Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Science
versão On-line ISSN 1996-7489
versão impressa ISSN 0038-2353
S. Afr. j. sci. vol.104 no.9-10 Pretoria Set./Out. 2008
SCIENCE POLICY
South Africa's bioprospecting, access and benefit-sharing legislation: current realities, future complications, and a proposed alternative
Neil R. CrouchI, II, *; Errol DouwesI, III; Maureen M. WolfsonIV; Gideon F. SmithIV, V; Trevor J. EdwardsIII
IEthnobotany Unit, South African National Biodiversity Institute, P.O. Box 52099, Berea Road 4007, South Africa
IISchool of Chemistry, University of KwaZulu-Natal, Durban 4041, South Africa
IIISchool of Biological and Conservation Sciences, University of KwaZulu-Natal, Private Bag X01, Scottsville 3209, South Africa
IVResearch and Scientific Services, South African National Biodiversity Institute, Private Bag X101, Pretoria 0001, South Africa
VSchweickerdt Herbarium, Department of Botany, University of Pretoria, Pretoria 0002
ABSTRACT
Globally, many nations are legislating access for bioprospecting purposes to their biological and genetic resources. South Africa, as a megadiverse country, has recently regulated bioprospecting, access and benefit-sharing activities in accordance with its obligations as a ratifying party to the Convention on Biological Diversity. The context and process of key legislation developments in South Africa are discussed, prior to our presenting a critique which emphasizes the practical impacts, especially on drug discovery, arising from the newly introduced systems. Probable effects on existing bioresource-based industries within South Africa, together with current as well as future bioprospecting activities, are assessed. Several practicalities of bioprospecting methods have been poorly accommodated, resulting in the development of impracticable and unnecessarily restrictive regulations. We conclude that though well-intentioned, these non-facilitative regulations have placed a dead hand on value-addition to South Africa's biodiversity. Bioprospectors will find it difficult to continue with broad-scale screening programmes given their user insecurity, legal uncertainty, and cost-inefficiency. Existing bioresource-based industries within South Africa face potential closure in view of onerous bioprospecting permit application requirements. An alternative, practical, CBD-compliant model on which to base urgently required legislative reforms is presented.
Introduction
South Africa, considered one of 17 megadiverse countries (nations that collectively account for 70% of global biodiversity),1 holds the Cape Floristic Region (CFR),2,3 one of the six most significant concentrations of plant diversity in the world, being the smallest but most diverse floristic kingdom known. With over 19 500 indigenous plant species in about 350 plant families, South Africa indeed has the richest temperate flora in the world. In addition, three global hotspots of biodiversity are currently recognized within the country: the CFR, the Succulent Karoo, and Maputaland–Pondoland–Albany.4 Regional biotic elements other than plants are similarly diverse, exemplified by the fungi which have recently been estimated conservatively at 200 000 species5 and coastline marine life at over 10 000 taxa.6 The rich floristic and geological diversity7 of the country supports immensely rich and diverse faunas, both invertebrate and vertebrate.6 Although the potential for new commercial product development from South African bioresources is high, such that South Africa has been, and remains, a country of significant interest to bioprospectors, it may never prove to be the land of 'green gold'. Especially with respect to the quest for new pharmaco-active materials, it should be remembered that behind every commercially successful bioproduct lies substantial expenditure in the form of extensive screening programmes that will have discarded literally thousands of commercially non-exploitable candidates before discovering a single commercially exploitable product.
Historically, a lack of bioprospecting legislation and associated regulations has permitted almost unconstrained access to South African bioresources, with materials being harvested, sometimes in destructively excessive quantities, and being exported to research and development nodes abroad, for innovative value addition, and off-shore financial benefit. The consequence has been that the country as a whole, including traditional knowledge (TK)-holding communities and bioresource providers, have not benefited equitably from the commercial and other gains derived from local bioresource commercialization. Records, fifteen years ago, reveal that the Dutch flower industry earned almost R300 million annually from the sale of freesias alone.7 The genus Freesia is near-endemic to South Africa. Similarly, large areas are under cultivation, with Gladiolus, Zantedeschia and Nerine plants in Holland and New Zealand.8 Revenue generated from sales of Pelargonium cultivars, derived from South African species and protected by Plant Breeders' Rights in the Netherlands, Germany and Belgium approximates US$6 billion annually, with no associated revenue returns to South Africa.8
Several notable industries have developed within South Africa,9 and have already brought direct and indirect benefits to its citizens: these include the cut flower [primarily fynbos species (proteas, leucospermums and the like)],10 aloe (principally Aloe ferox Mill.),11,12 marula (Sclerocarya birrea (A. Rich.) Hochst. subsp. caffra (Sond.) Kokwaro), honeybush tea (Cyclopia spp.), and rooibos tea (Aspalathus linearis (Burm. f.) R. Dahlgren)13 industries, amongst others. Recently introduced bioprospecting legislation14 and regulations15 in South Africa have sought to redress disparities in the sharing of benefits derived from bioprospecting. They reflect i) national sovereignty over access to bioresources, ii) a recognition of traditional knowledge and related intellectual property rights (IPR), iii) a need to share benefits equitably with stakeholders, and iv) a need to use bioresources sustainably. Each of these elements serves to align national law with the objectives of the Convention on Biological Diversity (CBD).16 Broadly speaking, this approach to developing access and benefit-sharing (ABS) legislation is supported increasingly by countries both rich and poor in bioresources. What is critical is that the implementation of such regulations should lead to a positive outcome. An emerging economy such as South Africa can ill afford to restrain its own bioprospecting activities through unreasonable and cost- prohibitive regulations. One threatened programme is the Novel Drug Development Platform (NDDP) (www.sahealthinfo.org/noveldrug/), a broad-based consortium of South African researchers from clinical and scientific disciplines based in local parastatals, universities and science councils. The objective of NDDP research has been to develop, from ethnomedicinal plants, novel drugs that are effective against serious and comparatively neglected diseases of the developing world: malaria, tuberculosis and diabetes mellitus. The Department of Science and Technology (DST), through the agency of the National Research Foundation (NRF), has provided Innovation Funds to support the activities of the NDDP.
A critique of recent South African legislation and ensuant regulations governing access to biological resources for bioprospecting purposes, and benefit-sharing, is the focus of this paper. This review does not question the stated intentions of the legislators. Our emphasis is placed on assessing the practicalities of these newly introduced systems, and we discuss the impact that these are anticipated to have on existing bioprospecting activities and bioresource-based industries both within and outside South Africa, now, and in time to come. Particular emphasis is placed on consequences for drug development. The implications of this legislation for academic research (expanding the knowledge base of science without a primary commercial interest) on South African biodiversity, and the conservation thereof, form the subject of a separate review.
Bioprospecting in South Africa: historical context
Bioprospecting involving traditional knowledge
High costs associated with laboratory assays, drug trials and subsequent registrations have largely limited bioprospecting for new drugs within South Africa to collaborative efforts between researchers based at science councils, parastatals and universities. Such ventures have necessarily applied focused selection procedures to identify the most promising subjects prior to initial screening programmes. This improves cost-effectiveness. These projects focus predominantly on ethnomedicinal knowledge, as it is widely acknowledged that ethno-directed approaches provide advantages in the search for novel drugs.17–21 There is a wealth of documented traditional ethnomedicinal knowledge in South Africa, particularly relating to plant use.22 That value is indeed inherent in this local knowledge base is exemplified by the study of Fourie and co-workers,23 who reported that approximately 81% of 300 evaluated medicinal plants show biological activity in a range of target assays. Similarly, considerable success has been achieved when an ethno-directed approach is adopted for the search for new antimalarial drugs from South African plants. Extracts of 49% of all the species assayed exhibited promising antiplasmodial activity (IC50 <10 µg/ml).24 These regional experiences substantiate the notion that bioresources used within local traditional medicine systems constitute good starting points for drug development research. However, the acceptance that such selections are starting points only, and not end products of a drug development pipeline, has fundamental implications for interpreting the value of i) the bioresource, and ii) any traditional knowledge associated with that resource which may have led to its selection for investigation in the first instance. The same may be stated of non-medicinal bioprospecting activities for, for example, foods, beverages, fibres, and horticultural cultivars, all of which require similarly innovative and costly research and development over extended periods. The notion that bioresources associated with traditional knowledge are for the most part commercializable entities, simply awaiting 'cherry picking' by industry, is false. In reality, only a small minority of initial research subjects yield commercial or industrial products.25 Market forces, toxicity and efficacy standards, production, processing and formulation challenges, and the sustainable use/supply of bioresources are among numerous factors that ultimately limit the industrialization or commercialization of any concept product. For CBD-ratifying countries, which include South Africa, further obligatory factors apply (Article 8j), namely, the approval and involvement of the holders of related traditional knowledge and the equitable sharing of benefits arising from the wider application of such knowledge.16
Intellectual property rights
Since the signing of the CBD,16 there has been contention26–28 over how countries should secure returns from intellectual property rights to ensure equitable sharing of benefits derived from the utilization of natural resources and their associated indigenous knowledge. It is recognized that cultural groups who contribute knowledge regarding the use of bioresources should benefit where such knowledge (intellectual property)29 is the basis of successful new product developments.30 The difficult issues of determining traditional knowledge ownership may have given rise to the reported reduction in bioprospecting activities at several large pharmaceutical companies.31 Risks faced by companies include financial losses, legal conflicts over IPR ownership, and negative publicity linked to perceived biopiracy. The historic absence of formalized means of sharing economic benefits derived from the commercial exploitation of biodiversity with landowners and/or custodians of traditional knowledge has led to accusations that businesses are involved in biopiracy.32–35 However, it has been argued that many cases of biopiracy have been de facto consequences of clumsy permit systems that are too costly, time-consuming or impracticable to implement.35 Along with several other factors, poor legislation governing the use of natural resources and traditional knowledge has been cited as a limiting factor in the global advancement of bioprospecting.36–38
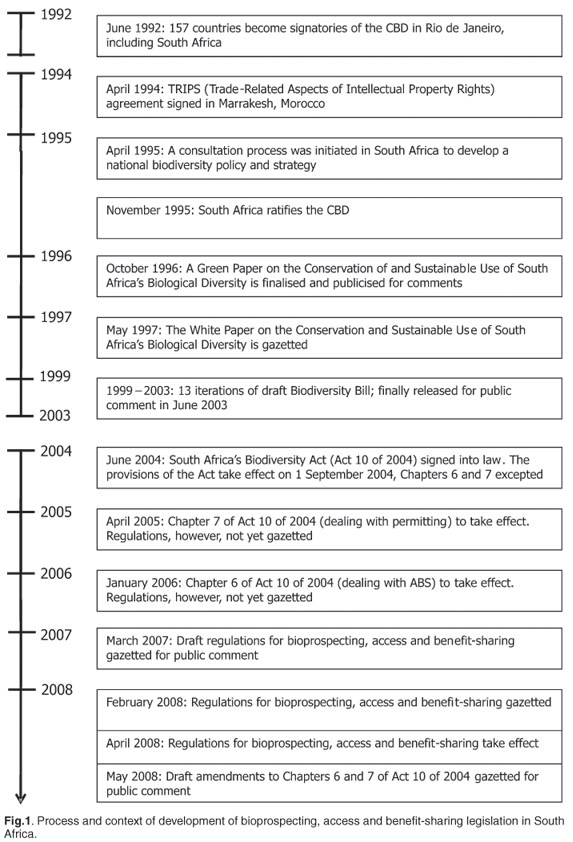
Bioprospecting industries within South Africa have generally not aligned with best practice, or shared equitably their financial profits with TK-holding communities from November 1995 to April 2008, this being the period between ratification of the CBD by South Africa and the implementation of regulations relating to benefit-sharing (Fig. 1). Doubtless this relates in part to problems in identifying the true holders or owners of the TK. Indirect benefits (such as improved nutrition and health, job creation, and infrastructural development through taxation) have nonetheless in the interim been distributed broadly. While IPR returns may accordingly facilitate the distribution of commercial benefits to TK-holding communities, a universally accepted means of implementing intellectual property recognition is yet to be formulated. Certain provisions made in the 1995 World Trade Organisation (WTO) Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS)39 were regarded to conflict with proposals made by the CBD.40 The convention has been seen to enshrine equity, and importantly, accessibility to bioresources. In particular, the CBD acknowledges the collective rights of indigenous and local communities to exchange and develop biodiversity. In contrast, TRIPS strongly favours private ownership of IPR and profit-based systems.39,40 The absence of legislation protecting private ownership of IPR in developing countries was reported to be costing industrialized countries some US$200 billion in lost royalties per annum.41 TRIPS attempted to narrow the gaps in the ways these rights were protected globally, incorporating them into common international rules. The disparity between TRIPS and the CBD objectives has led to much debate, particularly when WTO members have been faced with possible trade sanctions, if they declined endorsement of the TRIPS agreement. This agreement did, however, acknowledge the right of countries to decide on the details of their own patent systems40 and have been accordingly advised to exclude all life forms and related knowledge from patentability, as was permitted under the WTO.42 South Africa's Department of Trade and Industry (DTI) has subsequently published the Patents Amendment Act of 2005,43 which requires an applicant for a patent to furnish information relating to any role played by an indigenous biological resource, or traditional knowledge, that may form part of the submitted invention and, if so, provide proof that applicant(s) have received permission to make use of the indigenous resource or traditional knowledge or traditional use. Furthermore, there has been increased international support for an amendment to the WTO agreement on TRIPS that would require the disclosure of origin of genetic resources and traditional knowledge. Presently, this amendment is supported by nearly 80 of the total WTO membership of 151.44
Genetic and biological property rights
A new system of property and commercial transactions has been created under the CBD for genetic resources. The assignment of property rights is based on the need to exercise physical control over the property, which in this case is genetic information.45 South African legislation —like that of many other countries—does not create a property rights system for this component, but relies on the property rights relating to the physical aspect, such as the biological resource or its parts (such as seeds) to define the legal status of their genetic and biochemical resource.45 There may be a basis in the case of genetic resources for distinction between the rights over the physical entity (physical property) and over the genetic information that the resources contain (intangible property), which represents the real value of the resources, and where the judicial problems are especially complex.46 In some countries, notably Ethiopia, the state is considered the owner of the genetic resources, although the owner of the land, who is deemed to be the owner of the biological resources, has to be contacted to obtain permission to collect the biological resource.47 In South Africa, legislation14 does not vest ownership of genetic resources in the state, unless they occur on state land.47 The landowner in South Africa, in terms of common law, owns both the biological and the genetic resources on or under his/her property.
Not all ownership cases are clear-cut, particularly with respect to communal land ownership, and related culture-based concepts of control. To avoid undermining resource user certainty, especially when South African bioresources are exported for the purpose of bioprospecting, it is essential that users and providers share a common understanding of the exact nature of the rights that are being granted to the user by the ABS agreement. Accordingly, there is a need to determine and legally acknowledge the differences between the owners of the land from which the specimen is collected, the owner of the biological resource, and the owner of the genetic resource. This problem is exacerbated when one is dealing with trans-boundary biological and genetic resources where different property regimes exist in different countries. To maximize certainty, users will naturally tend to seek out resource owners whose ownership status is most straightforward, resulting in a marginalization of communal landowners.
Key legislative developments
White paper
The process and context of the development in South Africa of legislation and regulations on bioprospecting, access and benefit-sharing is summarized in Fig. 1, highlighting key events. A white paper48 on the Conservation and Sustainable Use of South Africa's Biological Diversity was gazetted in May 1997, with subsequent minor modifications by Cabinet, and adopted by Parliament.49 This policy48 outlined the necessity for establishing legislation and institutional structures to control access to South Africa's indigenous genetic resources. In addition, the proposed legislation was to ensure that benefits arising from South African bioresources served the nation. Significantly, the white paper recognized that it was in the country's best interest to ensure that access to biodiversity was not unnecessarily restrictive, and further recommended that legislative conditions should stimulate economic activity. Our critique considers whether these key policy considerations have been successfully achieved, given that both legislation (Act 10 of 2004, hereafter referred to as the Act)14 and Regulations15 have subsequently been ratified. Relatively few parties to the CBD have reached this commendable goal, with fewer than 10% of the CBD parties having adopted access and benefit-sharing legislation, ten years after the emergence of the convention. Virtually none has claimed that their ABS arrangements function effectively.50
The National Environmental Management Biodiversity Act (NEMBA)
NEMBA and the CBD
The draft Biodiversity Bill, formulated by the Department of Environmental Affairs and Tourism (DEAT), evolved through 13 iterations prior to its release for public comment on 1 June 2003. Following inputs received and subsequent amendments made, the Act14 was gazetted in early June 2004 (Fig. 1). Besides regulating bioprospecting, access and benefit-sharing activities involving indigenous biological resources within South Africa, the Act (Ch. 6) frames Regulations relating to the export from South Africa of indigenous biological resources for the purpose of bioprospecting or any other kind of research. The implementation of various chapters in the Act has been staggered to allow for appropriate regulations to be developed by the DEAT (Fig. 1).
The Act has sought to give effect to ratified international agreements (for instance, CBD, CITES), binding on the country, which relate to biodiversity. It legislates for, in part, i) the management and conservation of biological diversity within the country and of the components of such biological diversity, ii) the use of indigenous biological resources in a sustainable manner, and iii) the fair and equitable sharing among stakeholders of benefits arising from bioprospecting involving indigenous biological resources, coinciding closely with the cornerstones of the CBD. The convention refers to access to genetic resources, but does not define either access or use, implying that it is possible to access biological resources for commercial exploitation, without access to the genetic resources in the material. Accordingly, it is not clear in the CBD to what extent the use of the whole medicinal plant, herbal plants or nutraceutical food is regarded as access. In South Africa, however, access legislation refers to biological resources which, by definition, also includes the genetic resources.14 The definition of bioprospecting qualifies the activities, basing the distinction on the intended use by the applicant at the time of access, that is, for bioprospecting or for research other than bioprospecting. The Regulations develop this further, especially within the exemptions included in them.15
Consideration of definitions
Central to our treatise on the practicalities of newly implemented systems are definitions provided by the Act14 of 'bioprospecting' [Section 1(1)] and 'indigenous biological resources' [Sections 1(1); 80(2)]:
'bioprospecting', in relation to indigenous biological resources, means any research on, or development or application of, indigenous biological resources for commercial or industrial exploitation, including:
a) the systematic search, collection or gathering of these resources or making extractions from these resources for purposes of research, development or application;
b) the utilization for purposes of research or development of any information regarding any traditional uses of indigenous biological resources by indigenous communities; or
c) research on, or the application, development or modification of, any traditional uses, for commercial or industrial exploitation.
In relation to bioprospecting, 'indigenous biological resources' have been defined to include:
(a)
i) [any living or dead animal, plant or other organism of an indigenous species]; b) [any derivative of such animal, plant or other organism]; or c) [any genetic material of such animal, plant or other organism], whether gathered from the wild or accessed from any other source, including any animals, plants or other organisms of indigenous species cultivated, bred or kept in captivity or cultivated or altered in any way by biotechnology; ii) any cultivar, variety, strain, derivative, hybrid or fertile version of an indigenous species or of any animals, plants or other organisms referred to in subparagraph i); and:
iii) any exotic animals, plants or other organisms, whether gathered from the wild or accessed from any other source which, through biotechnology, have been altered with any genetic material or chemical compound found in any indigenous species or any animals, plants or other organisms referred to in subparagraph i) or ii); but excludes:
(b)
i) genetic material of human origin;
ii) any exotic animals, plants or other organisms, other than exotic animals, plants or other organisms referred to in (a) iii); and
iii) indigenous biological resources listed in terms of the International Treaty on Plant Genetic Resources for Food and Agriculture.
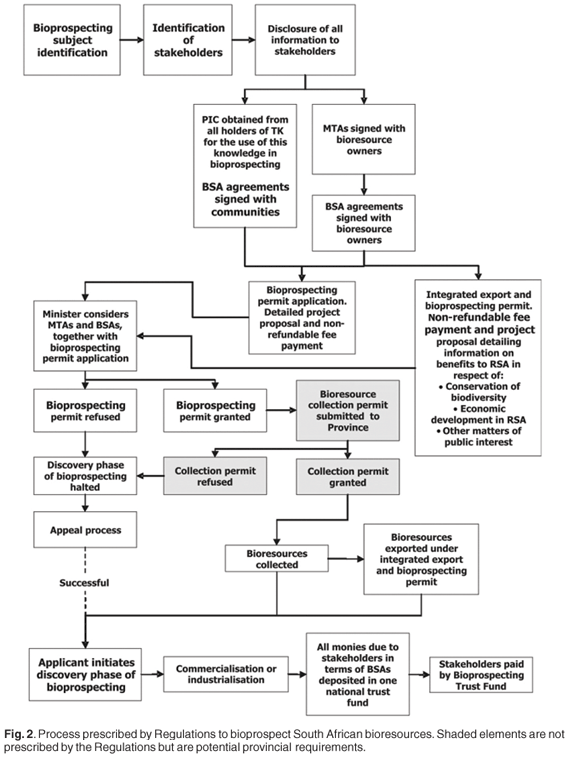
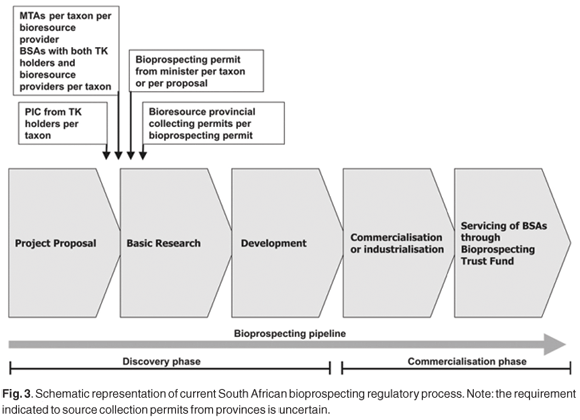
No definitions have been provided in the Act14 for 'indigenous community' or 'commercial or industrial exploitation'. Stakeholders who are expected to benefit from entering into benefit-sharing agreements (BSAs) with bioprospecting permit applicants were defined as 'a person, including any organ of State or community, providing or giving access to the indigenous biological resources to which the application relates [Section 82 (1)(a)], and an indigenous community i) whose traditional uses of the indigenous biological resources to which the application relates have initiated or will contribute to or form part of the proposed bioprospecting; or ii) whose knowledge of or discoveries about the indigenous biological resources to which the application relates are to be used for the proposed bioprospecting' [Section 82(1)(b)]. The Act14 requires [Section 82(2)] that the applicant enters into BSAs with the above stakeholders, as well as a material transfer agreement (MTA) with the bioresource provider, approved by the minister, before the actual bioprospecting permit application will be considered (Figs 2 and 3). Of particular significance are two elements prescribed by the Act for the BSAs: first, [Section 83(1)(d)] that ' the manner in which and the extent to which the indigenous biological resources are to be utilized or exploited for purposes of such bioprospecting' be set out and [Section 83(1)(e)] ' the manner in which and which the extent to which the stakeholder will share in any benefits that may arise from such bioprospecting' be given. These onerous requirements well reflect the 'cherry picking' notion of the bioprospecting process that has been adopted by the legislators. Indeed, the Act prescribes processes that better support the ABS context of imminent bulk bioresource extraction rather than the searching of bioresources for potential value, or bioprospecting. The full benefits that are likely to arise from the discovery phase of a bioprospecting project are impossible to determine, as is the manner and extent to which the bioresources might be utilized (if, ultimately, at all). Based on the above perceptions, realistic BSAs are thus extremely difficult to negotiate, with the process being unduly difficult, costly and onerous for the applicant. To minimize expenses associated with MTAs and their related BSAs, applicants will naturally seek out resource owners whose ownership status is unambiguous, and who are least demanding, complicated, or unobstructive as negotiating partners.
Mandates, responsibilities and procedures
The Act14 has gone as far as detailing specific mandates and responsibilities [Section 82(4)], and prescribes various permitting procedures (Section 81) that should arguably have been better assigned to the Regulations. The focus on the details of processes has detracted effectively from key principles considered by the white paper,48 namely, needs to facilitate access and stimulate sustainable economic development. Although the Act14 has delegated a range of responsibilities to undefined 'issuing authorities' (Section 82), the Regulations15 later make clear [Ch. 2, 6(1)(a)(b)] that the minister of the DEAT is the issuing authority for bioprospecting permits, as well as for integrated export and bioprospecting permits. In terms of institutional mechanisms, the Act does not identify a single 'ABS focal point', a desirable feature stressed by the Bonn Guidelines.51 However, the office of the minister of the DEAT deals with the issuing of bioprospecting-related permits, and could thus be considered the national focal point for bioprospecting issues.
Bioprospecting, Access and Benefit-sharing Regulations
Act–Regulations conformity
Because the Act14 prescribes inappropriate and detailed processes, little latitude is available for subsequent implementation of a practicable system in terms of regulations. Rather than redress the very obvious deficiencies of the Act's Chapter 6 through an immediate legal reform process, the inherent problems have been compounded through promulgation of Act-compatible Regulations15 that became effective from 1 April 2008 (Fig. 1). Procedures not prescribed by the Act have been subsequently detailed by the Regulations, and detailed application forms have been appended to facilitate permit applications of various types, as well as MTAs and BSAs. The minister is required to issue permits after: i) ensuring that the interests of stakeholders (including indigenous communities) are protected (through BSAs and MTAs), ii) that prior informed consent has been obtained, and iii) full disclosure of information relating to the proposed bioprospecting has been submitted to the stakeholders (Fig. 2). The discovery phase and/or commercialization phase of a bioprospecting project may only be initiated with a bioprospecting permit issued by the minister [Ch. 1, 4(1)].15
Consideration of definitions
A number of definitions not provided by the Act are contained in the Regulations. Of particular interest in relation to BSAs is that for 'indigenous community', meaning 'any community of people living or having rights or interests in a distinct geographical area within the Republic of South Africa with a leadership structure and a) whose traditional uses of the indigenous biological resources to which an application for a permit relates, have initiated or will contribute to or form part of the proposed bioprospecting; or b) whose knowledge of or discoveries about the indigenous biological resources to which the application for a permit relates are to be used for the proposed bioprospecting.'15 The Regulations do not define the required 'leadership structure' of the indigenous community, to allow for the unambiguous identification of appropriate TK-holding indigenous communities eligible to enter into BSAs. The 'commercialization phase of a bioprospecting project' means 'any research on, or development or application of, indigenous biological resources where the nature and extent of any actual or potential commercial or industrial exploitation in relation to the project is sufficiently established to begin the process of commercialization'. The definition provided15 for 'commercialization' (pp. 7–8) includes the following activities in relation to indigenous biological resources—
a) the filing of any complete intellectual property application, whether in South Africa or elsewhere;
b) obtaining or transferring any intellectual property rights or other rights;
c) commencing clinical trials and product development, including the conducting of market research and seeking pre-market approval for the sale of resulting products; or
d) the multiplication of indigenous biological resources through cultivation, propagation, cloning or other means to develop and produce products, such as drugs, industrial enzymes, food flavours, fragrance, cosmetics, emulsifiers, oleoresins, colours and extracts.
In relation to this definition (part c), 'product development' is not defined, but may reasonably include drug formulations, which would in fact be required prior to the clinical trial phase. Product development could also include new products based on established and well-branded commodities, such as rooibos: the development of soaps and handcreams from a plant historically marketed as a beverage. Further, in relation to c), the conducting of market research is typically undertaken by bioprospectors prior even to embarking on the costly discovery phase of a project. If there is no market niche, then there is no motive to develop a product. Accordingly, the inclusion of market research as a defined component of 'commercialization' is inappropriate. Similarly, so is part d) of this definition, for agroprocessing research is at times an essential prerequisite to determining, during the discovery phase, whether a product can ever be taken beyond the point of proof of principle. When regulations relating to access to bioresources are too commercially biased, they typically tend to be very restrictive, and impact negatively on basic research.52,53 Widespread suspicion that bioprospecting will take place under the guise of basic research stimulates over-regulation, well-exampled by legislation drawn up by several developing nations, including Sri Lanka, Brazil and the Philippines.53 It is therefore important to designate clear limits between access to material for bioprospecting purposes and access for any other kind of research. Given the broad definition of 'bioprospecting' in the Act,14 this has largely been achieved— with the exception of research required to identify candidate bioprospecting subjects. Logically, the initial identification of a bioprospecting subject inevitably involves some research, which by definition [Section 1(1)]14 is bioprospecting. Initially, this research would involve fieldwork that includes interaction with communities, as well as mining of data held in both public and privileged domains. Basic research of this nature forms a standard and essential part of the discovery phase of any bioprospecting project. In terms of the Regulations,15 no distinction is made between the various components of the discovery phase, which is defined (p. 9) as: 'any research on, or development or application of, indigenous biological resources where the nature and extent of any actual or potential commercial or industrial exploitation in relation to the project is not sufficiently clear or known to begin the process of commercialization'. Accordingly, the undertaking of research for the purposes of identifying a subject worthy of further (discovery phase) bioprospecting is illegal without a bioprospecting permit. However, bioprospectors are nonetheless required to identify their subjects before applying for a permit,15 placing them in a paradoxical Catch 22 predicament: they will have contravened the law in attempting to comply with it.
The seriousness of this situation may be appreciated when one considers that a person is guilty of an offence if that person, without a permit, undertakes bioprospecting involving indigenous biological resources [Ch. 4, 20(a)(i)].15 A person, so convicted, is liable to imprisonment for a period not exceeding five years, an appropriate fine, or to both a fine and imprisonment [Ch. 4, 21(1) (a)(b)(c)].15 Despite the above implications, 'research' is not defined in either the Act or the Regulations. While there is a clear need to ensure that the rights of all stakeholders (sensu Act)14 will be protected in instances where a commercially or industrially valuable discovery is made during basic research, the researchers, so involved, clearly also need protection. The regulations have appropriately made allowance for serendipitous events which lead to an application for a bioprospecting permit, although full explanations regarding activities leading to such finds are required by the minister (Ch. 2, 9(2)(b)].15
Applicants for bioprospecting permits – who need apply?
Importantly, the definition of those who engage in the multiplication of indigenous biological resources through cultivation or propagation to generate products as engaging in commercialization requires that they, too, obtain bioprospecting permits from the minister. Accordingly, for example, every grower who supplies the rooibos tea industry with plant material needs to apply for a permit, and to secure BSAs with relevant stakeholders. This outcome raises the issue of the identity of the party in the value-adding chain that needs to secure a bioprospecting permit to operate with legal compliance. In this regard, the Regulations exempt those who trade in commercial products purchased from a bioprospector, provided that the bioprospector has a relevant bioprospecting permit (Exemptions 2.3).15 However, those involved in the 'commercialization phase of a bioprospecting project' would, by definition, include all those who develop or apply indigenous biological resources where the nature of the project is sufficiently established to start a process of commercialization. Not only would growers require bioprospecting permits, but also commercial gatherers/wild-crafters, formulators, processors, packaging firms, and marketers, besides those who hold IPR in the form of patents. Neither contractor nor sub-contractor involved in the bioprospecting process appears to be exempted. The requirement in the regulations for non-South Africans to apply for a permit in conjunction with a South African judicial body or citizen should encourage the establishment of partnerships, and lead to technology transfer and information sharing. Such partnerships nevertheless place an onerous legal risk on the South African party should the non-South African party(ies) renege on any agreement.
Position of existing industries
In that a bioprospecting permit is now legally required for any stage of a bioprospecting project, definitions provided by the Regulations15 for 'the discovery phase of a bioprospecting project', and the 'commercialization phase of a bioprospecting project', serving principally to inform existing bioresource-based industries that they are indeed engaged in a commercialization phase, and are required to apply for a bioprospecting permit within six months from 1 April 2008 [Section 22(2)].15 This means that most existing industries (for example, rooibos, marula, honeybush, Aloe and Hoodia) need to take into consideration stakeholder interests [Section 22(3)(a)(b)]: failure to secure BSAs acceptable to the minister, notwithstanding his/her intervention, will legally require the industry(ies) to terminate their operations [Section 22(4)(a)].15 This positions newly-defined stakeholders who may regard their past benefits (if any) to be inequitable, to hold a veto on whether established industries may continue their business. Genuine TK-holding communities, in particular, are now empowered through being dealt an extremely strong negotiating hand by the DEAT. Although exact figures of the size of communities involved in, for example, the Cape aloes (Aloe ferox) industry in the southern Cape are not available, it is likely that several thousand individuals benefit directly or indirectly from harvesting, processing and selling aloe juice, and increasingly, aloe gel.11,54,55
Inexplicably, only two industry subdivisions have been exempted from the Regulations15 (though seemingly without undergoing the public consultation process prescribed by Sections 86, 99 and 100):14 the aquaculture or mariculture sector that produces material for consumption (Exemptions 2.7), and the existing local and international cut flower and ornamental plant market segment (and those who supply them)(Exemptions 2.6).15 However, the South African horticultural industry has little respite, as its inherent potential to stagnate is now framed by the same regulations: the development of new plant varieties and related products—competitive advances essential for sustaining this fashion-linked sector—are explicitly not exempted (see Exemptions 2.4.2).15
Practical implications for future bioprospecting
The Act and Regulations require that prior to engagement in the discovery phase of bioprospecting, the applicant needs to obtain MTAs (which are reasonably attainable) and BSAs (which have practical and financial complications). It does not make sense to negotiate a BSA before a permit has been granted (if required) to collect a bioresource for research purposes. Nor for that matter should the negotiation of BSAs precede the assessment of the nature and extent of any actual commercial or industrial value in relation to a bioresource, prior to any process of commercialization. Negotiations surrounding BSAs will focus in the vast majority of cases on biodiversity elements that make merit-worthy subjects for preliminary discovery-phase research—being of unknown value—but in reality may never lead to a commercial or industrial product. As few as one in 250 000 plant samples leads directly to a commercial drug.25 Such negotiations should take place at the start of the commercialization phase rather than prior to the discovery phase, as is currently required (Fig. 3). Memoranda of understanding (MOUs) agreeing to negotiate BSAs at a later stage would suffice in the case of landowners signing MTAs: indeed, the MOU should be incorporated into such MTAs. Further, there is a risk that considerable resources (both human and financial) will be wasted if the minister approves a range of negotiated agreements and a bioprospecting permit, only to have a provincial conservation authority refuse a collection permit on ecological grounds. In the current system (Figs 2 and 3) qualified approval to collect the amounts required should accordingly first be obtained from the provincial authorities. However, neither the Act nor the Regulations stipulate that bioresource collection permits are required following the successful granting of a bioprospecting permit. This is currently presumed (Figs 2 and 3), particularly in relation to CITES and Threatened or Protected Species (TOPS), and as national legislation (e.g. NEMBA)14 overrules existing provincial legislation only if the latter is in conflict. Clarity in this respect is essential, as well as on the nature of related 'integrated permits' provided for in the Act [Section 92(1)(b)],14 but surprisingly not dealt with in the Regulations.15 Provision for appeals against decisions by the minister are accommodated [Ch. 3(14)(15)]15 (Fig. 2), as are challenges to decisions made by provincial conservation authorities, which need be addressed through the Promotion of Administrative Justice Act.
Dynamic BSAs make allowance for changes in benefit-sharing arrangements as concepts develop towards products. The Act does allow for amendments to such BSAs [Section 83 (1)(f)],14 going as far in the Regulations as requiring their regular review (Annexure 8, 11).15 However, applicants will undoubtedly have to deal with the perceptions of TK-holding communities and/or bioresource providers that bioprospecting generates financial returns in the form of fees, upfront payments, milestone payments and royalties. The South African legislation's 'cherry picking' construal of the usual bioprospecting process could transfer to a TK-holding community level, especially if the legislating department develops and implements concurring community education programmes. The very process prescribed by the legislation/regulations will reinforce this perception, given the inappropriate timing of the BSA negotiations (Figs 2 and 3), and the bureaucratic character of the permit process (Annexures 2 and 4).15 The prescribed format for BSAs (Annexure 8)15 includes suggestions for non-monetary and 'in kind' benefits, as well as monetary benefits. The benefits identified in BSAs negotiated at the beginning of the bioprospecting process cannot realistically consider any major financial returns; the total benefits derived may ultimately take the form of only new or augmented information, and voucher and other preserved specimens having been lodged with museums or herbaria. Such actual benefits from the discovery phase of bioprospecting may be perceived as irrelevant and insufficient to communities, whose expectations are probably higher and financially orientated, given the prevalence of rural poverty. However, some international case studies have shown that some traditional people do not always seek financial rewards in such situations.56,57 BSAs and their linked PIC component, however, may well prove to be one of the most complex, cumbersome and difficult aspects for a bioprospecting permit applicant. While the regulations require that PIC be obtained from every stakeholder who will be providing the biological resource and/or the indigenous knowledge, they do not elaborate on the preferred means of engaging with communities.
The Act legislates for the establishment of a Bioprospecting Trust Fund (Section 85)14 into which all moneys arising from BSAs and due to stakeholders need be deposited. The Regulations [Ch. 3, 19(3)(b)] require that the director-general should manage and be accountable for the Trust Fund, and oversee the payment of what is due to stakeholders [Ch. 3, 19(4)(c)].15
In addition, it is sometimes very difficult to determine the identity of the bioresource owner, and more so, the identity of the genetic resource owner (see Box 1). Such potential stakeholders include private landowners, tribal authorities, municipalities, provincial authorities and the state. In this regard, harmonized benefit-sharing legislation, compatible with that of adjacent countries, is desirable. Much hinges here on decisions made at a global scale, particularly with regard to intellectual property and the patenting of biological material59 as well as the negotiation of an International Regime on Access and Benefit-Sharing. Whereas individual landowners signing MTAs may benefit through BSAs, the Regulations make no provision for individual TK-holders (for example, a traditional healer with unique knowledge) entering into a BSA; rather only for indigenous communities (Annexure 8, 4).15 Problems can also arise from claimants being located in different countries, e.g. Hoodia species are used as appetite suppressants by the San who, together with Hoodia, are located in several southern African countries.60,61 Some of these communities may give consent while others may not, and others may remain completely unaware, despite the best of efforts to inform them. However, assistance may be requested from the minister to attempt to conclude the necessary agreements (Annexure 2, Part 1, 34).15 There is also a possibility of identification or emergence of additional stakeholders or claimants during a discovery phase of bioprospecting (which typically extends for several years). In addition to TK-holding communities and bioresources providers, there are scientific experts who, through their knowledge of botany, pharmacology, phytochemistry, and drug development, may be eligible and legitimate claimants for benefits, even though they were not identified in the original approved BSA. These considerations need to be tabled during reviews of BSAs. By and large, benefits should be directed in such a manner as to promote the sustainable use and conservation of biodiversity (Bonn Guidelines, clause 48).51

From the example (see Box 2) it is clear that the mandatory bioprospecting process (Fig. 2) places an impediment on value-addition to South Africa's biodiversity. Bioprospectors will be disinclined to continue with broad-scale screening, given the uncertainty and cost-inefficiency. Similarly, bioresource access is affected adversely, which begs the question of whether South Africa has, in implementing bioprospecting and ABS legislation and regulations,14,15 ultimately failed to meet its sustainable-use CBD obligations. These require the promotion of the sustainable use of South African components of biological diversity and ensuring the fair and equitable sharing of benefits arising out of the exploitation of genetic resources (Article 1).16
Alternative regulatory model
Less restrictive, and more appropriate and practical would be to negotiate BSAs only after the commercial or industrial value of a bioresource has been determined. In the above illustrative example, if only two leads show commercial potential, then between two and seven BSAs are likely to require negotiation. Figure 4 presents an alternative model for practical bioprospecting regulatory processes which will conform to CBD objectives. We interpret Article 8(j) of the CBD16 as indicating that approval and involvement of holders of TK is required for the wider application (namely, commercialization or industrialization), but not for the initial explorative research (the discovery phase). The research undertaken here is notionally distinct from its application; were it not, then all research associated with both traditional knowledge and bioresources (for example, fundamental ethnopharmacology, natural products chemistry and ethnobiology) would be subject to PIC from TK-holding communities. The implementation of this model requires significant reform of both the relevant chapters (6 and 7) of the Act14 and Regulations.15
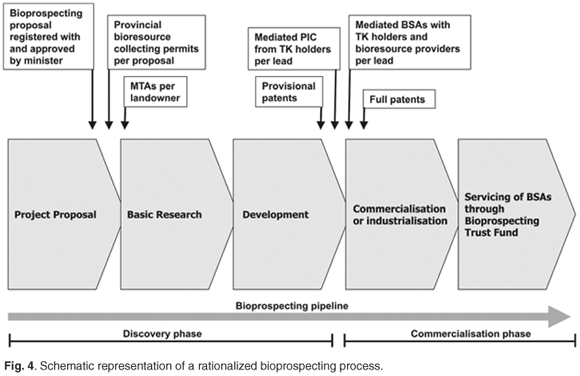
The model respects (through MTAs) the bioresource provider's permission to access bioresources, and positions TK-holding communities—with their veto option—to offer PIC before the wider application of their knowledge. These communities then also share benefits according to mediated BSAs negotiated with the applicants. Mediators involved in negotiations should include representatives of the DTI, as well as the DEAT. Significantly, once a promising lead has been identified and the commercialization phase is to commence, the BSA-negotiating positions of communities and/or bioresource providers become strengthened substantially. This is particularly so, given that a large investment would already have been made by the bioprospector in the discovery of a commercializable lead. Bioprospectors may thus reasonably argue against this veto clause, and seek instead to resolve BSAs through arbitration, if necessary. However, national CBD compliance may be compromised thereby. The existing costly and potentially wasteful requirement for generating numerous contracts that could soon become defunct is also avoided. Regulatory requirements, such as monitoring of the BSA agreements and ensuring compliance, imply the availability of informed capacity, as these are not simple processes to implement. The introduction of a system that generates fewer BSAs will ease the workload on the office of the minister, and release capacity therein to mediate the conclusion of MTAs and their regular reviewing, and to liaise further with provincial authorities whose permitting decisions may impact upon value-adding activities of national relevance.
According to the model (Fig. 4), a detailed bioprospecting proposal is considered by the minister, and following authorization, the discovery phase proceeds. This phase is tracked by DEAT. When the commercialization phase of bioprospecting is reached, and appropriate BSAs (with PIC from TK-holding communities) have been negotiated for a particular lead, the minister is then approached for a bioprospecting permit to continue work. We advocate that provisional patents be lodged prior to negotiations with either the bioresource providers, or indigenous communities, to protect IP generated through innovative research.
Proposed amendments to the Act and Regulations
The National Environment Laws Amendment Bill,62 which was published in the Government Gazette No. 31075 of 20 May 2008, drafts proposed amendments to Chapters 6 and 7 of Act 10 of 2004. These amendments stipulate that anyone engaging in the discovery phase of bioprospecting has to notify the minister, providing particulars in a format yet to be published. Bioprospectors involved in this discovery phase need to sign a commitment to comply with the legal requirements when the commercialization phase of bioprospecting is reached. Thus a bioprospecting permit (and the associated need to negotiate BSAs) would only be required at the start of the commercialization phase of bioprospecting. The bill also contains a proposal to expand the concept of stakeholders to include specific individuals whose traditional uses of knowledge of, or discoveries about, the indigenous biological resources will form part of the proposed bioprospecting. The proposed amendments would entitle the director-general of DEAT to appoint a trustee to administer the Bioprospecting Trust Fund on his/her behalf, and/or to allow individual trusts to be established, as long as they are managed in compliance with relevant legislation. Provision is also made for the renewal and amendment of permits, and the increase in fines. Possibly the most significant proposal relates to the expansion of arbitrary powers provided to the minister under Section 86 of an amended Act:
1) The Minister may by notice in the Gazette –
(b) declare that this Chapter [on bioprospecting, access and benefit-sharing] does not apply to certain categories of research involving indigenous biological resources or commercial exploitation of indigenous biological resources.
Notably, the minister is still required in terms of the Act [Section 86(2)] to follow a consultative process, in accordance with Sections 99 and 10014 prior to publishing an exemption notice. Such consultation includes giving notice of the proposed execution of this power through publication in both the Gazette and at least one national newspaper, allowing due time for public comment.
The above amendments have been drafted for public comment, but remain to be accepted, possibly in modified form.
Conclusion
South Africa, with its unique and remarkably rich biodiversity, and a strong foundation of biodiversity research (for example, in botany63 and horticulture64), could be a source of many new commercially exploitable leads and beneficial products. Directed research must be allowed to progress with minimal restraint, especially in our emerging economy. Such development, coupled to sound natural resource management, can generate both financial and non-financial benefits. This is desirable in South Africa, where exploitation of bioresources is sought in a sustainable manner. The rider is whether the regulations will encourage those with an interest in biodiversity and its products to act responsibly and equitably in sharing the benefits with qualifying parties. Or will the regulations coerce interested and affected parties further into a situation where they are tempted, either to 'fly under the radar' when accessing bioresources (evasion, in preference to compliance) or to seek resources from adjoining countries with either less restrictive laws or a legislative void? It is essential that regulations allow bioprospectors to comply with regulatory measures with ease. Such facilitative legislation can nurture value-adding activity, both locally and abroad, for the benefit of South Africa as well as the global community. Current well-intentioned but impracticable legislation14 and regulations15 could impose severe restraint on bioprospecting activity (and benefits derived therefrom) in years to come, besides collapsing our existing bioresource-based industries within the country. While legislators may argue for purposive interpretation of their bioprospecting definition, the exemptions given to some industries, and not others, indicate otherwise. Gazetted proposals for expansion of the powers of the minister to exempt selected bioprospecting/commercialization activities62 raises uncertainty for bioprospecting, given the arbitrary nature of this process. What are the criteria for exemption, and who is deemed eligible thereto? Positive alternatives to the current legislative scenario will require substantial revisions of both the relevant chapters of the Act and associated Regulations—beyond current proposals.62 These include changes to, or the addition of, definitions to provide legal certainty: both 'commercialization' and 'research' require reconsideration as they are critical to the determination of the commercialization phase of bioprospecting. Crucial to amendments would be a fundamental shift in emphasis from an implicit 'cherry-picking' perspective of the bioprospecting process, to one that recognizes bioprospecting as a course of action involving extensive search (prospecting) for value in bioresources. This search phase requires cost-efficient facilitation by regulators, providing for user certainty, rather than the restrictive and expensive bureaucracy that now exists.
We outline an alternative, practical model on which to base legislation that allows South Africa to add value to its biodiversity, while also remaining compliant with the CBD. The South African legislature should take heed of the potentially detrimental consequences for the national economy if appropriately facilitative laws and regulations are not put in place with the greatest of speed.
1. Mittermeier R. A., Gil P. R. and Mittermeier C. G. (1997). Megadiversity: Earth's Biologically Wealthiest Nations. Conservation International, Washington, D.C. [ Links ]
2. Cowling R.M. and Richardson D.M. (1995). South Africa's Unique Floral Kingdom. Fernwood Press, Cape Town. [ Links ]
3. Van Wyk A.E. and Smith G.F. (2001). Regions of Floristic Endemism in Southern Africa. A Review with Emphasis on Succulents. Umdaus Press, Pretoria. [ Links ]
4. Steenkamp Y., Van Wyk B., Victor J., Hoare D., Smith G., Dold T. and Cowling R. (2004). Maputaland-Pondoland-Albany. In Hotspots Revisited: Earth's Biologically Richest and Most Endangered Ecoregions, eds R.A. Mittermeier, P.R. Gil, M. Hoffman, J. Pilgrim, T. Brooks, C.G. Mittermeier, J. Lamoreux and G.A.B. Da Fonseca, pp. 218–229. CEMEX S.A. de C.V., Mexico City. [ Links ]
5. Crous P.W., Rong I.H., Wood A., Lee S., Glen H., Botha W., Slippers B., De Beer W.Z., Wingfield M.J. and Hawksworth D.L. (2006). How many species of fungi are there at the tip of Africa? Stud. Mycol. 55, 13–33. [ Links ]
6. Klopper R.R., Smith, G.F. and Van Rooy, J. (2002). The biodiversity of Africa. In Rebirth of Science in Africa. A Shared Vision for Life and Environmental Sciences, eds H. Baijnath and Y. Singh, pp. 60–86. Umdaus Press, Pretoria. [ Links ]
7. Ivey P. (1993). South African petaloid geophytes: at home and abroad. M.Sc. thesis, Universities of Cape Town and Birmingham. [ Links ]
8. Jaques R. (1996). The Commercial Use of South Africa's Horticultural Resources. Genetic Resources Conservation Workshop Report. Land and Agriculture Policy Centre, Johannesburg. [ Links ]
9. Mander M. and McKenzie M. (2005). Southern African Trade Directory of Indigenous Natural Products. Commercial Products from the Wild Group, Stellenbosch. [ Links ]
10. Goldblatt P. and Manning J.C. (2000). Cape Plants. A Conspectus of the Cape Flora of South Africa. Strelitzia 9. National Botanical Institute, Pretoria. [ Links ]
11. Newton D.J. and Vaughan H. (1996). South Africa's Aloe ferox Plant Parts and Derivatives Industry. TRAFFIC East/Southern Africa, Johannesburg. [ Links ]
12. Botha M.C. and Kogler L.K. (1998). L'Aloe ferox. La Liliacée Miraculeuse du Desert. Publications Elkomed, Ennetbül. [ Links ]
13. Anon. (n.d.). Rooibostee. Die Unieke Smaak Uit die Sederberge. Rooibosteebeheerraad, Clanwilliam. [ Links ]
14. NEMBA (2004). National Environmental Management: Biodiversity Act (No. 10 of 2004). Government Gazette No. 26436, 7 June 2004. Department of Environmental Affairs and Tourism, Pretoria. [ Links ]
15. DEAT (2008). National Environmental Management: Biodiversity Act 2004 (Act 10 of 2004): Commencement of Bio-prospecting, Access and Benefit Sharing Regulations 2008. Government Gazette No. 30739, 8 February 2008. Department of Environmental Affairs and Tourism, Pretoria. [ Links ]
16. CBD (1992). Convention on Biological Diversity. United Nations Environment Programme, Geneva. [ Links ]
17. Balick M.J. (1990). Ethnobotany and the identification of therapeutic agents from the rainforest. In Ethnobotany and the Search for New Drugs. Ciba Foundation Symposium 185, eds D.J. Chadwick and J. Marsh, pp. 22–39. John Wiley, Chichester. [ Links ]
18. Cox P.A. (1990). Ethnopharmacology and the search for new drugs. In Ethnobotany and the Search for New Drugs. Ciba Foundation Symposium 185, eds D.J. Chadwick and J. Marsh, pp. 40–55. John Wiley, Chichester. [ Links ]
19. Du Toit B.M. (1998). Modern folk medicine in South Africa. S. Afr. J. Ethnol. 21, 145–152. [ Links ]
20. Moerman D.E. (1991). The medicinal flora of Native North America: an analysis. J. Ethnopharm. 31, 1–42. [ Links ]
21. Taniguchi M. (1993). Ethnobotanical drug discovery based on medicine men's trials in the African savanna: screening of east African plants for antimicrobial activity II. J. Nat. Prod. 56, 1539–1546. [ Links ]
22. Arnold T.H., Prentice C.A., Hawker L.C., Snyman E.E., Tomalin M., Crouch N.R. and Pottas-Bircher C. (2002). Medicinal and Magical Plants of Southern Africa: an Annotated Checklist. Strelitzia 13. National Botanical Institute, Pretoria. [ Links ]
23. Fourie T.G., Swart I. and Snyckers F.O. (1992). Folk medicine: a viable starting point for pharmaceutical research. S. Afr. J. Sci. 88, 190–192. [ Links ]
24. Clarkson C., Maharaj V.J., Crouch N.R., Grace O.M., Pillay P., Matsabisa M.G., Bhagwandin N., Smith P.J. and Folb P.I. (2004). In vitro antiplasmodial activity of medicinal plants native to or naturalised in South Africa. J. Ethnopharm. 92, 177–191. [ Links ]
25. Macilwain C. (1998). When rhetoric hits reality in debate on bioprospecting. Nature 392, 535–540. [ Links ]
26. Cordell G.A. (2000). Biodiversity and drug discovery – a symbiotic relationship. Phytochemistry 55, 436–480. [ Links ]
27. Soejarto D.D. (2001). Intellectual property rights and indigenous knowledge: credit where credit is due. J. Ethnopharm. 74, iii. [ Links ]
28. Wynberg R. (2004). Bioprospecting delivers limited benefits in South Africa. Eur. Intell. Prop. Rev. 26, 239–243. [ Links ]
29. Gollin M.A. (2002). Elements of commercial biodiversity prospecting agreements. In Biodiversity and Traditional Knowledge. Equitable Partnerships in Practice, ed. S.A. Laird, pp. 312–332. Earth- scan, London. [ Links ]
30. Aylward B. (1995). The role of plant screening and plant supply in biodiversity conservation, drug development and health care. In Intellectual Property Rights and Biodiversity Conservation: an Interdisciplinary Analysis of the Values of Medicinal Plants, ed. T. Swanson, pp. 93–126. Cambridge University Press, Cambridge. [ Links ]
31. Soejarto D.D., Xuan L.T., Vu B.M., Dac L.X., Bich T.Q., Southavong B., Sydara K., Bouamanivong S., Zhang H.J., Fong H.H.S., Tan G.T., Pezzuto J.M., Franzblau S., Gyllenhaal C., Riley M.C., Hiep N.T., Loc P.K. and Hung N.V. (2002). Implementing IPR and benefit sharing arrangements: experiences in the University of Illinois at Chicago-Vietnam-Laos ICBG. In Intellectual Property Rights and Traditional Knowledge on Genetic Resources in Pharmaceutical and Cosmetic Business. Symposium Organised by Japan Bioindustry Association and National Institute of Technology and Evaluation, pp. 47–83. Tokyo, Japan. [ Links ]
32. Fenwick S. (1998). Bioprospecting or biopiracy? Drug Discov. Today 3, 399. [ Links ]
33. Masood E. (1998). Old scores surface as African states face new opportunities. Nature 392, 540. [ Links ]
34. Van Wijk J. (2000). Phytobusiness requires social chemistry. Phytochemistry 55, 93–95. [ Links ]
35. Hirsch L.P. (2005). Provider and user country measures – do two wrongs ever make a right? Expert International Workshop on ABS, co-hosted by Norway and South Africa, Cape Town. [ Links ]
36. Farnsworth N.R. and Bigel A.S. (1977). Problems and prospects of discovering new drugs from higher plants by pharmacological screening. In New Natural Products and Plant Drugs with Pharmaceutical, Biological or Therapeutic Activity, eds H. Wagner and P. Wolff, pp. 1–22. Springer-Verlag, Heidelberg. [ Links ]
37. Tyler V.E. (1986). Plant drugs in the twenty-first century. Econ. Bot. 40, 279–288. [ Links ]
38. Soejarto D.D. (1993). Logistics and politics in plant discovery. In Human Medical Agents from Plants, eds A.D. Kinghorn and M.F. Balandrin, pp. 96–111. American Chemical Society, Washington, D.C. [ Links ]
39. WTO (1995). Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS) (Annex 1C of the Marrakesh Agreement). Agreement Establishing the World Trade Organization. World Trade Organization, Geneva. [ Links ]
40. Masood E. (1998). Social equity versus private property: striking the right balance. Nature 392, 537. [ Links ]
41. Gaia and GRAIN (1998). TRIPS versus the CBD: conflicts between the WTO regime of Intellectual Property Rights and sustainable biodiversity management. Global Trade and Biodiversity in Conflict. Issue 1. Gaia Foundation and GRAIN, London and Barcelona. [ Links ]
42. Jha V. and Vossenaar R. (1999). Breaking the deadlock: a positive agenda on trade, environment and development? In Trade, Environment and the Millennium, eds G.P. Sampson and W.B. Chambers, pp. 65–95. United Nations University Press, Tokyo. [ Links ]
43. Patents Amendment Act (2005). Government Gazette No. 28319, 9 December 2005. Department of Trade and Industry, Pretoria. [ Links ]
44. Mara K. and New W. (2008). TRIPS council: half of WTO membership backs biodiversity amendment. Intellectual Property Watch. Online at: www.ip-watch.org/weblog/index.php?p=961. [ Links ]
45. Medaglia J.C. and Silva C. L. (2007). Addressing the problems of access: protecting sources, while giving users certainty. IUCN Environmental Policy and Law Paper No. 67/1. [ Links ]
46. Febres M.E. (2002). La Regulación del Acceso a Los Recursos Genéticos en Venezuela. Serie Mención Publicación, Centro de Estudios del Desarrollo. Universidad Central de Venezuela, Caracas, Venezuela. [ Links ]
47. Wynberg R., Taylor M. and Laird S. (2007). Access and benefit sharing in South Africa: an analysis of legal frameworks and agreements. DEAT Report No. 1/07/274. Southern African Development Community Biodiversity Support Programme. [ Links ]
48. DEAT (1997). White Paper on the Conservation and Sustainable Use of South Africa's Biological Diversity. Government Gazette No. 1095, May 1997. Department of Environmental Affairs and Tourism, Pretoria. [ Links ]
49. Wynberg R. and Swiderska K. (2001). South Africa's Experience in Developing a Policy on Biodiversity and Access to Genetic Resources. International Institute for Environment and Development, London. [ Links ]
50. Tvedt M.W. and Young T. (2007). Beyond access: exploring the implementation of the fair and equitable sharing commitment in the CBD. IUCN Environmental Policy and Law Paper No. 67/2. [ Links ]
51. Secretariat of the Convention on Biological Diversity (2002). Bonn Guidelines on Access to Genetic Resources and Fair and Equitable Sharing of the Benefits Arising Out of Their Utilisation. Montreal. [ Links ]
52. Pethiyagoda R. (2004). Biodiversity law has had some unintended effects. Moves to prevent unfair exploitation of resources could restrict conservation research. Nature 429, 129. [ Links ]
53. Pethiyagoda R., Gunatilleke N., De Silva M., Kotagama S., Gunatilleke S., De Silva P., Meegaskumbura M., Fernando P., Ratnayeke S., Jayewardene J., Raheem D., Benjamin S. and Ilangakoon A. (2007). Science and biodiversity: the predicament of Sri Lanka. Curr. Sci. 92, 426–427. [ Links ]
54. Dekenah J. (1951). The story of aloe tapping. Afr. Wild Life 5, 303–313. [ Links ]
55. Smith G.F. and Van Wyk B. (2008). Aloes in Southern Africa. Struik, Cape Town. [ Links ]
56. Hardison P. (2000). PIC/PIA Part II. ICBG: A case study in prior informed consent. Month. Bull. Canad. Indig. Cauc. CBD 16, 1–4. [ Links ]
57. Ready T. (2002). 'Biopiracy' issue stops research. Nature Med. 8, 9. [ Links ]
58. Watt J.M. and Breyer-Brandwijk M.G. (1962). The Medicinal and Poisonous Plants of Southern and Eastern Africa. E. & S. Livingstone, London. [ Links ]
59. Ganguli P. (1998). Intellectual property rights in transition. World Pat. Inf. 20, 171. [ Links ]
60. Victor J.E., Nicholas A., Bruyns P.V., Venter H.T.J. and Glen H.F. (2003). Apocynaceae. In Plants of Southern Africa: an Annotated Checklist. Strelitzia 14, eds G. Germishuizen and N.L. Meyer, pp. 132–177. National Botanical Institute, Pretoria. [ Links ]
61. Wynberg R. (2004). Rhetoric, realism and benefit sharing: use of traditional knowledge of Hoodia species in the development of an appetite suppressant. J. World Intell. Prop. 7, 851–874. [ Links ]
62. DEAT (2008). National Environment Management Laws Amendment Bill. Government Gazette No. 31075, 20 May 2008. Department of Environmental Affairs and Tourism, Pretoria. [ Links ]
63. Smith G.F., Davis G.W., Donaldson J. and Rourke J.P. (1995). Research in the National Botanical Institute of South Africa. S. Afr. J. Sci. 91, 597–604. [ Links ]
64. Smith G.F., Brown N.A.C., Botha D.J., Rutherford M.C., Donaldson J.S., De Lange J.H. and Davis G.W. (1999). Horticultural research in the National Botanical Institute of South Africa: past achievements and future thrusts. S. Afr. J. Sci. 95, 344–348. [ Links ]
Received 1 July. Accepted 23 September 2008.
Abbreviations: ABS, access and benefit-sharing; BSA, benefit-sharing agreement; CBD, Convention on Biological Diversity, the Rio Convention; CITES, Convention on the Trade in Endangered Species; DEAT, Department of Environmental Affairs and Tourism; DST, Department of Science and Technology; DTI, Department of Trade and Industry; IP, intellectual property; IPR, intellectual property rights; MOU, memorandum of understanding; MTA, material transfer agreement; NEMBA, National Environmental Management Biodiversity Act (Act 10 of 2004); PIC, prior informed consent; TK, traditional knowledge; TOPS, Threatened or Protected Species; TRIPS, Agreement on Trade-Related Aspects of Intellectual Property Rights; WTO, World Trade Organisation.
* Author for correspondence. E-mail: crouch@sanbi.org














