Golden Sun Moth Synemon plana Walker (Lepidoptera: Castniidae): implications for restoration. Biol. Conserv. 89, 131–141. [ Links ]
6. Lubke R.A., Hoare D., Victor J. and Ketelaar R. (2003). The vegetation of the habitat of the Brenton Blue butterfly, Orachrysops niobe (Trimen), in the southern Cape, South Africa. S. Afr. J. Sci. 99, 201–206. [ Links ]
7. Terblanche R.F., Morgenthal T.L. and Cilliers S.S. (2003). The vegetation of three localities of the threatened butterfly species Chrysoritis aureus (Lepidoptera: Lycaenidae). Koedoe 46(1), 73–90. [ Links ]
8. Kent M. and Coker P. (1994). Vegetation Description and Analysis – a practical approach. John Wiley, Chichester. [ Links ]
9. Illenberger W.K. (1996). The geomorphic evolution of the Wilderness dune cordons, South Africa. Quat. Internat. 33, 11–20. [ Links ]
10. Bateman M.D., Holmes P.J., Carr A.S., Horton B.P. and Jaiswal M.K. (2004). Aeolianite and barrier dune construction spanning the last two glacial–interglacial cycles from the southern Cape coast, South Africa. Quat. Sci. Rev. 23, 1681–1698. [ Links ]
11. Schulze R.E. and McGee O.S. (1978). Climatic indices and classifications in relation to the biogeography of southern Africa. In Biogeography and Ecology of Southern Africa, ed. M.J.A. Werger, pp. 19–55. W. Junk, The Hague. [ Links ]
12. Schulze R.E. (1997). Climate. In Vegetation of Southern Africa, eds R.M. Cowling, D.M. Richardson and S.M. Pierce, pp. 21–42. Cambridge University Press, Cambridge [ Links ]
13. Mucina L., Rutherford M.C. and Powrie L.W. (eds) (2005). Vegetation map of South Africa, Lesotho and Swaziland, 1: 1 000 000 scale sheet maps. South African National Biodiversity Institute, Pretoria. [ Links ]
14. Vlok J.H.J., Euston-Brown D.I.W. and Cowling R.M. (2003). Acocks's Valley Bushveld 50 years on: new perspectives on the delimitation, characterisation and origin of subtropical thicket vegetation. S. Afr. J. Bot. 69(1), 27–51. [ Links ]
15. Edge D.A. (2002). Some ecological factors influencing the breeding success of the Brenton Blue butterfly, Orachrysops niobe (Trimen) (Lepidoptera: Lycaenidae). Koedoe 45(2), 19–34. [ Links ]
16. Mueller-Dombois D. and Ellenberg, H. (1974). Aims and Methods of Vegetation Ecology. John Wiley, New York. [ Links ]
17. Werger M.J.A. (1974). On concepts and techniques applied in the Zürich- Montpellier method of vegetation survey. Bothalia 11, 309–323. [ Links ]
18. Westhoff V. and Van der Maarel E. (1980). The Braun-Blanquet approach. In Classification of Plant Communities, eds R.H. Whittaker and W. Junk, pp. 287–399. W. Junk, The Hague. [ Links ]
9. Bohnen P. (1986). Flowering Plants of the Southern Cape. Cape & Transvaal Book Printers, Parow. [ Links ]
20. Bohnen P. (1995). More Flowering Plants of the Southern Cape. The Still Bay Conservation Trust, Still Bay. [ Links ]
21. Moriarty A. (1997). South African Wild Flower Guide 2: Outeniqua, Tsitsikamma & Eastern Little Karoo. Botanical Society of South Africa, Kirstenbosch. [ Links ]
22. Goldblatt P. and Manning J. (2000). Cape Plants: a conspectus of the Cape flora of South Africa – Strelitzia 9. National Botanical Institute of South Africa & Missouri Botanical Garden, Cape Town. [ Links ]
23. Manning J. (2001). South African Wild Flower Guide 11: Eastern Cape. Botanical Society of South Africa in association with the National Botanical Institute, Kirstenbosch. [ Links ]
24. Germishuizen G. and Meyer N.L. (eds) (2003). Plants of Southern Africa: an annotated checklist – Strelitzia 14. National Botanical Institute of South Africa, Pretoria. [ Links ]
25. Edwards D. (1983). A broad-scale structural classification of vegetation for practical purposes. Bothalia 14, 3 & 4, 705–712. [ Links ]
26. Council of Soil Testing and Plant Analysis (1974). Handbook on Reference Methods for Soil Testing. Athens, GA. [ Links ]
27. Hennekens S.M. (1996). TURBO (VEG) for Windows: Software package for input, processing and presentation of phytosociological data. Version 1.7 IBN-DLO: user's guide. University of Lancaster, U.K. [ Links ]
28. Hennekens S.M. (1996). MEGATAB: a visual editor for phytosociological tables. Giesen & Geurts, Ulft. [ Links ]
29. Hill M.O. (1979). DECORANA – a Fortran program for detrended correspondence analysis and reciprocal averaging. Cornell University, New York. [ Links ]
30. Mucina L. and Van Tongeren O.F.R. (1989). A coenocline of the high-ranked syntaxa of ruderal vegetation. Vegetation 81, 117–125. [ Links ]
31. Begon M., Harper J.L. and Townsend C.R. (1996). Ecology, 3rd edn. Blackwell Science, Oxford. [ Links ]
32. Spjotvoll E. and Stoline M.R. (1973). An extension of the T-method of multiple comparison to include cases with unequal sample sizes. J. Am. Stats 68, 976–978. [ Links ]
33. Statistica for Windows (1995). Computer program manual. StatSoft, Tulsa, OK. [ Links ]
34. Connell J.H. (1978). Diversity in tropical rainforests and coral reefs. Science 199, 302–1310. [ Links ]
35. Huston M. (1979). A general hypothesis of species diversity. Am. Nat. 113, 81–101. [ Links ]
36. Schwilk D.W., Keeley J.E. and Bond W.J. (1997). The intermediate disturbance hypothesis does not explain fire and diversity pattern in fynbos. Plant Ecol. 132, 77–84. [ Links ]
37. Cowling R.M., Richardson D.M. and Mustard P.J. (1997). Fynbos. In Vegetation of Southern Africa, eds R.M. Cowling, D.M. Richardson and S.M. Pierce, pp. 99– 130. Cambridge University Press, Cambridge. [ Links ]
38. Manders P.T. (1990). Fire and other variables as determinants of forest/fynbos boundaries in the Cape Province. J. Veg. Sci. 1, 483–90. [ Links ]
39. Cowling R.M. and Holmes P.M. (1997). Flora and vegetation. In Ecology of Fynbos – Nutrients, fire and diversity, ed. R.M. Cowling, pp. 23–61. Oxford University Press, Cape Town. [ Links ]
40. Thomas J.A. (1984). The behaviour and habitat requirements of Maculinea nausithous (the dusky large blue) and M. teleius (the scarce large blue) in France. Biol. Conserv. 28, 325–347. [ Links ]
41. Thomas J.A. (1995). The ecology and conservation of Maculinea arion and other European species of Large Blue butterflies. In Ecology and Conservation of Butterflies, ed. A.S. Pullin, pp. 180–197. Chapman & Hall, London. [ Links ]
42. Thomas J.A., Simcox D.J., Wardlaw J.C., Elmes G.W., Hochberg M.E. and Clarke R.T. (1998). Effects of latitude, altitude and climate on the habitat and conservation of the endangered butterfly Maculinea arion and its Myrmica ant hosts. J. Ins. Conserv. 2, 39–46. [ Links ]
43. Edge D.A., Robertson, H.G. and van Hamburg H. (2008). Ant assemblages at three potential breeding sites for the Brenton Blue butterfly Orachrysops niobe (Trimen). Afr. Entomol. 16, 253–262. [ Links ]
44. Mucina L. and Rutherford M.C. (eds) (2006). The Vegetation of South Africa, Lesotho and Swaziland. Strelitzia 19. South African National Biodiversity Institute, Pretoria. [ Links ]
45. Burge M.N. and Kirkwood R.C. (1992). The control of bracken. Crit. Rev. Biotech. 12, 299–333. [ Links ]
46. Skinner J.D. and Chimimba C.T. (2005). The Mammals of the Southern African Subregion. Cambridge University Press, Cape Town. [ Links ]
46. Milewski A.V. (2002). Elephants and fynbos. Veld & Flora March 2002, 28. [ Links ]
Received 10 May. Accepted 3 September 2008.
This article is accompanied by a supplementary phytosociological table and a location map online at www.sajs.co.za
* Author for correspondence. E-mail: daveedge@xnets.co.za
Supplementary material to:
Edge D.A., Cilliers S.S. and Terblanche R.F. (2008). Vegetation associated with the occurrence of the Brenton blue butterfly. S. Afr. J. Sci. 104, 505–510.