Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.104 n.9-10 Pretoria Sep./Oct. 2008
REVIEW ARTICLE
Application of active biomonitoring within an integrated water resources management framework in South Africa
V. Wepener
Centre for Aquatic Research, Department of Zoology, University of Johannesburg, P.O. Box 524, Auckland Park 2006, South Africa. E-mail: victorw@uj.ac.za
ABSTRACT
Because waste water and runoff from surrounding catchments are a main source of direct and continuous input of pollutants in aquatic ecosystems, the study of the effects of in-stream exposure on organisms has a high ecological relevance. However, correlating observed effects with specific pollutants or even classes of pollutants remains difficult, due to the usually unknown, complex and often highly variable composition of these source waters. By integrating multiple endpoints at different ecologically relevant levels of organization within one test organism, it is possible to gain an understanding of how different levels of organization within this organism respond to toxic exposure, and how responses at these different levels are interrelated. The use of biological markers in transplanted organisms, referred to as active biomonitoring (ABM), is demonstrated in this paper. The correct choice of bioindicator organism and suite of biomarkers makes it possible to assess the effects of wastewater and runoff water in terms of known environmental effects (such as effluents containing endocrine-disrupting chemicals or pulp mill effluent) as well as runoff water with an uncharacterized composition of contaminants (for instance storm-water runoff from industrial complexes). The applicability of ABM as a cause–effect assessment protocol is demonstrated through a case study in South Africa that relates to stressor identification within a system exposed to urban and industrial waste water. This paper proposes a multi-tiered framework that allows for the incorporation of ABM within the existing South African integrated water resources management framework.
Introduction
Because water is the basis for all organisms and ecosystems, the protection of aquatic resources is essential in preserving entire ecosystems.1 The original environmental problems that influenced the development of aquatic toxicology were relatively simple and involved pollutants such as uncharacterized organic matter, hazardous metals, other chemical species and pesticides.2 Regulatory goals, which generally drive the development of toxicological monitoring methods, have thus been initially reactive, affording assessment tools for the restoration of aquatic ecosystems. The toxicity tests were primarily based on a single-component, chemistry-based approach. Aquatic contamination problems have now become more complex, involving pollutant compositions in one or more environmental matrices (involving, for example, water, sediment, and living organisms).3 Effects can thus be more insidious than assessed when the endpoint of measurement is simply mortality. When effects are detected, the causative agent(s) might not be readily apparent. It is becoming more evident that effluents are a main source of direct and continuous pollutant input in aquatic ecosystems and represent a major threat to communities in receiving aquatic ecosystems. This is especially the case in densely populated and industrial areas where point and diffuse sources of xenobiotics pollute aquatic ecosystems with complex mixtures of problem chemicals, and are associated with other anthropogenic perturbations (such as habitat modification and/or loss) that have impacted severely on the integrity of ecosystems.4,5 Regulatory goals have consequently shifted from reactive treatments to pre-emptive approaches that are aimed at protecting, enhancing and preserving the ecosystem. This has necessitated a shift away from isolated surrogate methods, based on single-species toxicity tests and chemical measurements, to a more integrated and holistic exposure assessment, to reflect the total health status of ecosystems. This holistic approach draws physico-chemical, toxicological and ecological lines of evidence into an integrated assessment of aquatic ecosystem health.6
Ecosystem health assessment in South Africa
The realization in South Africa of an integrated procedure has brought about two divergent approaches to ecosystem health assessment. The concept of ecosystem health as an acceptable and meaningful term has elicited much debate7 in itself. It is a truism to state that for an ecosystem to be healthy—that is, to function optimally—the constituent biota (both fauna and flora) comprising it also have to be healthy. The two approaches adopted to assess aquatic health have been in response to specific information needs. The first involves biomonitoring, which has been used initially in the South African River Health Programme (RHP), which was developed to assess the ecological status of riverine ecosystems in relation to all the anthropogenic disturbances affecting them. It was never intended to assess site-specific impacts or conditions. It is a screening-level monitoring programme that uses a low sampling frequency and a low resolution of sites scattered semi-randomly across catchments.8 The RHP assessment is based on the concept of biological integrity, and makes use of identified biological indices (fish, invertebrates, riparian vegetation), as well as other measures for evaluating in-stream and riparian habitats. The biological responses, as indicators of an ecosystem, are recorded in terms of a river health classification scheme that allocates a specific category of health to each river reach. The health categories used by the RHP are simply termed natural, good, fair, and poor.8 This classification system provides an elementary 'front end' to a much more intricate assessment process, and is depicted by colour-coded icons for each of the applied indices (Fig. 1).
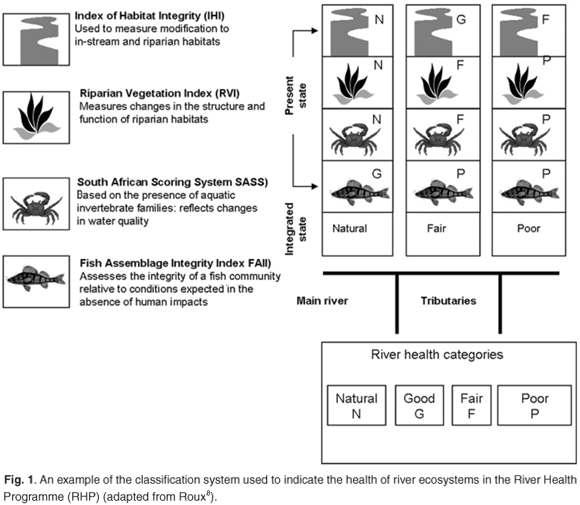
However, these biological responses deal only with indistinct cause–effect relationships between environmental drivers and consequential biological responses. Should the biological integrity indicate the likelihood of unacceptable conditions, then there should be a process to assess these conditions and responses in a critical manner. The biological indices that are applied in the RHP were expanded upon as part of the Ecological Reserve process, and have given rise to the development of the EcoClassification indicators. These measures [e.g. the macroinvertebrate response assessment index (MIRAI) and fish response assessment index (FRAI)] interpret the biophysical components of a river in terms of drivers and biological responses and endpoints in an integrated way, thereby deriving a realistic and reproducible conclusion as to the EcoStatus of the river.9 The EcoStatus in this case can be defined as the totality of the features and characteristics of the river and its riparian areas that bear upon its ability to support an appropriate natural floral and faunal biome.9
The second water quality assessment approach has been to adopt whole effluent toxicity (WET) testing as a tool, to evaluate the acceptability of potentially hazardous effluents for discharge into receiving waters.10 The WET testing approach is an integrative tool that measures the toxic effect of an effluent mixture as a whole, and assesses uncharacterized sources of toxicity and for toxic interactions (such as synergism and antagonism).6 The methods used in WET focus on acute and chronic toxicity testing using standardized laboratory-based bioassays involving laboratory-reared organisms.10 These WET methods have been incorporated into the Direct Estimation of Ecological Effects Potential (DEEEP) toxicity tests,11 the current application of choice within the National Toxicity Monitoring Programme.
Smolders et al.6 proposed that a more ecologically sound approach would be to use the WET concept within an in situ environment to assess in-stream toxicity, rather than conducting laboratory toxicity tests on water samples collected from the environment. Table 1 shows the advantages of using in-stream toxicity testing over the traditional WET approach. The difference between the two approaches is that WET predominantly uses mortality to describe toxicity whereas in-stream toxicity assessment makes use of assessment endpoints that measure biological responses, which in turn can provide a measure of exposure, and sometimes also of toxic effect.6 These biological responses are termed biomarkers and the fundamental assumption upon which they are based centres on some biochemical process (biomarker) being compromised as a result of pollutant exposure.11,12 However, the use of biomarkers in resident organisms is severely constrained by a substantial variability in endpoints, as prolonged pre-exposure to any ambient environmental condition can drastically alter the 'normal' range of a biomarker response. The use of caged organisms has been successful in reducing this degree of variability.13 Active biomonitoring (ABM) is thus conducted within this context.

Active biomonitoring
An ABM approach uses organisms that are collected from a (generally) unstressed, unpolluted population. They are then translocated to selected polluted sites, for instance, along a pollution gradient, or at regular intervals downstream from known pollution sources.14 The chemical and biological consequences of this translocation, which usually involves caging of the organisms, can then be monitored in space and time, to assess the effects of exposure on selected endpoints.6,14,15 The deployment period ranges between 4 and 6 weeks, which allows sufficient time for organisms to recover from any stress incurred as a consequence of the translocation process, as well as a period to react to the environmental conditions that are being monitored.15 The mainstay of the ABM procedure is that transplanted organisms can respond to ambient environmental conditions at an earlier stage and to a greater degree than resident species.
The second approach in which organisms are used to evaluate environmental health is passive biomonitoring (PBM), which involves 'the collection of organisms from their habitats at sites where a natural population exists'.2,6 Although PBM is the most frequently used method in aquatic health assessment, ABM has a number of features that favour its use in aquatic health monitoring.
Smolders et al.6 list the advantages of ABM for biomonitoring as follows:
• There will only be a limited effect of field exposure through, for instance, climatological shock, as the transplanted organisms are adapted to changing environmental conditions, compared to organisms that are cultured in a stable laboratory setting.
• The test exposure period is exactly known in the case of organisms from laboratory sources.
• Laboratory-cultured organisms have a complete record of conditions (usually standardized) under which they were bred and maintained.
• Results from different sites are comparable (constancy of the test organism) even if the test organisms are not normally present at the exposure locations.
• A comparison between transplanted and resident organisms can indicate to what extent the indigenous organisms have adapted to the location. This can also give information about the uptake kinetics of pollutants.
• Resident (site-specific) species can be 'genetically protected' through adaptive change. Use of transplanted organisms can exclude adaptive factors, making the comparison between different sites more valid. The use of non-adapted species can raise the sensitivity of the method.
Although these features favour the use of ABM under field conditions, there are also drawbacks that have to be considered when evaluating and interpreting the results of ABM studies:6
• Food availability at the test site may skew results, either overor underestimating the in-stream toxicity. For example, mussels depend mainly on algae as a food source. Exposure to nutrient-poor but clean water may induce stressful conditions that are unrelated to pollutants, the real reason being inferior quality of food resources.17 On the other hand, enhanced amounts of food in eutrophic streams may mitigate the impact of pollutant exposure on aquatic organisms. Several authors have demonstrated that even though there are adverse effects associated with eutrophication, certain non-negative responses by test organisms may be due to abundant food, actually promoting growth and reproduction of the pollutant-exposed organisms.18
• The loss of cages by theft and vandalism can destroy the value of work done. The problem is widespread in South Africa.
• ABM is regarded to give a worst-case scenario, as prolonged exposure to low levels of contamination can lead to genetic or physiological adaptation of caged organisms. ABM does not take account of adaptation processes of this kind.
Selection of bioindicator organisms for use in ABM
Organisms selected for both ABM and PBM purposes have generally differed from those used in standardized laboratory toxicity tests because of the required assessment outcomes involving vastly differing endpoints. PBM is often selected for higher sensitivity, whereas ABM needs to be able to tolerate and reflect responses to a relatively wide range of pollutant conditions. The choice of the organism will impact on the relevance, success and interpretability of the test results. The organisms should fulfil a number of the following criteria to be recognized as suitable bioindicators, bearing in mind the objectives of the particular transplantation study:19
• They should be representative of the water body or aquatic environment, that is, species indigenous to the region.
• They should be confined in a suitable cage.
• They should be easy to collect, identify and handle.
• They should be relatively long-lived so that longer-term exposure assessment can be carried out.
• They should be of a reasonable size to provide adequate tissue samples for analyses.
• They should be easily cultivated and maintained in the laboratory, as instances may arise where there are no 'unpolluted' species available from a reference site. It would then require the use of indigenous species, cultured under laboratory conditions, for transplantation purposes.
• They should accumulate the pollutant of interest at levels present in the environment without lethal toxic effects and should be relatively tolerant to pollutant exposure.
• They should give a rapid response to an early exposure of various pollutants.
Several bioindicator species indigenous to South Africa have been used in ABM and ABM-related studies. These include indigenous riverine freshwater insect larvae, e.g. Ephemeroptera species, freshwater gastropods, for example: Burnupia stenochoria20 and Melanoides tuberculata,21 crustaceans, e.g. Caradina nilotica,20 and indigenous fish species, e.g. Oreochromis mossambicus.21 Other, unpublished ABM studies conducted throughout South Africa by students from the Ecotoxicology laboratory at the University of Johannesburg, have made use of additional freshwater insect larvae (Chironomus sp.), freshwater molluscs (Corbicula africana, Physa acuta), marine molluscs (Perna perna) and freshwater fish species (Barbus argenteus and Barbus trimaculatus) as bioindicator organisms.
Selection of assessment endpoints in ABM
It is essential to define clearly what is meant by assessment endpoints, as they will be used to define those ecological attributes (that is, different levels of biological organization) that are assessed in an explicit manner and provide a focus for assessments such as ecological risk assessments (ERAs).22 The assessment endpoints used in ABM are aimed at different levels of biological organization and range between sub-organism and ecosystem level (Fig. 2). Owing to the level of complexity in responses and experimental procedure experienced at population level and higher, ABM studies concentrate mainly on sub-organism and organism levels. The ABM approach does not exclude other forms of biomonitoring and is therefore also combined with PBM (e.g. EcoClassification procedures) and traditional laboratory-based WET to provide an integrated and holistic overview on how waste water, runoff and effluents affect receiving water bodies. An assessment of responses at higher levels of biological organization to effluent can be obtained by including PBM. However, this paper focuses primarily on the assessment endpoints applied in ABM.
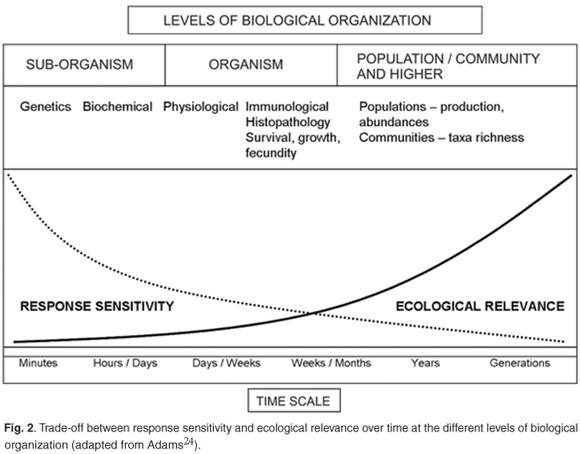
When aquatic organisms are exposed to pollutants, a cascade of biological events takes place, if the concentration is high enough and/or the duration of exposure is sufficiently long to induce adverse effects. The general physiological basis of stress responses of organisms is usually referred to as the General Adaptation Syndrome (GAS).23 In this concept, there are three distinct phases in the stress response: alarm, compensation, and exhaustion. These three phases are placed in an ecological context by identifying three sequences of alteration: 1) primary alterations at a biochemical level, accompanied by, for example, the release of 'stress hormones'; 2) secondary alterations on a physiological level, and 3) tertiary effects on the whole animal level.6 It becomes clear that these three sequences in GAS are a function of exposure duration, external and internal concentration of potentially harmful substances, and biological organization.
Wu et al.13 reviewed the variability in responses and have come to the conclusion that many biomarkers may show adaptation to pollution following long-term exposure to pollution, or may quickly recover in the face of decreasing levels of contaminants in the environment. A generalized representation of biomarker responses is given in Fig. 3. It is essential to have an understanding of the temporal changes of biomarker responses before applying them in environmental management.13 Primary responses are rapid and reversible at a (intra)cellular biochemical level. Secondary responses are generally physiological changes that take more time to occur in organisms, and they can also be reversible. Tertiary responses (at both individual and population level) are the least reversible, occur at the highest level of biological organization, and have the longest-lasting effect.6,24

A number of different methods have been proposed to select the most suitable biomarkers for environmental monitoring. To obtain a holistic and integrative overview of how instream effluent exposure affects bioindicator organization, it has been proposed that a link be established between different levels of biological organization within the same organism.6 The reasoning is that a unit with higher ecological relevance is attained with an increase from one hierarchical level of biological organization to the next (e.g. from sub-organism to organism). However, responses at lower levels of biological organization can usually be detected much earlier and could ultimately affect higher levels. Thus, to extrapolate between different levels of organization, a common parameter has to be found that allows for the linking between the different levels. Energy budgets have been proposed as one such parameter, not only because these budgets can be determined at different levels of biological organization, but also because they can provide a causal relationship between the different levels, potentially relating cellular effects to reproduction.6,24
A number of approaches have been applied to analyse large biomarker data sets. These data sets often consist of biomarker responses covering all the levels of biological organization in a vast array of bioindicator organisms representing different trophic levels. The most promising application involves multivariate statistical techniques, which also identify those biomarkers that are responsible for clearly distinguishing contaminated sites from clean sites.21,25 The biomarkers selected for implementation in this particular study on the Rietvlei wetland system have been based on the most commonly used suites of markers that are currently used in this field of study.12,13
Generalized ABM deployment protocol
Bioindicator organisms may be collected from a variety of reference (clean or non-contaminated) sources. These sources may include laboratory or hatchery-reared organisms,20,21 from reference sites within the catchment of the system to be monitored20 or even from a non-impacted site within the same system. A sufficient number of bioindicator organisms are then deployed, depending on the experimental design (e.g. 10 organisms in duplicate or triplicate exposure cages) at the selected sampling sites in polyethylene cages, with polyethylene mesh screens that allow for free water movement through the cage. The cages are firmly anchored at the sites to prevent their displacement. Food is initially placed in the cage so that the bioindicator organisms undergo minimized stress from lack of feeding during the first week of exposure. This is only applicable to bioindicators that do not filter feed.
The period during which the bioindicators are exposed to the ambient conditions at the sampling site will depend on the specific organism—for instance, the exposure period for bivalves and fish can be up to eight weeks, whereas for aquatic insect larvae up to one week. The frequency at which cages may be checked depends on several factors such as species used, type of pollutant, and rate of biological response. Samples can be collected during this period for biomarker analyses and verification of food availability. Supplementary food is given where there is inadequate algal growth, e.g. for the grazing group of bioindicators. Water and/or sediment samples are taken for physico-chemical analyses. This is repeated for the remainder of the exposure period.
Case study illustrating the application of ABM in South Africa: industrial and urban run-off in a peri-urban river
An urban nature reserve near Pretoria, which covers an area of about 3500 ha, arose from what was named the Rietvlei Water Scheme. A dam within the reserve and its environs provide about 27% of the city's water requirement. The catchment area receives effluent from industries, agricultural activity, informal settlements and municipal sewage treatment plants. Increased housing and industrial developments are considered to be the biggest threats to the water quality of this reserve.26 A study of the dam indicated that there are endocrine-disrupting pollutants present. This was not confirmed as present in the streams leading to and flowing from the dam.26 A biological assessment of the stream that flows into the dam was carried out using standardized PBM protocols. This assessment led to the conclusion that the health of this riverine system is in a severely degraded condition.27 No toxicity was observed when WET testing (using fish, daphnid and algal growth inhibition tests) was conducted under laboratory conditions on water from the stream above and below the dam.21 The water quality parameters under scrutiny were not deemed in terms of the test to be of such a poor quality that it would lead to mortalities of aquatic organisms. The poor health-status of the riverine area was then ascribed, for the most part, to habitat destruction, subject to any additional test indicating otherwise.
Wepener et al.21 initiated an ABM study of this system. Summarized findings are reported here. An aquatic mollusc species (M. tuberculata) and fish species (O. mossambicus) were used at three locations on stream flowing through the reserve and retrieved after a 6-week exposure period. Effects of field exposure were determined using a range of cellular and biochemical biomarkers that are both indicators of exposure and effect [that is, DNA damage, heat shock protein (HSP 70), metallothionein (MT), acetylcholine esterase (AChE), lactate dehydrogenase, catalase (CAT), cellular energy allocation (CEA) and ethoxyresorufin-o-diethylase activity (EROD)].20 The results clearly indicate that although the conventional mortality-based whole-effluent toxicity testing did not reveal toxicity, all the biomarker responses showed changes from the norm, indicating that the in situ exposed organisms were stressed. A multivariate statistical approach provided an interpretation of the biomarker responses, and highlighted sites at which more detailed analysis of pollution would be useful. Based on the contribution of the individual biomarker results towards the distinct groupings, we concluded that the site upstream of the impoundment complexes in the nature reserve receives organic pollutants, whereas the sites directly upstream and directly downstream of the main dam undergo the effects of a combination of metallic and organic pollutant stressors. The specific stressors and their concentrations in the two dams have been confirmed by independent chemical analyses that were undertaken during a separate but associated research project on the endocrine disruptive activity in this system.28 These two forms of stressors result in anti-oxidant and genotoxic symptoms.
Two important aspects were highlighted in this study and their effect on the use of ABM. The first is that the two bioindicator organisms used gave different biomarker responses. These differences were in terms of the intensity of response from differences in exposure routes and trophic levels. Nevertheless, the general nature of the response is common, both species showing the same site-specific trend described. These results support the findings of Wu et al.13 showing that there is no bioindicator species group that is notably more sensitive than another. All groups (whether crustaceans, molluscs or fish) show similar patterns of induction and recovery. We have conducted the same biomarker analyses on the test organisms that were subjected to the traditional laboratory-based WET bioassays (author's unpublished data). Although the objective of the WET bioassay was to monitor lethality (but not shown to be evident), the bioindicators gave the same response trends as was found in the in situ ABM bioindicators. The repeatability of this finding is currently being assessed through further work.
Application in integrated water resources management
The contemporary approach to water resource management recognizes that legislative procedures cannot focus on chemical criteria alone, as they are inadequate for the protection of ecosystems that are exposed to multiple impacts.29 The 'weight of evidence' characteristics that biomarker results provide make this form of biomonitoring ideal within a risk assessment scenario.30 The general application of standard chemical quality guidelines (e.g. in the case of South Africa the water quality guidelines for the aquatic environment) becomes problematic as they do not take into consideration site-specific chemical and biological tolerances.12 Internationally, there is a greater awareness of the development of cohesive methods of determining environmental quality by considering biological, ecological, physico-chemical and hydrological criteria.25 Implicit in the risk-based monitoring approach that is currently being practised in South Africa is the requirement to classify and protect the 'ecological health' of rivers and streams in a transparent and integrated manner.
The question that arises is where ABM fits into the integrated water resources management (IWRM) framework of South Africa. The ABM approach seems to be the appropriate choice when relevant toxicity test methods have been selected for the National Toxicity Monitoring Programme (NTMP) (surface water status and trends monitoring) of the Department of Water Affairs and Forestry. The choices relating to toxicity testing were made at a workshop attended by local aquatic toxicologists, ecotoxicologists and water resources managers who would be responsible for implementing the NTMP. Notwithstanding the advantages, the resolution was to defer the ABM approach. The constraints identified by the workshop participants were vandalism of monitoring equipment (a problem that exists internationally); choice of organism and biomarker; what can be done during 'no-water' months?; the lack of clear links to the ecological relevance of the ensuing results; the limited number of laboratories that are able to carry out biomarker analyses; and the capacity within the water resource management structures to interpret biomarker results. A conservative approach was followed by selecting the following laboratory tests: semi-static zebrafish development test; the daphnid (Daphnia pulex) reproduction test; the 24-well microplate algal growth inhibition test; and a recombinant yeast test for oestrogenicity.31 The drawback here remains that these protocols are laboratory-based and may not take site-specific factors into account.
ABM within the National Water Act (NWA, No. 36 of 1998) has, however, been identified as having application value within the context of water resource management, such as the 'Classification and Resource Quality Objectives' and 'Monitoring Ecosystem Health' requirements stipulated by the act. The aforementioned contexts in the act do not specifically require legally defensible test methods at this stage31 as is required for the standard toxicity tests that are used for effluent discharge permitting processes. It therefore stands to reason that these NWA contexts could serve as an ideal platform to evaluate the as yet untested ABM protocols within South African IWRM.
Although ABM has the potential for being an excellent tool to monitor and therefore manage ambient water quality, the approach is still largely used for research purposes. In cases where it is applied on a routine basis, the emphasis is on data compilation and not on management and control of pollutants (that is, as the chemical criteria or guideline value may be applied), primarily due to its not forming part of any regulatory requirement. Given the wealth of information validating biomarker responses both in the laboratory and the field, the application of biomarker and ABM within a regulatory framework is long overdue.12
Handy et al.12 have identified a number of important steps that need to be followed within the European regulatory environment to bridge the divide that exists between research and development, and the policy-management framework. These are:
• agreement between participating member states on how biomarkers should be incorporated into a regulatory framework;
• the development of standard operating procedures (SOPs);
• inter-laboratory testing and validation of SOPs in the same manner that has been done to validate acute toxicity protocols; and
• the modification of legislation to include biomarkers as one of the environmental monitoring tools. We are in a fortunate position in South Africa as we have the provision within the NWA to develop and adopt innovative monitoring techniques such as ABM and biomarkers within the aquatic health assessment framework.
Until such time as internationally acceptable and validated SOPs exist, ABM can be applied within the current South African IWRM context through the ERA framework. Jooste and Claassen32 indicate that the nature of the ERA framework addresses the requirements of the NWA, and takes into account the inherent characteristics of aquatic ecosystems and the diverse nature of stressors affecting them. By integrating ABM with other existing monitoring protocols through a multi-tiered guidance framework, it should be possible to define whether an effect is present, whether it is related to a particular activity and whether the organism's responses are characteristic of a particular stressor agent (Fig. 4). The multi-tiered framework follows the same approach as the Environmental Effects Monitoring framework that was developed for the Canadian pulp and paper and metal mining industries.33 The framework consists of multiple tiers of progressive assessment from the identification of a problem (Tier 1) through to identification of the specific pollutant species responsible for the observed effects.
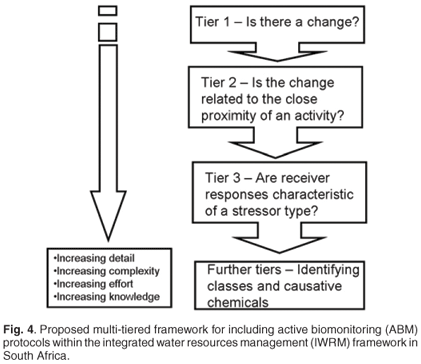
Tier 1 identifies whether a potential quality-related problem or even quality-related effects exist in the receiving water body and is based on, and uses, existing national monitoring programmes such as the Chemical Monitoring Programme (in the case of water quality issues) or RHP (in the case of aquatic health-related issues). If no effects are detectable (i.e. the monitoring programme identifies whether a problem is evident or not) then the routine monitoring frequency will be adhered to, without initiating any of the following tiers. The second tier (Tier 2) comprises the traditional mortality-based protocols and bioassay (that is, NTMP protocols); testing that results in fail or pass. This identifies whether, for instance, in the case of the RHP, the ecological effects observed during Tier 1 are related to a specific activity. Based on these results, the Tier 3 assessment would be conducted to determine whether the aquatic ecosystem response patterns are characteristic of a particular type of stressor. These first three tiers are focused on the receiving water body, that is, in the mixing zones beyond any 'end-of-pipeline' conditions. Further tiered investigations could be carried out within the boundaries of the activity to identify the particular contributor responsible for the stressors, characterizing the chemical classes and identifying the specific chemicals responsible for the responses. Thus, within the existing South African water resources monitoring framework, biomarkers and ABM could potentially be of value within Tier 3 and the later tier structures, which could be related to water licensing applications. Within the current South African monitoring structures, Tier 1 would constitute routine chemical and biological monitoring through the existing monitoring programmes (i.e. Chemical Monitoring Programme and RHP), and Tier 2 would be represented by more specialized monitoring programmes (e.g. NTMP). Where these two tiers are inconclusive in identifying aquatic health impacts in a particular water body, the ABM protocol of Tier 3 can be applied.
The case study that has been presented demonstrates that ABM and biomarkers can be used successfully to indicate sub-lethal exposure and effect responses to low levels of toxicants when the traditional approaches of PBM and mortality-based bioassays do not reveal any specific stressor-based responses. A total of 27 different biomarkers was assessed during the course of the case studies in this project, and other ABM studies conducted by the Centre for Aquatic Research at the University of Johannesburg. It now remains to select the most appropriate suite of biomarkers that could be applied in ABM studies on a wider scale and form part of routine monitoring initiatives in a similar manner to the ECOMAN programme in the United Kingdom.30
The research projects from which ABM-based data were generated were primarily funded by the National Research Foundation, THRIP and the Water Research Fund of Southern Africa. I wish to thank the following postgraduate students for their contributions to the formulation of these ideas: Gordon O'Brien, Martin Ferreira, Wynand Malherbe, Zvegerei Mbizi, Sibonani Mlambo, Fungayai Chatiza, Kerry Mills, Kristin Schüring and Sabine Schwientek.
1. Train R.E. (1979). Quality Criteria for Water. Castle House Publishers, London. [ Links ]
2. Chapman P.M. (1995). Ecotoxicology and pollution—Key issues. Mar. Pollut. Bull. 31, 167–177. [ Links ]
3. Chapman P.M. (2002). Integrating toxicology and ecology: putting the 'eco' into ecotoxicology. Mar. Pollut. Bull. 44, 7–15. [ Links ]
4. Sarakinos H.C., Bermingham N. and White P.A. (2000). Correspondence between whole effluent toxicity and the presence of priority substances in complex industrial effluents. Environ. Toxicol. Chem. 19, 63–71. [ Links ]
5. Smolders R., De Coen W. and Blust R. (2004). Ecologically relevant exposure assessment for a polluted river using an integrated multivariate PLS approach. Environ. Pollut. 132, 245–263. [ Links ]
6. Smolders R., Bervoets L., Wepener V. and Blust R. (2003). A conceptual framework for using mussels as biomonitors in whole effluent toxicity. Hum. Ecol. Risk Assess. 9, 741–760. [ Links ]
7. Calow P. (2000). Critics of ecosystem health misrepresented. Ecosystem Health 6(1), 3–4. [ Links ]
8. Roux D.J. (2004). From monitoring design to operational program: facilitating the transition under resource-limited conditions. In Environmental Monitoring, ed. G.B. Wiersma, pp. 631–647. CRC Press, Boca Raton. [ Links ]
9. Kleynhans C.J., Louw M.D., Thirion C., Rossouw N.J. and Rowntree K. (2005). River EcoClassification: Manual for EcoStatus determination (Version 1). Joint Water Research Commission and Department of Water Affairs and Forestry report. WRC Report No. KV 168/05, Department of Water Affairs and Forestry, Pretoria. [ Links ]
10. Slabbert J.L., Oosthuizen J., Venter E.A., Hill E., Du Preez M. and Pretorius P.J. (1998). Development of procedures to assess whole effluent toxicity. WRC Report No. 453/1/98, Water Research Commission, Pretoria. [ Links ]
11. Slabbert L. (2004). Methods for Direct Estimation of Ecological Effect Potential (DEEEP). WRC Report 1313/01/04, Water Research Commission, Pretoria. [ Links ]
12. Handy R.D., Galloway T.S. and Depledge, M.H. (2003). A proposal for the use of biomarkers for the assessment of chronic pollution and regulatory toxicology. Ecotoxicology 12, 331–343. [ Links ]
13. Wu R.S.S., Sui W.H.L. and Shin P.K.S. (2005). Induction, adaptation and recovery of biological responses: Implications for environmental monitoring. Mar. Pollut. Bull. 51, 623–634. [ Links ]
14. De Coen W., Robbens J. and Jansen C. (2005). Ecological impact assessment of metallurgic effluents using in situ biomarker assays. Environ. Pollut. 141(2), 283–294. [ Links ]
15. Smolders R., Voets J., Bervoets L., Blust R. and Wepener V. (2005). Active biomonitoring (ABM) by translocation of bivalve molluscs. In Water Encylcopedia Vol. 2, Water Quality and Resource Development, eds J.H. Lehr and J. Keeley, pp. 33–37. John Wiley, New York. [ Links ]
16. Bervoets L., Voets J., Chu S., Covaci A., Schepens P. and Blust R. (2003). Comparison of accumulation of micropollutants between indigenous and transplanted zebra mussels (Dreissena polymorpha). Environ. Toxicol. Chem. 23(8), 1973–1983. [ Links ]
17. Borcherding J. (1995). Laboratory experiments on the influence of food availability, temperature, photoperiod and gonad development in the freshwater mussel Dreissena polymorpha. Malacologia 36(1-2), 15–27. [ Links ]
18. Stuijfzand S.C., Helms M., Kraak M.H.S. and Admiraal W. (2000). Interacting effects of toxicants and organic matter on the midge Chironomus riparius in polluted river water. Ecotox. Environ. Safe. 46(3), 351–356. [ Links ]
19. Forbes V.E. and Forbes T.L. (1994). Ecotoxicology in Theory and Practice. Chapman & Hall, London. [ Links ]
20. Palmer C.G., Muller W.J., Gordon A.K., Scherman P-A., Davies-Coleman H.D., Pakhomova L. and De Kok E. (2004). The development of a toxicity database using freshwater macroinvertebrates, and its application to the protection of South African water resources. S. Afr. J. Sci. 100, 643–664. [ Links ]
21. Wepener V., Van Vuren J.H.J., Chatiza F.P., Mbizi Z., Slabbert L. and Masola B. (2005). Active biomonitoring in freshwater environments: early warning signals from biomarkers in assessing biological effects of diffuse sources of pollutants. Phys. Chem. Earth 30, 751–761. [ Links ]
22. Suter II G.W., Norton S.B. and Fairbrother A. (2005). Individuals versus organisms versus populations in the definition of ecological assessment endpoints. Int. Environ. Assess. Manag. 1, 397–400. [ Links ]
23. Selye H. (1950). Stress and the general adaptation syndrome. Br. Med. J. 1, 1384–1392. [ Links ]
24. Adams S.M. (2001). The use of biomarkers in ecological risk assessment: recommendations from the Christchurch conference on Biomarkers in Ecotoxicology. Biomarkers 6(1), 33–44. [ Links ]
25. Smolders R., Bervoets L. and Blust R. (2002). Transplanted zebra mussels (Dreissena polymorpha) as active biomonitors in an effluent-dominated river. Environ. Toxicol. Chem. 21(9), 1889–1896. [ Links ]
26. Barnhoorn E.J., Bornman M.S., Pieterse G.M. and Van Vuren J.H.J. (2004). Histological evidence of intersex in feral sharptooth catfish (Clarias gariepinus) from an estrogen polluted water source in Gauteng, South Africa. Environ. Toxicol. 19, 603–609. [ Links ]
27. River Health Programme (RHP) (2005). State of the Rivers Report: Monitoring and managing the ecological state of rivers in the Crocodile (West) Marico water management area. State of the Rivers Report No. 9, Department of Environmental Affairs and Tourism, Pretoria. [ Links ]
28. Bornman M.S., Van Vuren H.J., Bouwman H., De Jager C., Genthe B. and Barnhoorn E.J. (2007). The use of sentinel species to determine the endocrine disruptive activity in an urban nature reserve. WRC Report 1505/1/047, Water Research Commission, Pretoria. [ Links ]
29. Blöch H. (1999). The European Union water framework directive: Taking European water policy into the next millennium. Water Sci.Technol. 40, 67–71. [ Links ]
30. Galloway T.S., Brown R.J., Browne M.A., Dissanayake A., Lowe D., Depledge M.H. and Jones M.B. (2006). The ECOMAN project: a novel approach to defining sustainable ecosystem function. Mar. Pollut. Bull. 53(1-4), 186–194. [ Links ]
31. Slabbert J.L. and Murray K. (2006). Guidelines for the selection of toxicity tests in support of the information requirements of the National Water Act (No. 36 of 1998). WRC Report No. K5/1211/1/05, Water Research Commission, Pretoria. [ Links ]
32. Jooste S. and Claassens M. (2001). Rationale for an ecological risk approach for South African water resource management. Water SA 27(3), 283–292. [ Links ]
33. Hewitt L.M., Dubé M.G., Culp J.M., MacLatchy D.L. and Munkittrick K.R. (2003). A proposed framework for the investigation of cause for environmental effects monitoring. Hum. Ecol. Risk Assess. 9, 195–211. [ Links ]
Received 11 April 2007. Accepted 3 September 2008.














