Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Journal of Science
versão On-line ISSN 1996-7489
versão impressa ISSN 0038-2353
S. Afr. j. sci. vol.104 no.7-8 Pretoria Set./Ago. 2008
RESEARCH LETTERS
Comparison of lipid and fatty acid profiles of commercially raised pigs with laboratory pigs and wild-ranging warthogs
L-B. Fine; B.C. Davidson*
School of Physiology, University of the Witwatersrand Medical School, 7 York Road, Parktown 2193, South Africa
ABSTRACT
The perception that commercial livestock meat products have adverse nutritional effects, especially from the point of view of lipids and cardiovascular disease, is linked to assumptions concerning their inherent lipid profile. However, when commercially reared domestic pig meat samples were compared with those from laboratory pigs or wild-ranging warthogs, the warthog and laboratory pig samples shared similar lipid profiles, whereas the commercially reared pig samples were different. Pork is not inherently high in saturated fatty acids, unless this has been induced by the current intensive agricultural practices employed in raising the animal. We conclude that problems of high saturated fatty acid content in commercially raised pork could be ameliorated by appropriate changes to current intensive animal production methods.
Introduction
A high red meat intake continues to be popular in South Africa, despite awareness of the high incidence of coronary artery disease, correlated with this dietary habit.1 Globally, most commercially available meat is the product of intensive agricultural, and highly selective breeding methods, and many have blamed meat, at least in part, for the increase in cardiac disease-associated morbidity and mortality.2–7 Studies in Australia limiting saturated fat intake, but supplementing with lean meat that is comparatively rich in polyunsaturates, have shown a reduction in circulating cholesterol, paralleling results when fish oils are consumed.8–10 Thus, meat in general may not be the problem, but specifically the high saturated fat content present in intensively-produced meats.11–13
Before the emergence of agriculture, especially intensive agricultural practices, human dietary meats would have shown lipid profiles approximating those of wild meats, and thus would probably have displayed lower total lipid, but with higher proportions of polyunsaturates.12,14,15
Many reports in the literature document the lipid and fatty acid profiles of commercial meats arising from current agricultural practices, usually intensive in nature, aimed to fatten and bring livestock to market as quickly as possible.16–19 In contrast, animals reared for use in animal-based research need to be as 'natural' as possible, while maintaining a degree of genetic and phenotypic uniformity. These two contrasting requirements determine the diet to be fed, in each case. For the former, it is a diet that addresses rapid attainment of slaughter weight (optimized body-mass and time-frame product conversion of input costs from feed). For the latter, it is a diet that addresses the animal's need for healthily balanced nutrition. In the wild, the diet is usually more restricted in nature, and at times may constrain an animal's growth, and the animal will undergo significant exercise in the process of foraging. It is thus possible that significant health-related differences exist between commercial, laboratory, and wild animals of the same or similar species.
Laboratory animals are fed a more balanced, and usually more varied, spectrum of foods, whereas animals under intensive stock-farming are given high carbohydrate and possibly high fat diets. This diet may pro vide more saturated fat, which is less inclined to go rancid and thus has a longer shelf life. Moreover, excess carbohydrate has a tendency for con version to saturated fat by the animal's liver. This surplus fat is stored around abdominal organs, subcutaneously and as adipose tissue that in filtrates muscle fibres (marbling).
Animals in the wild forage for their diet, and will take in a wide range of foodstuffs that have a correspondingly wide range of lipid profiles, including a significant proportion of polyunsaturates. The latter are not stored as future energy sources, but preferentially utilized for membrane structure and eicosanoid synthesis.
They also stimulate the uncoupling of chain, inducing an increased metabolic rate, and elevated substrate catabolism.20,21
Materials and methods
Samples of pork from shoulder, midback and abdomen were obtained from three different retailers, to avoid the possibility that all material came from the same animal. Similar samples were also obtained from three euthanased pigs provided by Badenhorst & Co., Bronkhorstspruit, Northern Province, South Africa, via the University of the Witwatersrand Central Animal Service. Data from analyses of samples obtained from culled wild warthogs, carried out in previous research, were used for comparison.
Pork sample lipids were extracted using chloroform:methanol (2:1).22 A 1-ml aliquot of each extract was then used to determine lipid dry weight. A further aliquot equivalent to 20 mg of lipid was transmethylated using 10% boron trifluoride in methanol to prepare methyl esters of the fatty acids.23 These were then extracted into hexane, and the methyl esters separated on a Varian 3400 gas chromatograph, with a 10% SP2330 6' × ⅛" packed column and FID detection. Quantification was with a Varian 4270 integrator. The peaks were identified by comparison of retention times with FAME standards. The warthog samples were subjected to the same analytical protocol during the course of the previous work.
Results
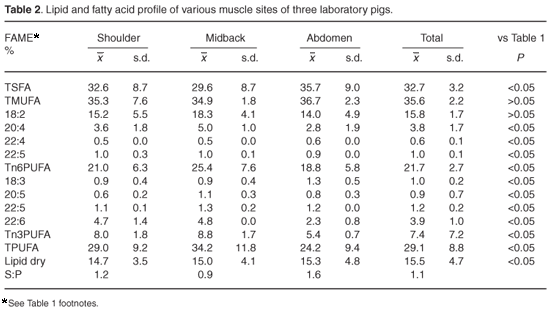
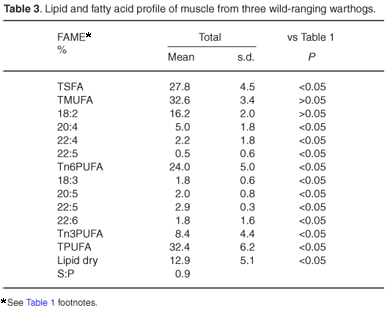
Table 1 shows the results from analysis of the pig samples from the various retail outlets, and these have been treated as the reference data for all statistical comparisons. Table 2 shows comparable results from the pigs of laboratory origin, whereas Table 3 illustrates the comparative data from the wild-caught warthogs.
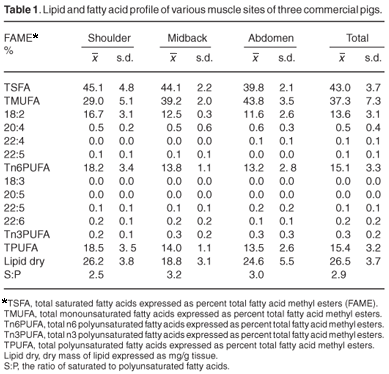
A large proportion of the commercial sample total fatty acids (Table 1) comprises saturates and monounsaturates (43.0% and 37.3% respectively), whereas only 15.4% are polyunsaturates. Within this fraction, 15.1% are n6 polyunsaturates, with only trace amounts of n3 polyunsaturates. The lipid dry mass is 26.5 mg/g wet mass. The mass ratio of saturates to polyunsaturates is 2.89:1. Whereas there was a moderate degree of variability between the samples from different anatomical sites, there are no significant differences between these sites for any of the fatty acids.
By comparison, the laboratory pig samples (Table 2) show somewhat lower total saturates (32.7%), similar monounsaturates (35.6%), and markedly higher polyunsaturates (29.1%). Within the polyunsaturates, amounts of n3 are significantly greater than those in Table 1 (7.4%). The n6 are also greater (21.7%). However, the total lipid dry mass is lower (15.5%) and the mass ratio of saturates to polyunsaturates is less at 1.06:1.
Discussion
There are only slight or insignificant compositional differences between the wild warthog and laboratory pig samples. We conclude from this that domestic pigs (when not intensively reared) are not inherently high in saturates or low in polyunsaturates and share a similarity with the related wild species. There are also only minor differences between these two groups in respect of their total lipid dry masses and saturatedto-polyunsaturated mass ratios. Information on the precise anatomical sampling sites from the warthogs was not available, but there is little variation between the samples. This could indicate that the sample sites were similar, or that there is little variation between different sites. The different sites sampled in the laboratory pigs also show small variability, except for variation in fatty acid profile between the shoulder and midback samples, and the abdominal samples. Differences are deemed to be insignificant.
Examination of the data from the retail meat samples shows striking differences, with the total saturates being markedly higher, the total monounsaturates similar, and the total, n6 and n3 polyunsaturates considerably lower. There is insignificant variation between muscle sites sampled. The mass ratio of saturates to polyunsaturates is greater, more than twice found in the laboratory pig samples, whereas the total lipid dry mass is also almost double that of the laboratory pigs. The retail meat has much more fat, with a greater proportion as saturates.
Laboratory pigs and warthogs, in contrast, show significantly lower levels of saturates compared with the stock-reared pigs, together with significantly higher total, n6 and n3 polyunsaturates, and significantly lower saturate-to-polyunsaturate mass ratios and total lipid dry masses. This reflects a lower total fat, together with a greater proportion of fat comprising polyunsaturates.
The fatty acid profiles reported above indicate that domesticated pig meat is not naturally high in saturates. The high levels, found by us and others,19 are a consequence of the intensive stock-farming practices used to accelerate animal growth to slaughter-weight. The trend is paralleled by lower proportions of polyunsaturates.
Given the prevailing dogma that high dietary saturated fatty acid intake is deleterious to health, especially in terms of cardiac and circulatory health,16,24–28 we judge current intensive pig-farming as prejudicial to consumer health. While we accept that commercial survival is predicated by optimized agro-economics, we believe that practices should not be to the detriment of the customer. Research is needed in the development of stock-farming methods that optimize agro-economic considerations without compromising the nutritional value of the product to the consumer.
We wish to thank the National Research Foundation of South Africa and the University of the Witwatersrand, Johannesburg, for their financial support of this project.
1. Love P., Maunder E., Green M., Ross F., Smale-Lovely J. and Charlton K. (2001). South African food-based dietary guidelines. S. Afr. J. Clin. Nutr. 14(1), 9–19. [ Links ]
2. Armstrong B., Clarke H., Martin C., Ward W., Norman N. and Masars J. (1979). Urinary sodium and blood pressure in vegetarians. Am. J. Clin. Nutr. 37, 755–762. [ Links ]
3. Bursten J., Schonfeld G., Howald M.A., Weidman S.W. and Miller J.P. (1978). Plasma apoprotein and lipoprotein lipid levels in vegetarians. Metabolism 27, 711–719. [ Links ]
4. Gutierrez Fuentes J.A. (1996). What food for the heart? World Health Forum 17, 157–163. [ Links ]
5. Hugo A., Osthoff G. and Jooste P.J. (1999). Effect of slaughter weight on the intramuscular fat composition of pigs. Proc. 45th Int. Congr. Meat Sci. Tech., Yokohama, 496–497. [ Links ]
6. Phillips R.L., Lemon F.R., Beeson W.L. and Kuzma J.W. (1978). Coronary heart disease mortality among Seventh Day Adventists with differing dietary habits: a preliminary report. Am. J. Clin. Nutr. 31, S191–198. [ Links ]
7. Sanders K., Johnson L. O'Dea K. and Sinclair A.J. (1994). The effect of dietary fat level and quality on plasma lipoprotein lipids and plasma fatty acids in normocholesterolemic subjects. Lipids 29(2), 129–138. [ Links ]
8. O'Dea K., Traianedes K., Chisholm K., Leyden H. and Sinclair A.J. (1990). Cholesterol-lowering effect of low-fat diet containing lean beef is reversed by the addition of beef fat. Am. J. Clin. Nutr. 52, 491–494. [ Links ]
9. Sachs F.M., Donner A. and Castelli W.P. (1981). Effect of ingestion of meat on plasma cholesterol of vegetarians. J. Am. Med. Assoc. 246, 640–644. [ Links ]
10. Watts G.F., Ahmed W., Quiney J., Houlston R., Jackson P., Iles C. and Lewis B. (1988). Effective lipid-lowering diets including lean meat. Br. Med. J. 296, 235–237. [ Links ]
11. Bull N.L., Day M.J.L., Bunt R. and Buss D.H. (1983). Individual fatty acids in the British household food supply. Hum. Nutr. Appl. Nutr. 37A, 373–377. [ Links ]
12. O'Dea K. (1984). Marked improvement in carbohydrate and lipid metabolism in diabetic Australian aborigines after temporary reversion to traditional lifestyle. Diabetes 33, 596–603. [ Links ]
13. Sinclair A.J., O'dea K., Dunstan G., Ireland P.D. and Niall M. (1987). Effects of plasma lipids and fatty acid composition of very low fat diets enriched with fish or kangaroo meat. Lipids 22, 523–529. [ Links ]
14. Cordain L., Watkins B.A., Florant G.L., Kehler M., Rogers L. and Li Y. (2002). Eur. J. Clin. Nutr. 56, 181–191. [ Links ]
15. Sinclair A.J. and O'Dea K. (1991). In Fats in Human Diets Through History: Is the western diet out of step? eds J.D. Wood and A.V. Fisher, pp. 1–47. Elsevier, London. [ Links ]
16. Enser M., Hallet K., Hewitt B., Fursey G.A.J. and Wood J.D. (1996). Fatty acid content of English beef, lamb and pork at retail. Meat Sci. 42(4), 443–456. [ Links ]
17. Crawford M.A. (1968). Fatty acid ratios in free-living and domestic animals: possible implications for atheroma. Lancet 22, 1329–1333. [ Links ]
18. Crawford M.A., Gale M.M., Woodford M.H. and Casped N.M. (1970). Comparative studies on fatty acid composition of wild and domestic meats. Int. J. Biochem. 1, 295–305. [ Links ]
19. McCance R.A. and Widdowson E.M. (1978). In The Composition of Foods, 4th edn, eds A.A. Paul and D.A.T. Southgate, p. 295. H.M.S.O., London. [ Links ]
20. Beck V., Jaburek M., Demina T., Rupprecht A., Porter R.K., Jezek P. and Pohl E.E. (2007). Polyunsaturated fatty acids activate human uncoupling proteins 1 and 2 in planar lipid bilayers. FASEB J. 21, 1137–1144. [ Links ]
21. Rodriguez V.M., Portillo M.P., Pico C., Macarulla M.T. and Palou A. (2002). Olive oil feeding up-regulates uncoupling protein genes in rat brown adipose tissue and skeletal muscle. Am. J. Clin. Nutr. 75, 213–220. [ Links ]
22. Folch J., Lees M. and Sloane-Stanley G.H (1957). A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 226, 497–509. [ Links ]
23. Moscatelli E.A. (1972). Methanolysis of cerebrosides with boron trifluoridemethanol. Lipids 7, 268–271. [ Links ]
24. Hu F.B., Manson J.E. and Willett W.C. (2001). Types of dietary fat and risk of coronary heart disease: a critical review. J. Am. Col. Nutr. 20(1), 5–19. [ Links ]
25. Li D., Ng A. Mann N.J. and Sinclair A.J. (1998). Contribution of meat fat to dietary arachidonic acid. Lipids 33(4), 437–440. [ Links ]
26. Phillips R.L., Beeson W.L., Kuzma J.W. and Lotz T. (1980). Influence of selection versus lifestyle on risk of fatal cancer and cardiovascular disease among Seventh Day Adventists. Am. J. Epidemiol. 112, 296–314. [ Links ]
27. Sachs F.M., Castelli W.P., Donner A. and Kass E. (1975). Plasma lipids and lipoproteins in vegetarians and controls. N. Engl. J. Med. 292, 1148–1151. [ Links ]
28. Snowdon D.A, Phillips R.L. and Frazer G.E. (1984). Meat consumption and fatal ischemic heart disease. Prev. Med. 13, 490–500. [ Links ]
Received 6 July 2007. Accepted 15 August 2008.
* Author for correspondence. E-mail: bruce.davidson@wits.ac.za














