Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Science
versión On-line ISSN 1996-7489
versión impresa ISSN 0038-2353
S. Afr. j. sci. vol.104 no.7-8 Pretoria sep./ago. 2008
RESEARCH LETTERS
Neutralization of acid mine drainage using fly ash, and strength development of the resulting solid residues
V.R. Kumar VadapalliI, *; M.J. KlinkII; O. EtchebersI; L.F. PetrikI; W. GitariI; R.A. WhiteIII; D. KeyIII; E. IwuohaII
IEnvironmental and Nano Sciences Group, Department of Chemistry, University of the Western Cape, Private Bag X17, Bellville 7535, South Africa
IISensor Research Lab, Department of Chemistry, University of the Western Cape
IIIDepartment of Chemistry, University of the Western Cape
ABSTRACT
Acid mine drainage (AMD) from a South African coal mine was neutralized with fly ash (FA) from a local power station. An immediate increase in pH and subsequent decrease in the electrical conductivity (EC) values were observed with the addition of FA. A pH buffering region was observed for all the AMD:FA ratios investigated. This was attributed to precipitation and hydrolysis of the main AMD constituents such as Al and Fe and adsorption of the precipitates upon the ash particles. A high percentage of major, minor and trace elements and SO42– attenuation was achieved after contact of AMD with FA in solution; this removal depended on the final pH of the product water. Most of the contaminants were removed to acceptable levels in one simple procedure by contacting the AMD with FA in suitable ratios. Solid residues (SR) recovered from neutralization reactions were tested for unconfined compressive strength and elastic modulus in order to assess their suitability as backfill material. Strength testing was carried out for 410 days with and without the addition of ordinary Portland cement. The SR with a pozzolanic binder added gained 300% greater strength than without, both of which increased in strength over time. The implementation of this FA treatment process would not only be environmentally beneficial but also would be to the advantage of coal mines and power stations as a way of constructively using the large volumes of waste that they generate.
Introduction
Throughout the world, fly ash (FA), an abundant by-product of coal combustion in power stations, is mostly disposed of in landfills; only a small part of it is used constructively. Poor management of FA is responsible for environmental problems such as loss of usable land, contamination of plants and soil, air pollution and eyesores.1
Most of South Africa's power stations are coal-fired. Eskom, the country's largest electrical utility, generates nearly 20 Mt of FA per annum, of which only 5% is used for cement and brick making; the rest is disposed of in ash dams or stacked on land as ash dumps (www.eskom.ac.za/annreport). FA tends to accumulate as heavy metals in toxic concentrations2 and is considered an environmental hazard in South Africa. Metals, anions and cations are leached from the ash heaps by the wastewater derived from the ash slurry or by subsequent infiltration by rain. This subsequently imposes a risk on surface and ground water. There is therefore mounting pressure on power stations to find alternative ways of using ash productively to protect the environment and to reduce its disposal on land.
Combustion of South African coal produces FA that is highly alkaline due to the presence of free lime. Several authors3–6 have shown that FA can control acid mine drainage (AMD) generation in situ in mine spoils and for remediation of acidic soils. The ash increases the pH and immobilizes the contaminant elements as amorphous hydroxides or oxyhydroxides.
AMD is produced when sulphide minerals, such as pyrite, found in association with the coal or overburden, come into contact with oxygen and water during mining. Sulphide minerals undergo oxidation, which results in the generation of sulphuric acid that mobilizes rock constituents. AMD is characterized by high acidity (pH 2–4), high sulphate concentrations (1–20 g/l) and frequently contains high concentrations of Fe, Mn, Al, Cu, Zn, Pb and Cd. The extreme pH and high ionic content are responsible for highly toxic wastewater. Its handling requires expensive storage, remediation and disposal techniques.
The detrimental effect of AMD on the environment has been widely studied and reported.7–9 Various options to neutralize AMD have been proposed. One of the widely used and well-accepted methods of treating AMD in South Africa is neutralization with lime (CaO) or limestone (CaCO3). In view of the costs involved in the liming process and diminishing lime reserves, and because the efficiency of neutralization by limestone progressively declines because of armouring due to the ferric oxyhydroxides that form, alternative treatment methods are being explored. Previous studies indicated that the treatment of AMD with FA promises to be a viable alternative to liming.10 The successful implementation of this process is expected to address two main issues: the reduction of the cost involved and the effective management of FA. The cost involved compared with liming will be less because FA is freely available in close proximity to coal mines that generate AMD. Unlike FA, the liming treatment requires secondary processes, such as biological methods, for sulphate reduction and addition of reagents like flocculants, which add to the costs. The treatment of AMD with FA, however, generates solid residues (SR) that require disposal.
Mines are backfilled to avoid subsidence, provide support to pillars and walls and to reduce the void volume in underground coal mines.11,12 Greater mine stability prevents rock falls and associated air blasts, which improves mine safety.13 Partial backfilling does not completely eliminate the chance of subsidence but may reduce it by 33% compared with non-backfilled mines.14
The suitability of FA as a backfill material has been studied by various authors.15 The ash has pozzolanic properties and has been investigated for its suitability as a cement replacement.16 The permeability of FA paste backfill is lower than that of soils or mine spoils and unlikely to leach harmful elements in any great quantity.17 That study resulted in FA being reclassified from special waste to inert waste. It has also been reported that backfilling by FA reduces subsidence and the production of AMD.18 No studies were found, however, relating to the suitability of SR resulting from AMD neutralization with FA as possible backfill material. X-Ray fluorescence (XRF) analysis has shown that, despite being used for the active neutralization of AMD, the recovered SR were still strongly alkaline and possessed pozzalinic properties.19 The suitability of SR for backfilling can be physical or chemical. Those of SR to treat AMD seepage passively in a backfill set-up generated alkalinity and long sustained the pH of the leachates at neutral to circumneutral pH levels.20 In addition, it was observed that the leachability of the trace elements was insignificant and so it was safe to dispose of SR as a backfill material. This is a bonus as the sludge emanating from the AMD-FA treatment can be managed effectively. In view of these encouraging results, we performed further evaluation of the strength development of the SR recovered from different AMD-FA neutralization reactions.
We report the conditions for the efficient use of South African FA to neutralize and remove contaminants from AMD. The neutralization reactions were carried out with the object of determining the optimum amount of ash to be used to treat a given volume of AMD. The variations in pH and electrical conductivity (EC) over time were monitored during these reactions and are critically discussed below. We also report on a preliminary investigation of the suitability of SR as backfill material using unconfined compressive strength (UCS) and elastic modulus as parameters of strength development.
Materials and methods
The description of the materials and methods used in the current study is provided in Appendix 1.
Results and discussion
The pH and EC trends for different AMD-FA neutralization reactions are provided in Appendix 2.
Removal of major, minor and trace elements and sulphate
Table 1 shows the initial concentration of sulphates and various elements in the AMD, as well as their concentration in the process waters recovered after the neutralization reactions using different amounts of FA. The contaminants were removed more completely as the amount of FA was increased in the reaction mixture. The final pH attained in the reaction mixture determined the degree of contaminant removal in the final process water. Aluminium values decreased with increasing FA content; the removal was optimum at lower AMD:FA ratios (except for 100:1). The formation of amorphous Al(OH)3 at pH ~5 was most likely responsible for reduced Al in the process waters recovered.21 Slight leaching from FA was observed for the 100:1 ratio for Al, which is attributed to the low pH attained for this mixture.
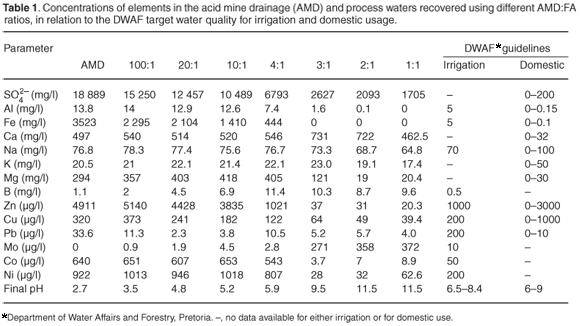
Total Fe decreased in the process water with declining AMD: FA ratios—no Fe was detected for 3:1, 2:1 and 1:1 mixtures. These AMD:FA ratios all attained a pH > 9 over the reaction time, when both Fe3+ and Fe2+ were expected to be out of solution.22 As the pH increased, these cations underwent hydrolysis, forming insoluble amorphous hydroxides/oxyhydroxides [Equation (2B)]. The sulphate concentration decreased with decreasing AMD:FA ratio, which indicated that the formation and precipitation of a secondary mineral phase was responsible for its removal. Research has shown that gypsum is one of the mineral phases that form.19
Several mechanisms could be involved in the removal of sulphate, for example: the initial dissolution of CaO from FA in the presence of the acidic AMD; Ca2+ released in the reaction mixture [Equation (2A)] and its interaction with SO42– to form CaSO4.2H2O (gypsum). This caused the initial sharp decrease in SO42– content. Dissolution of Ba and Sr salts from FA and their consequent interaction with SO42– may also lead to the formation of barite (BaSO4) and celestite (SrSO4) or a mixed (Ba, Sr)SO4 salt. As the pH increases to 3, Fe3+ precipitates to form amorphous ferric hydroxides and oxyhydroxides, which are known to have large surface areas and adsorb some of the SO42–. Oxidation of Fe2+ occurs in the presence of oxygen and is maximum at pH 5–7, with the hydrolysis of the resulting Fe3+ resulting in the formation of amorphous ferric hydroxides, which adsorb more of the SO42–.23 In this pH range a significant decrease in SO42– concentration was observed.19 Formation of amorphous Al(OH)3 and incorporation of SO42– at pH >4 is also believed to contribute to reduced SO42– content. At pH >9 a mineral phase known as ettringite [Ca6Al2(SO4)3(OH)] forms and also contributes to the decrease in SO42– concentrations.19
There was a marginal increase in K and Ca concentrations in the process waters recovered from most of the AMD:FA mixtures. These results follow the trend observed by Jenke and Pagenkopf,22 who showed that the treatment of acidic waters with alkaline reagents left these elements in the process waters. Leaching of Na from the FA was observed only at a 100:1 ratio but thereafter the concentration decreased gradually in the process water with smaller mixture ratios. An increase in Mg concentrations was observed in the process waters recovered from mixture ratios. For lower AMD:FA ratios, however, significant removal of Mg was observed within the pH range 9.5–11.5. Optimum Mg removal between pH 10.49 and 11 has previously been noted.24 Boron and Mo leached significantly from the FA, especially at the low ratios of 3:1, 2:1 and 1:1. The leaching strongly correlated with the amount of FA in the mixture, confirming that the source of these trace elements was dissolution of soluble salts in FA. It has been observed that as B occurs mainly as surface oxide precipitates or is incorporated in the glassy matrix of FA,25 its concentration in the process waters is controlled by the rate at which surface precipitates and glassy phases dissolve and also by the adsorption on the precipitates that form as a result of the AMD:FA neutralization reaction. Boron concentrations were, however, observed to decrease in the process waters recovered from the 3:1, 2:1 and 1:1 mixtures over time when the pH of the final recovered waters was >9 (Table 1). The decrease in B concentration at this pH is attributed to adsorption on calcite. Other studies show that boron concentrations are likely to be low in alkaline leachates that are actively precipitating CaCO3(s).26 These neutralization reactions were conducted in the open, so that ingress of CO2 and formation of calcite was likely.
The low values of Ni observed in process waters for 3:1 and 2:1 mixtures can be attributed to precipitation as Ni(OH)2 or increased adsorption on the precipitating amorphous Al, Fe-oxyhydroxides. Lead values were low up to a ratio of 10:1, after which a slight increase was observed for a 4:1 mixture. However, Pb levels were stabilized for lower ratios. Cobalt, Zn and Cu decreased in the process waters recovered from the low AMD:FA ratios. Copper precipitates as cupric and cuprous ferrite and can be adsorbed on the surface of FA at pH values between 5 and 6.21,27 At pH 6–7, Zn co-precipitates with Si that is solubilized from FA and forms willemite.21,27,28
We conclude that the main mechanisms that governed the removal of contaminants were precipitation, co-precipitation and adsorption at the surface of the FA particles and of the precipitating amorphous Al and Fe hydroxides and oxyhydroxides. Certain trace elements such as B and Mo were observed to leach with increasing FA in the reaction mixture and would have limited this treatment process. It should be noted, however, that most contaminants were significantly removed to trace levels.
The South African Department of Water Affairs and Forestry (DWAF) guidelines indicate different patterns for different elements in process waters (Table 1). Those from 3:1–1:1 and 2:1–1:1 mixtures have 'acceptable' levels of Al for irrigation and domestic purposes, respectively. For Fe, Ni and Co the waters recovered from our 3:1–1:1 mixtures met water quality target. For Cu, even the water recovered from higher AMD:FA ratios had acceptable levels for both irrigation and domestic purposes. For Pb, except for the 100:1 and 4:1 mixtures, the water recovered from the other mixtures met water quality targets. For Zn, the water recovered from 4:1–1:1 mixtures was acceptable for either domestic purposes or for both irrigation and domestic use. The final concentrations of Na and K were within the acceptable range either for irrigation or for both irrigation and domestic use. None of the mixtures managed to remove Ca to the desired limits. Process waters from 2:1 and 1:1 mixtures, however, met the DWAF specifications for Mg. Although a maximum of approximately 91% of the very high level of SO42– was removed from the AMD, none of the waters recovered from any of the mixtures reached the DWAF target. Boron and Mo seemed to leach from FA in the waters recovered from all the mixtures; the leaching was significant for low ratios. Although Mo was observed to leach from FA, the concentrations of this element were within the limits of the target water quality in the process waters recovered from the high ratios (100:1 to 4:1). The waters recovered from 3:1 and 4:1 mixtures had more or less the same pH range specified by DWAF. The rest of the waters were either too acidic or too alkaline and would need post-treatment correction of pH. Because all buffering constituents had been removed by the ash treatment, pH correction would be trivial. Based on the results indicated in Table 1, we conclude that the main benefit would occur for AMD:FA ratios of between 4:1 and 3:1; in our study the active treatment and neutralization of AMD with FA to circumneutral or alkaline pH was optimized at a ratio of 3:1.
Strength testing
Solid residues without a pozzolanic binder
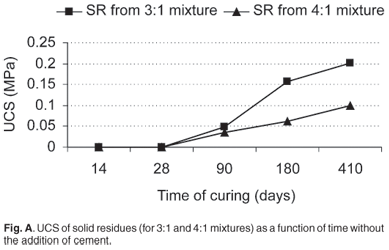
The UCS and elastic modulus are the two most important mechanical properties of a backfill material. The chemistry of the water used to prepare the samples plays an important role in the strength development of the material being tested as demonstrated by Benzaazoua et al.29 To avoid any discrepancy in the chemistry of the waters that were used for mixing in this study, the process water recovered from the 3:1 reaction mixture was used to prepare a slurry with the solid residues recovered from the neutralization reactions. The pH of the process water was 8.83 and the average moisture content of the solid residues recovered from 3:1 and 4:1 mixtures was 29.5% and 33.4%, respectively. The development of UCS and elastic modulus of SR without binder is shown in Figs A and B in supplementary material online. The mean specific gravities of the SR recovered from the 3:1 and 4:1 mixtures were 2.3 and 2.2, respectively.
From Fig. A, it can be observed that after 14 days of curing, no material was strong enough to be tested. After 90 days, the samples had developed some strength. The UCS increased with an increasing amount of ash used during the neutralization reaction, which related to the remaining portion of free lime of the ash after it was applied for neutralization. After 90 days, compressive strengths increased to 0.05 MPa and 0.04 MPa for the 3:1 and 4:1 mixtures, respectively. After 180 days, a further significant increase in strength was again noted for both mixtures. After 410 days, the increase in strength was even more evident. Materials prepared using a 3:1 mixture achieved a UCS of 0.20 MPa whereas that for 4:1 was 0.10 MPa. Strength development of the material was very slow, possibly due to the slow release of unreacted CaO from the solid residues during calcium silicate hydrate (CSH) gel formation.19 An adequate UCS for a backfill material in a typical mine is in the range 0.7–2 MPa,30 which clearly was not achieved within our testing period.
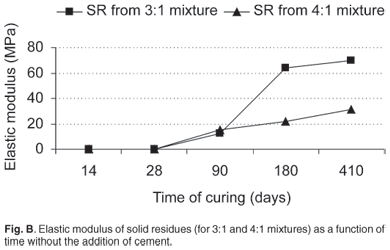
The elastic modulus could not be measured after 14 and 28 days of curing because of the instability of the material (Fig. B). After 90 days, the modulus could be measured and the materials gained some strength, with no major difference between the 3:1 and 4:1 mixtures. After 180 days, the distinctive behaviour of the 3:1 mixture was observed, with a much higher elastic modulus. The same distinction was maintained after 410 days, with the elastic modulus of the 3:1 mixture reaching 70.1 MPa. In general, the SR obtained from the 3:1 mixture developed a significant strength and the elastic modulus of the sample from the 4:1 mixture increased over time to acceptable, but to lower levels (Fig. B). An adequate elastic modulus for a backfill material varies depending upon the mining conditions, which include mining method and depth.31 In our investigations, the elastic modulus was generally greater the more ash in the AMD neutralization reaction.
The greater compressive strength and elastic modulus of solid residue samples recovered from low AMD:FA ratios indicate that strength development was a function of reactions taking place between unconsumed pozzolanic components in the ash and the FA matrix. It also demonstrated that the components contributing to strength development were similar to those influencing the neutralization efficiency of the FA. It has been observed that the less compacted the backfill material, the lower the strength that is developed.29 The small difference in the specific gravity observed between the two SR samples derived from the 3:1 and 4:1 neutralization reactions may also have contributed to the difference in the strength acquired by the two materials.
Solid residues with a pozzolanic binder
The strength testing results of samples that were prepared by blending the slurry of SR with 3% of pozzolanic binder (OPC) are presented in Fig. C online. Stability testing with binder was carried out on only the SR recovered from the 4:1 mixture, over a period of 410 days.
After 14 days, the UCS of SR blended with binder was lower than 0.07 MPa, but significantly higher than the corresponding material without binder (Fig. A). After 28 days, the strength reached 0.11 MPa and after 90 days achieved an acceptable value of 0.23 MPa for backfill purposes. The strength seemed to stabilize after 180 days and was nearly 4 times more than that of the SR without a binder. After 410 days, the UCS was ~0.30 MPa.
The elastic modulus with 3% pozzolanic binder was also tested only on the SR derived from the 4:1 mixture. It was unchanged between 14 and 28 days (53.5 MPa and 53.4 MPa, respectively), after which it increased sharply to 92 MPa (Fig. C online). The rise in elastic modulus continued between 90 and 180 days but a slight decrease was observed at 410 days.
The addition of cement as binder thus increased the UCS of solid residues by 300%. The enhanced strength is attributed to the extra pozzolanic components that favour the formation of various hydrates such as CSH gels.29 The enhanced formation of gypsum as a result of the interaction of Ca2+ and SO42– from the process waters could also have led to increased strength.30 Although the SR recovered from the 4:1 neutralization reaction with the addition of binder did not reach the critical UCS range of 0.7–2 MPa, the gain in strength over time is promising for its application as a backfill material, because it continues to strengthen after placement. As mentioned, the current study used 3% binder to enhance the strength development. The addition of more binder to the SR (for instance, 6%) would be interesting as it might enhance the strength development further. On the other hand, strength development due to addition of the same amount of binder as used in this study (3% binder) to the SR recovered from lower AMD to FA ratios (for example 3:1, as this section only dealt with the SR recovered from the 4:1 ratio) would also be interesting as the strength development of the SR recovered from the 3:1 mixture gained more strength than the SR recovered from the 4:1 mixture in the case of SR without binder.
Another study,19 that used ordinary municipal tap water (our study used process water recovered from the AMD-FA neutralization reaction) for testing the strength development of SR recovered from 3:1 (AMD:FA) mixture with the addition of 1% binder showed different results. It was observed that the SR gained sufficient strength, and took far less time to achieve it, than was observed in our work. This implies that the strength development of SR depends on both the binder and also on the water chemistry used. Similar phenomena relating to water chemistry have previously been noted during the strength development of cured mine tailings.31
Conclusions
The active treatment and neutralization of AMD with FA to circum-neutral or alkaline pH was optimized at an AMD:FA ratio of 3:1. The pH attained in the final process water was dependent on the AMD:FA ratio used. Major contaminants were more effectively removed as the FA content increased in the reaction mixture. The final pH attained seemed to determine the degree of contaminant removal in the final process water. Boron and Mo were leached from FA, more significantly at low AMD: FA ratios. The leaching was strongly correlated with the amount of FA in the mixture, confirming that the source of these toxic trace elements was soluble salts in the fly ash. The Co, Zn and Cu content was reduced mostly in the process waters recovered from the low AMD:FA mixtures.
We concluded that the main mechanisms that governed the removal of contaminants were precipitation, co-precipitation and adsorption at the surface of the FA particles and the precipitating amorphous Al and Fe hydroxides and oxyhydroxides. Specific trace elements were observed to leach with increasing FA in the reaction mixture; their presence in the recovered process water would be a limitation in this treatment process. Our study concentrated on the reactions and trends between AMD from a single coal mine and FA from one power station. Previous studies with materials from different sources, however, have shown that low pH and Fe- and Al-rich AMD can be successfully treated with FA with the same degree of contaminant attenuation. Most contaminants were removed by more than one AMD:FA mixture to levels of water quality set by the Department of Water Affairs.
The strength development of SR, especially that tested without the addition of binder, was slow. The SR tested with 3% OPC became three times stronger than that without.
The successful implementation of industrial processes to treat fly ash could be a solution to the effective use and proper management of waste fly ash, and thus of great benefit to power stations. Fly ash is freely available in close proximity to coal mines and its treatment does not require secondary processes or addition of reagents. This would considerably reduce the costs associated with AMD treatment and should be good news for the mining houses.
We thank the Water Research Commission, Coaltech 2020, the National Research Foundation and the Claude Leon Foundation for their financial support.
1. Carlson C.L. and Adriano D.C. (1993). Environmental impacts of coal combustion residues. J. Environ. Qual. 22, 227–247. [ Links ]
2. Campbell A. (1998). Chemical, physical and mineralogical properties associated with the hardening of some South African fly ashes. M.Sc. thesis, University of Cape Town, South Africa. [ Links ]
3. Stewart R.B, Lee Daniels W. and Jackson M.L. (1997). Evaluation of leachate quality from co-disposed coal fly ash and coal refuse. J. Environ Qual. 26, 1417–1424. [ Links ]
4. Abbott D.E., Essington M.E., Mullen M.D. and Ammons J.T. (2001). Fly ash and lime-stabilized biosolid mixtures in mine spoil reclamation: simulated weathering. J. Environ. Qual. 30, 608–616. [ Links ]
5. Seoane S. and Leirós M.C. (2001). Acidification-neutralization processes in a lignite mine spoil amended with fly ash or limestone. J. Environ. Qual. 30, 1420–1431. [ Links ]
6. Xenidis A, Evangelia M. and Paspaliaris I. (2002). Potential use of lignite fly ash for the control of acid generation from sulphidic wastes. Waste Manage. 22, 631–641. [ Links ]
7. Baird C. (1995). In Environmental Chemistry, pp. 301–302. W.H. Freeman, New York. [ Links ]
8. Evangelou V.P. and Zhang Y.L. (1995). Pyrite oxidation mechanism and acid mine drainage prevention. Crit. Rev. Environ. Sci. Technol. 25(2), 141–199. [ Links ]
9. Bell F.G., Halbich T.F.J. and Bullock S.E.T. (2002). The effects of acid mine drainage from an old mine in the Witbank coalfield, South Africa. Quart. J. Engng Geol. Hydrogeol. 35(3), 265–278. [ Links ]
10. Petrik L., White R., Klink M., Burgers C., Somerset V., Key, D., Iwuoha E., Burgers C. and Fey M.V. (2005). Utilization of fly ash for acid mine drainage remediation. WRC Report No.1242/1/05. Water Research Commission, Pretoria. [ Links ]
11. Barret J.R., Coulthard M.A. and Dight P.M. (1978). Determination of fill stability. Mining with Backfill – 12th Canadian Rock Mechanics Symposium, Canadian Institute of Mining and Metallurgy, Quebec. Special vol. 19, 85–91. [ Links ]
12. Donovan J.G. (1999). The effects of backfilling on ground control and recovery in thin seam coal mining. Master's thesis, Virginia Polytechnic Institute and State University, Blacksburg, VA. [ Links ]
13. Coates D.F. (1981). Rock Mechanics Principles. CANMET, EMRCan, Monograph No. 874, Varennes, Canada. [ Links ]
14. National Academy of Sciences (1975). Underground Disposal of Coal Mine Wastes. National Academy of Engineering, Washington, D.C. [ Links ]
15. Palarski J. (1993). The use of fly ash, tailings, rock and binding agents as consolidated backfill for coal mines. In Proc. Minefill 93, ed. H.W. Glen, pp. 403–408. South African Institute of Mining and Metallurgy, Johannesburg. [ Links ]
16. Grice A.G. (1998). Underground mining with backfill. The 2nd Annual Summit – Mine Tailings Disposal Systems, Brisbane. [ Links ]
17. Jorgenson R.R. and Crooke D.R. (2001). Fly ash paste utilization for placement as mine and landfill backfill at Great River Energy's Coal Creek Station. Proceedings of Minefill, Seattle, WA, 187–194. [ Links ]
18. Siriwardane H.J., Kannan R.S.S. and Ziemkiewicz P.F. (2003). Use of waste materials for control of acid mine drainage and subsidence. J. Environ. Engng 129(10), 910–914. [ Links ]
19. Gitari M.W. (2006). Evaluation of leachate chemistry and contaminant attenuation by fly ash blended acid mine drainage/fly ash solid residues. Ph.D. thesis, University of the Western Cape, Bellville, South Africa. [ Links ]
20. Gitari M.W., Petrik L.F., Etchebers O., Key D.L., Iwuoha E. and Okujeni C. (2008). Passive neutralisation of acid mine drainage by fly ash and its derivatives: a column leaching study. Fuel 87(8–9), 1637–1650. [ Links ]
21. Komnitsas K., Bartzas G. and Paspaliaris I. (2004). Clean up of acidic leachates using fly ash barriers: laboratory column studies. Global Nest 6(1), 81–89. [ Links ]
22. Jenke R.D. and Pagenkpof G.K. (1983). Chemical changes in concentrated, acidic, metal bearing waste waters when treated with lime. Environ. Sci. Technol. 17(4), 217–223. [ Links ]
23. Rose S. and Elliot Crawford W. (2000). The effects of pH regulation upon the release of sulphate from ferric precipitates formed in acid mine drainage. Appl. Geochem. 15, 27–34. [ Links ]
24. Britton H.T.S (1956). Hydrogen Ions, 4th edn. Chapman and Hall, London. [ Links ]
25. Hullet L.D. and Weinberger A.J. (1980). Some etching studies of the microstructure and composition of large aluminosilicate particles in fly ash from coal burning power plants. Environ. Sci. Technol. 14, 965–970. [ Links ]
26. Hollis J.F., Keren R. and Gal M. (1988). Boron release and sorption by fly ash as affected by pH and particle size. J. Environ. Qual. 7, 181–184. [ Links ]
27. Hequet V., Ricou P., Lecuyer Y. and Le Cloirec P. (2001). Removal of Cu2+ and Zn2+ in aqueous solutions by sorption onto mixed fly ash. Fuel 80, 851–856. [ Links ]
28. Manceau A., Lanson B., Schlegel M.L., Harge J.C., Musso M., Eybert-Berard L., Hazemann J.L., Chateigner D. and Lamble G.M. (2000). Quantitative Zn speciation in smelter-contaminated soils by EXAFS spectroscopy. Am. J. Sci. 300, 289–343. [ Links ]
29. Benzaazoua M., Belem T. and Bussière B. (2002). Chemical factors that influence the performance of mine sulphidic paste backfill. Cem. Concr. Res. 32, 1133– 1144. [ Links ]
30. Brackebusch F.W. (1994). Basics of a paste backfill systems. Min. Engng 46, 1175–1178. [ Links ]
31. Amaratunga L.M. and Yaschyshyn D.N. (1997). Development of a high modulus paste fill using fine gold mill tailings. Geotech. Geol. Engng 15, 205–219. [ Links ]
32. Petrik L., Vadapalli V.R.K., Gitari W., Etchebers O., Reynolds K., Surender D., Fester V., Slatter P. and Sery G. (2007). Large scale stability and neutralization capacity of potential mine backfill material formed by neutralization of fly ash and acid mine drainage. WRC Report No. 662, Water Research Commission, Pretoria. [ Links ]
33. Burgers C.L. (2002). Synthesis and characterization of sesquioxidic precipitates formed by the reaction of acid mine drainage with fly ash leachate. M.Sc. thesis, University of Stellenbosch, South Africa. [ Links ]
34. O'Brien R.D. (2000). The neutralization of acid mine drainage by fly ash. M.Sc. thesis, University of Cape Town, South Africa. [ Links ]
35. Ziemkiewicz P.F., Skousen J.G., Brant D.L., Sterner P.L. and Lovett R.J. (1997). Acid mine drainage treatment with armoured limestone in open limestone channels. J. Environ. Qual. 26, 1017–1024. [ Links ]
Received 31 August 2007. Accepted 8 May 2008.
This article is accompanied by supplementary figures and tables online at www.sajs.co.za
* Author for correspondence. E-mail: rvadapalli@uwc.ac.za
Materials and methods
Neutralization reactions
The chemical composition of AMD varies considerably according to its source, whereas the composition of FA is based on the mineralogy of the parent coal and the power station from which it was derived. We investigated only the neutralization reaction between the low pH, Fe- and Al-rich AMD derived from a single coal mine and ash from one power station in Mpumalanga province. AMD from different coal mines was successfully treated with FA from different power stations, however, and the results are provided in one of our technical reports.10
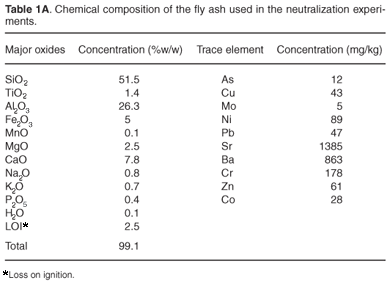
Fresh FA samples were collected from hoppers on the ash collectors. Samples were stored in tightly sealed, plastic containers. The major constituents (% w/w) and trace elements (mg/kg) of FA were determined using XRF (Table 1A in supplementary material online).
Sulphate, Fe and Al rich AMD from a coal mine was collected in high-density polyethylene containers, with air excluded, and refrigerated at <4°C until analysed. Chemical analysis of AMD was conducted using ion chromatography (IC) for sulphates; and inductively coupled plasma–mass spectrometry (ICP–MS) for major, minor and trace elements (Table 1).
Open beaker tests were carried out to determine the change in pH and EC with time for different mixtures of AMD and FA. The AMD was placed in a beaker and a known mass of FA was added, according to the AMD:FA mixture ratio required. For example, for a 1:1 (AMD:FA) ratio, 800 g ash was added to 800 ml AMD. The mixtures were stirred for 240 minutes using an overhead stirrer (with a multi-bladed radial impeller). The pH and EC were recorded at different time intervals using a calibrated Hanna HI 99 1301 portable pH/EC/TDS/temperature probe.
After the reaction, the mixture was allowed to settle until the solid and liquid phases were separated. Thereafter, the supernatant was decanted and filtered using a 0.45-µm pore membrane. The samples were split into two batches for cation and anion analysis. The samples for cation analysis were acidified with analytical grade HNO3, whereas samples for anion analysis were diluted with ultra-pure water and preserved in a refrigerator at 4°C.
Strength testing
Preparation of SR
The SR for strength testing was obtained by reacting AMD and FA in 3:1 and 4:1 ratios using a 250-l agitator, to facilitate neutralization. Agitation was carried out until the mixture attained a neutral or circum-neutral pH. At the end of the neutralization reactions, the mixtures were transferred into buckets and were left for a few hours to allow phases to separate. The supernatants were then separated from the wet SR and both were kept in tightly sealed plastic containers.
Preparation of the samples and curing for strength tests
The samples as cylinders for the strength tests were made by slurrying the SR with process water resulting from the neutralization of AMD with FA. Sufficient water was added to each sample to obtain pumpable slurry, as defined by the marsh cone tests.32 Once prepared, the slurries were split and poured into different cylindrical tubes for curing. The cylinders were prepared in duplicate for each AMD:FA ratio for each curing period. The cylinders were covered with plastic bags, a bottle of water being placed nearby to maintain the required level of humidity during the curing process. Additional samples were prepared for curing and strength testing, by blending the slurries of SR with 3% ordinary Portland cement (OPC), a pozzolanic binder. The schedule of curing and strength testing, with the different blends and periods of curing, is given in Table 1B online. The period of curing ranged from 14 to 410 days. Samples blended with 3% OPC were tested only for the 4:1 ratio up to 410 days.
This section presents results and discussion of the pH and EC trends during the neutralization reactions for various AMD:FA ratios.
The variation of pH with time during neutralization reactions
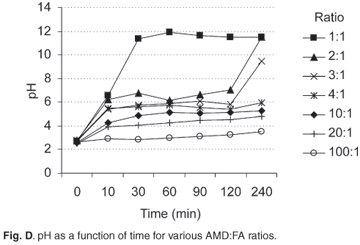
Figure D online shows the pH variation over time as the AMD was progressively neutralized with FA. Consistent trends were observed from high to low AMD:FA ratios, i.e. the more FA added, the higher the pH of the mixture. For all the AMD:FA ratios, on addition of FA there was an immediate increase in the pH of the reaction mixture (except for the 100:1 mixture) followed by a pH plateau and finally a neutral or higher pH (for 1:1, 2:1 and 3:1 mixtures). This immediate increase in the pH on initial contact with AMD can be attributed to the dissolution of free CaO on the surface of the FA spheres, which is readily soluble.
On contact with AMD, under the low acidic regime the free CaO in FA undergoes dissolution to release OH– ions.
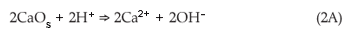
AMD constituents such as Fe3+, Fe2+, Mn2+ and Al3+ undergo hydrolysis, thereby consuming the released OH– ions and in turn releasing protons (H+)
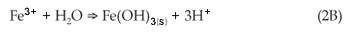
The protons released from the hydrolysis reactions offset the pH increase from the CaO dissolution and hence a buffer region, or pH plateau, was observed. The pH buffer region will only be overcome when all the AMD components have been fully hydrolysed. The time taken to overcome the buffer region also depended on the amount of FA in the mixture. The smallest AMD:FA ratio of 1:1 exceeded the buffer region in 10 minutes, whereas the 100:1 and 20:1 mixtures did not do so in the contact time investigated because of the insufficient neutralizing capacity of these mixtures (Fig. D). It should also be noted that the final pH attained also depended on the amount of FA in the mixture and increased with increase in FA. The buffer region was observed at a similar pH range for all mixtures, confirming the role played by the hydrolysis of the AMD components: Fe3+, Fe2+ and Al3+ undergo hydrolysis in the pH range 4–722 and the buffer region in these experiments was observed at pH 4.5–7. Another important observation was that the time taken to overcome the buffer region was inversely proportional to the amount of FA in the mixture: for the AMD:FA ratios 2:1 and 3:1 the buffer region lasted for nearly 120 minutes whereas for mixtures 4:1 and 10:1 the corresponding time was nearly 240 minutes (Fig. D). In the case of the 100:1 to 20:1 mixtures, the FA did not provide enough alkalinity to increase the pH of the solution to overcome this pH plateau in the reaction time tested.
The observed pH plateau indicate that after an initial stoichiometric neutralization, leading to an increase in pH to 4.5–6.5, the AMD components hydrolysed in the buffer zone, before the residual alkalinity of the FA allowed a further increase of pH. In addition, FA particles become coated ('armoured') with Fe and Al oxyhydroxides in the buffer region, preventing the neutralization reaction based on readily soluble species from continuing.33 The later pH increase may indicate that FA continued to release substantial alkalinity via the slow dissolution of its glassy phases.34 Abrasion of particles during continuous stirring may have enhanced this alkalinity release.35 Three factors thus seemed to dictate the pH and nature of the final solution obtained, namely, the amount of FA in relation to the volume of AMD, the quantity of hydrolysable constituents available in the AMD, and the reaction time.
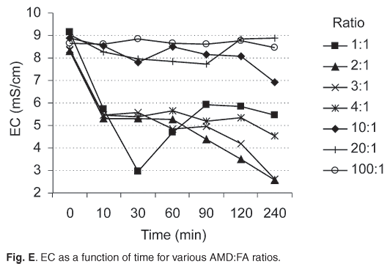
The variation of EC with time during neutralization reactions
Figure E online shows the variation in EC with time for different AMD: FA mixtures. High ratios (100:1 to 10:1) did not show any significant reduction in EC in the final water, as values remained between 7 and 9 mS/cm. This can be attributed mainly to the fact that, at such ratios, the pH did not exceed 5.5, and most AMD constituents remained in solution. Another factor may be the small amounts of FA added, so that the soluble species that would be released from the ash and thus their availability for reaction, were minimal. Ratios 4:1 to 2:1 showed a steady decrease in EC as contaminant species were removed from solution by precipitation or adsorption onto FA particles. For the lowest ratio (1:1), after a steep EC decrease in the first 30–60 minutes, a slight increase occurred. This can be attributed to the high FA content in the reaction mixture.
The factor that could lead to enhanced EC at high FA content was the dissolution of soluble salts on the surface of ash particles and dissolution of unreacted CaO, which contributed to increased concentration of unreacted OH– ions.
Vadapalli V.R.K., Klink M.J., Etchebers O., Petrik L.F., Gitari W., White R.A., Key D. and Iwuoha E. (2008). Neutralization of acid mine drainage using fly ash, and strength development of the resulting solid residues. S. Afr. J. Sci. 104, 317– 322.