Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Science
versión On-line ISSN 1996-7489
versión impresa ISSN 0038-2353
S. Afr. j. sci. vol.104 no.5-6 Pretoria may./jun. 2008
RESEARCH LETTERS
Middle Miocene vertebrate fauna from Pemba Island, Tanzania
Martin Pickford
Collège de France, UMR 5143 du CNRS, Case postale 38, 8, rue Buffon, 75005 Paris, France. E-mail: pickford@mnhn.fr
ABSTRACT
This paper records the presence of Middle Miocene terrestrial and marine vertebrates in sedimentary deposits on Pemba Island in the Indian Ocean, offshore Tanzania. Although the fossil assemblage is restricted, the affinities of the terrestrial mammals are typical of African continental faunas of basal Middle Miocene age, c. 16.5 million years ago. The Pemba Series has been reported to be of Lower–Middle Miocene age on the basis of marine invertebrates, in agreement with the age estimates based on a mandible of Libycochoerus massai from the deposits. The discovery is important because it provides a potential means of checking correlations between marine and continental biochronological time scales in a part of the world that has previously yielded no evidence of terrestrial palaeontology.
Introduction
The large islands offshore Tanzania were geologically surveyed during the 1950s by British Petroleum. During the surveys, P.E. Kent and his colleague R.E. Linton collected several vertebrate fossils from Pemba and Zanzibar islands, embedded in blocks of calcareous deposits rich in marine invertebrate micro- and macrofauna. The fossils, principally the microfauna, indicated correlation of the Pemba Series with the Lower–Middle Miocene (Kent letter to A.T. Hopwood, 1953).1
At the end of April 1953, the mammalian fossils were forwarded to A.T. Hopwood at the British Museum of Natural History (now the Natural History Museum, London), who arranged for them to be incorporated into the mammal collections of that institution. In a letter accompanying the fossils, P.E. Kent wrote that the fossils 'are of interest partly for dating purposes (although this mainly relies on micro-faunal evidence) and also for their relations to the faunas from continental beds inland'.
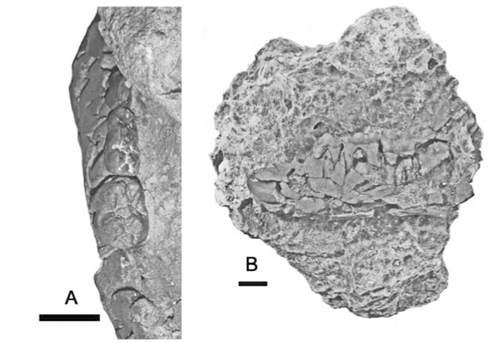
The fossils were collected from three localities; two on Pemba Island (Fig. 1) and one on Zanzibar. The Pemba locality at Chwaka, found by R.E. Linton, yielded a juvenile suid lower jaw containing elements of the deciduous and permanent dentition (Fig. 2), a large ruminant third phalanx, fragments of sirenian ribs and a trionychid scute, all of a brownish colour, whereas the Weti locality, found by P.E. Kent, yielded two crocodile teeth and a ruminant upper third premolar from grey clays on the beach (Fig. 2). The specimens from this site are shiny and black. Both these localities were estimated by Kent (letter to Hopwood dated 30 April, 1953) to be Lower–Middle Miocene in age.2,3 The M'bweni locality on Zanzibar, sampled by Kent, yielded several sirenian ribs from the top of the Chlamys werthi beds, at the time considered to be of Pliocene age. Later, the strata mapped on Zanzibar as Chlamys werthi beds were realized to span much of the Miocene as well as the Pliocene1,4 but the M'bweni deposits were retained in the Pliocene. A possible equid tooth from the site (label accompanying the fossil) is in fact a fragment of broken sirenian rib.

As far as I am aware, nothing detailed has ever been published about the mammalian and other vertebrate fossils from Pemba Island. The aim of this paper is to describe and interpret the Pemba fossils, and to examine their biochronological and palaeobiogeographic affinities, as outlined by Kent in his letter to Hopwood.
Geology
The detailed field context of the Pemba vertebrate fossils is not known. The Chwaka specimens are embedded in a pale cream to yellowish, poorly sorted calcareous deposit with small reddish patches here and there. An unpublished report of the locality indicates that it is on a islet half a mile (0.8 km) south of Tumbe just beneath high tide level in a grey sandy limestone containing Tubecellara (A. Belopolsky, pers. comm.). Kent's letter to Hopwood indicates that the pig jaw was collected from the top of the Pemba Series. The context of the Weti fossils was described as being Pemba Series, while unpublished notes by Kent indicate that the deposit was a grey clay at the head of the beach at Weti Creek. The Pemba Series comprises a lower unit, the fossiliferous Chake-Chake Beds overlain by the unfossiliferous Weti Beds.3
Given that calcareous deposits are prone to karstification, it is necessary to keep in mind the possibility that the deposit in which the Chwaka fossils are embedded may have accumulated in a karst pocket, and could thus post-date the Pemba Series. Karst processes are known to have affected large areas of Pemba Island (Kiuyu, Kojani and Kiweni districts)3 and it is possible that in the past some of the karst depressions may have acted as traps for animal remains. The presence of reddish discolouration in the deposit that embeds the suid jaw is suggestive of such a possibility, but clearly it is necessary to rediscover the outcrop to clarify the original depositional context of the fossils.
The Weti fossil assemblage differs from the Chwaka one, not only in the colour of the fossils (black instead of light brown) but also by being slightly rolled and polished. The polishing could have been produced either at the time of fossilization or when the specimens eroded out of the sediments. As with the Chwaka material, it is necessary to relocate the deposits which yielded the Weti fossils in order to ascertain their primary depositional context.
Interpretation
At the time that the Pemba and Zanzibar fossils were sent to the Natural History Museum, they would have been extremely difficult to interpret according to the request outlined in Kent's letter to Hopwood. This is because in the early 1950s almost nothing was known about Middle Miocene African fossil mammals. The only basal Middle Miocene localities known in the continent at the time were Maboko Island and Moruorot, Kenya, and Wadi Moghara, Egypt, but the suids and ruminants from these sites had not yet been described in detail, and in any case are poorly represented even today.5,6 The key to interpreting the Pemba fossils was discovered in 1961 at Gebel Zelten, Libya.7 The Libyan locality yielded a rich and well-preserved suid fauna5–9 and a good sample of ruminants,10,11 but by the late 1950s the Pemba fossils were lying neglected in a drawer, where they have lain ever since.
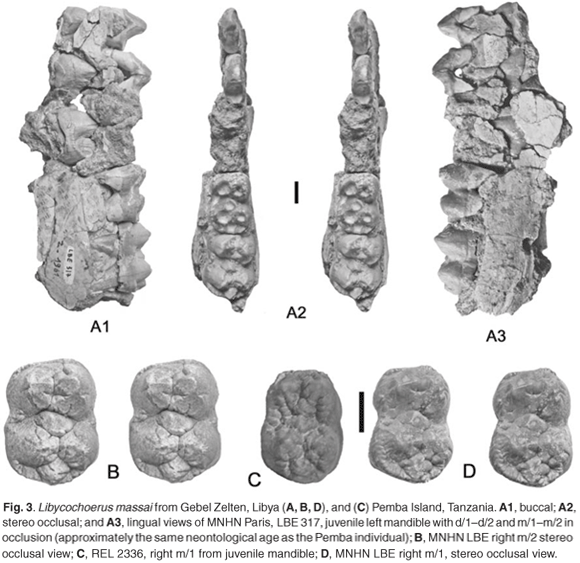
In February 2008, the author was browsing through the mammal collections at the Natural History Museum. On opening a drawer labelled Zanzibar, he realized that in it was a mandible embedded in a block of pale cream to yellowish limestone from Chwaka (labelled REL 2336), which was reminiscent of remains of Libycochoerus massai from Gebel Zelten,8,9,12,13 a terminal Early Miocene to basal Middle Miocene locality in Libya.14–16 Detailed study of the Chwaka specimen reveals that this is indeed the case, the deciduous and permanent dentition agreeing in both morphology and dimensions with the excellent sample of this species from Gebel Zelten5,6 (Figs 2–4).
Examination of the ruminant premolar (PEK 5559) from Weti (now known as Wete) (identified on the accompanying label as a suid tooth) reveals that it is close in dimensions and morphology to teeth of Canthumeryx syrtensis, a species defined at Gebel Zelten.10,11 However, since the specimen consists only of the buccal half of the tooth, there remains some doubt about this identification.
Systematic descriptions
Family Crocodylidae Cuvier, 1807 Genus indet.
Description and discussion
The sample labelled PEK 5559 from Weti contains two crocodile teeth. One possesses well-developed fluting, the other, which is slightly rolled, has abraded fluting. The teeth are typically crocodyliform, and are essentially unidentifiable at the generic level, although it is possible to conclude that they do not represent the genus Euthecodon, which possesses taller, sharper teeth.
Family Trionychidae Fitzinger, 1826 Genus indet.
Description and discussion
In sample REL 2336, there is a scute of a trionychid turtle attached to a small block of limestone similar to that in which the suid jaw is embedded. The scute has a dorsal surface adorned with small pimples of bone.
Class Mammalia Linnaeus, 1758
Order Sirenia Illiger, 1811
Genus and species indet.
Description and discussion
In a small block of limestone similar in composition to that in which the suid jaw is embedded, there is a fragmentary osteosclerotic rib typical of sirenians. It is not possible to identify such a fragmentary specimen, but the features of the bone are clearly those of a sirenian.
Order Artiodactyla Owen, 1848
Family Suidae Gray, 1821
Subfamily Kubanochoerinae Gabunia, 1958
Genus Libycochoerus Arambourg, 1961
Species Libycochoerus massai Arambourg, 1961
Description
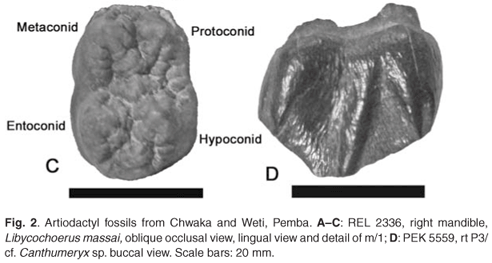
REL 2336 is a juvenile suid right mandible currently in a large block of poorly sorted pale yellowish marine limestone (Fig. 2A, 2B). The exposed lingual surface of the jaw is heavily fractured, but the pieces are more or less in their correct anatomical position. Teeth in occlusion are the d/3, d/4 and m/1, whereas the canine, p/3, p/4 and m/2 are in their crypts. Alveoli of a relatively large d/2 are present in front of the d/3. The crypt of the m/3 is partly preserved, but the tooth itself had fallen out before the fossil was collected. This individual was therefore probably unweaned at the time of death. The crowns of the deciduous premolars are in medium wear, and the m/1 is unworn, meaning that the detailed occlusal morphology of the tooth is well preserved (Fig. 2C). Judging from the shape and dimensions of the unerupted canine crown, the individual was a female.
Deciduous dentition. There are two prominent alveoli in front of the d/3, presumably for a large d/2. The d/3 is a single cusped slender tooth with the apex of the cusp above the gap between the two roots. The anterior crest is slightly concave in lingual view, and distally there is a swelling which almost forms a distal cusplet, but the details of the shape of the cusplet have been eradicated by wear. The enamel is thin.
The d/4 is trilophid with five roots, three buccal and two lingual, as is usual in artiodactyls. The anterior lophid is deeply worn, leaving little to describe. The second lophid is in medium wear and shows two cuspids, each of which sports anteriorly and posteriorly directed grooves and one leading towards the centre of the tooth. There is a prominent median accessory cusplet. The third lophid is lightly worn, and shows two cusps, each with grooves anteriorly, posteriorly and centrally orientated, and there is a prominent posterior accessory cusplet in the centre line of the tooth. There are remnants of a buccal cingulum in the ends of the anterior and posterior transverse valleys and the base of the buccal cusps is slightly swollen, but not to the extent of forming a cingulum. The lingual notches are moderately narrow.
Permanent dentition. The canine is still in its crypt, and only the lingual aspect is exposed. The crown is low for a canine, and it is hook shaped in profile and verrucosic in section (as far as can be judged from the visible parts).
The m/1 is unworn (Figs 2C, 3C), having erupted almost to occlusal level. There are four main cusps disposed in two lophids, an anterior one comprising the protoconid and metaconid, and a posterior one comprising the entoconid and hypoconid. The cusps are bundont, with prominent but not deep furchen. The preprotocristid and premetacristid are short and descend steeply towards the anterior accessory cusplet. The postprotocristid and postmetacristid are beaded and are directed distally and slightly towards the centre of the tooth. The inner surface of the protoconid and metaconid show accessory grooves, which on the metaconid are deep enough to individualize low cusplets near the centre line of the tooth. Between the apex of the metaconid and the median accessory cusplet, there is an additional cusplet (the 2–3 cusplet17), which is strongly joined to a crest which leads from the apex of the metaconid obliquely distally towards the median accessory cusplet. The 2–3 cusplet is almost as large as the median accessory cusplet. The internal surface of the entoconid is subdivided by two grooves which outline two incipient cusplets medially and disto-medially, the latter positioned opposite the posterior accessory cusplet (hypoconulid). The postentocristid and posthypocristid are beaded and descend steeply onto the posterior cingulum, which lies distal to the posterior accessory cusplet. The anterior, median and posterior accessory cusplets are low and bundont with wrinkled enamel. There is a beaded cingular remnant in the buccal end of the median transverse valley and the bases of the buccal cusps (protoconid and hypoconid) are swollen, but not to the extent of forming a cingulum. The anterior cingulum is finely beaded and is located below the anterior accessory cusplet. The rear lophid is slightly broader than the mesial one.
Only the lingual and radicular aspects of the m/2 are visible in its crypt. The tooth has not yet formed any roots, so the inside of the pulp cavities is visible. Only part of the crypt of the m/3 is preserved, the tooth itself having fallen from the specimen prior to collection.
Measurements of the teeth, in mm, are as follows: d/3 20.2 × 10e (e = estimate); d/4 29.6 × 14.4; m/1 27.8 × 19; m/2 31.8 × –.
Discussion
The Chwaka suid mandible is of a large suid with bunodont cheek teeth. Bunodont listriodonts have molars that tend to show the beginnings of lophodonty, do not generally possess swellings at the base of the buccal cusps and in any case are smaller than those of the Chwaka mandible (Fig. 4), and their deciduous teeth are different (smaller, broader distally). The molars of potamochoeres and bunodont suines have taller, more parallel-sided cusps with better expressed, deeper furchen, no sign of basal swelling of the buccal cusps and are also generally smaller than the Chwaka specimen. Bunodont nyanzachoeres (Fig. 4) such as Nyanzachoerus tulotos possess markedly divergent morphology of the deciduous teeth (premolars larger than the first molar, inflated cusps), and the anterior premolars are small, not large as suggested by the alveoli of the d/2 preserved in the Chwaka mandible. The only subfamily of suids with the combination of features of the Chwaka specimen is the Kubanochoerinae; among members of this subfamily, the dimensions of the teeth accord only with Libycochoerus massai (Fig. 4).18
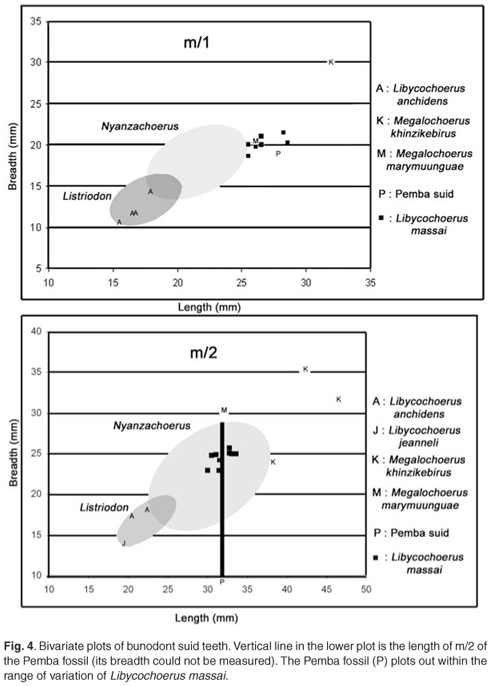
The deciduous lower teeth are so similar in shape and dimensions to those of Libycochoerus massai from Gebel Zelten, that there is little hesitation in identifying the Pemba mandible as such. In particular the detailed morphology of the m/1 with buccal swelling of the base of the crown, the shape and position of the anterior, median and posterior accessory cusplets, the position and relatively voluminous dimensions of the 2–3 cusplet and the heavily wrinkled enamel, all evoke this species. The dimensions of the lower first and second molars and the d/3 lend support to this identification.
Suborder Ruminantia Scopoli, 1777
Genus cf. Canthumeryx Hamilton, 1973
Species indet.
Description
Only the buccal part of the right P3/, PEK 5559 from Weti, is preserved (Fig. 2D). There are three swollen styles on the buccal surface of the tooth, an anterior one that pinches out to a point at the margin of the crown. The height of the style is about two-thirds the total height of the crown. There is a large central style that reaches the apex of the tooth, and a lower posterior style about half the total crown height. The depressed space between the anterior and central styles is sharply triangular, whereas the central and posterior styles are more or less parallel to each other and enclose a flattish depressed area. The surface of the enamel is lightly wrinkled. There is a basal swelling, but no signs of a cingulum. There appear to have been two buccal roots.
Although this tooth is fragmentary, its ruminant affinities are immediately apparent, including the morphology of the buccal wall and its wrinkled enamel. Among ruminants the tooth appears to be closer to giraffoids and climacoceratids than to bovids or other groups. The specimen is close in morphology and size to teeth of Canthumeryx syrtensis, but clearly, there are a number of other ruminants that it could belong to. Nevertheless it would not look out of place in a Middle Miocene assemblage.
The P3/ measures 27.8 mm anteroposteriorly.
Post-cranial skeleton. In the sample numbered REL 2336 from Chwaka, there is a third phalanx of a large ruminant embedded in a small block of limestone similar to that holding the suid jaw. The phalanx is medio-laterally narrow with a steeply angled articular facet for the distal articulation of the second phalanx, the plantar surface is narrow, and the dorsal ridge is high. This phalanx is compatible in size with the P3/ described above, but clearly it could belong to a number of different taxa.
Discussion
The Pemba Series is an accumulation of marine deposits of terminal Early Miocene to basal Middle Miocene age, cropping out on Pemba Island in the Indian Ocean, 60 km offshore the Kenya–Tanzania frontier (Fig. 1).1,3,4 The presence of crocodiles, trionychid turtles and land mammals in the deposits indicates that, at the time of deposition, dry land with freshwater bodies was not far from the localities that yielded the vertebrates. This suggests that the deposits may have accumulated in lagoons or a similar nearshore environment, although accumulation in a karst setting cannot be ruled out, at least for the Chwaka deposit. The Weti grey clays likely accumulated in paludal or lagoonal quiet water conditions.
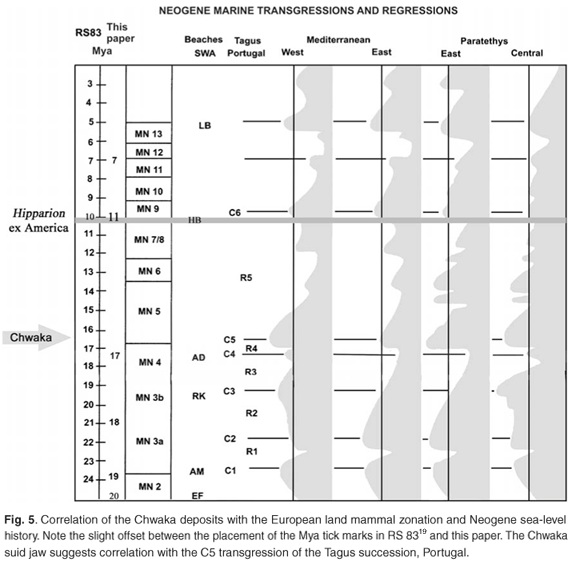
Crocodiles and trionychids are extremely widespread geographically and geochronologically and, being aquatic, would be expected to occur on islands such as Pemba and Zanzibar, which are not far removed from continental shores and are close to the equator. The mammals from Pemba, in contrast, being terrestrial, are potentially more informative about geochronology and palaeobiogeography. Libycochoerus massai and Canthumeryx sp. have been recorded previously only from deposits that accumulated about 16 ± 1 million years ago (Mya) in mainland Africa,16, 20 although related lineages are known from the Indian subcontinent and mainland Eurasia17 (Fig. 5). The suid from Chwaka, in particular, is most informative about age, as there is little chance that it could belong to a taxon other than Libycochoerus massai. The latter species is known from Gebel Zelten, the type locality, and from Wadi Moghara, Egypt, in deposits equivalent in age to MN 05 in Europe.21 Although the species has not previously been reliably reported from East Africa, it is intermediate in size between Libycochoerus jeanneli from Rusinga (17.8 Mya)22–24 and Moruorot, Kenya (17.2 Mya),25 on the one hand, and Libycochoerus khinzikebirus from Gebel Zelten, Libya, Cheparawa, Kenya (14.5 Mya),16 and Maboko, Kenya (14–16 Mya)25 on the other. It is therefore likely that the species Libycochoerus massai existed about 16 ± 1 Mya.16 Its identification at Pemba Island represents a major extension of its known geographic range, and accords with the geochronological age estimate of the strata based on marine organisms.
A debate was fuelled before and during the Second World War by the contrast between the close resemblance of the extant terrestrial fauna of Pemba with that of the African mainland opposite, suggesting, on the one hand, a Pleistocene to Recent isolation of the island fauna from its mainland counterparts,26 and on the other, the available geological evidence which indicated a much earlier (Miocene) separation of the island from the continent.3 This debate remains unresolved, despite further geological surveys1,4 which supported a Miocene separation of the island from the continent. Kent et al.4 considered that the 800-metre-deep Pemba Channel that separates the island from the Tanzania mainland is probably the site of a graben that came into play perhaps during the Pliocene.
A distance of 60 km between the island and the mainland is perhaps insufficient to form a significant faunal barrier over geological time spans, especially given the seasonally alternating directions of the ocean currents in the Pemba Channel (southwest monsoon drift during the northern summer and northeast monsoon drift during the northern winter),27 which favour transportation of terrestrial animals (on floating islands, for example) from the mainland towards the island, both from the north and from the south. Furthermore, Zanzibar Island to the south of Pemba is separated from the mainland by a shallower sea floor, which may have been exposed during Pleistocene low sea stands, and this would provide an alternative passage for continental faunas to approach Pemba from the south.
For comparison, Madagascar, which has a largely endemic terrestrial fauna, is 430 km from the African mainland, yet during the Pleistocene mammals such as hippopotamids and the suid Potamochoerus managed to colonize it from Africa.
Conclusions
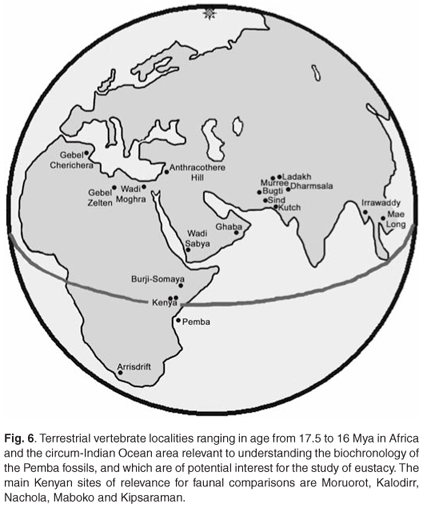
The identification of terrestrial mammals in strata exposed on Pemba Island, 60 km offshore Tanzania mainland, is important for several reasons, two of which were evoked by P.E. Kent in a letter that he wrote to A.T. Hopwood in 1953, which accompanies the specimens in their drawer at the Natural History Museum, London. They throw light on the age of the deposits and reveal that the island was inhabited by taxa that occurred on the African mainland during the base of the Middle Miocene. Even though the currently available fossil assemblage from Pemba is not very diverse, and the detailed geological context is uncertain (were the fossils within the Pemba Series, or in karst pockets developed in the Pemba Series?), it indicates that further palaeontological research in the region is warranted. Deposits yielding fossil vertebrates of similar age are extremely poorly known in Africa (Fig. 6), there being one locality in southern Africa (Arrisdrift, which is slightly older, c. 17.5 Mya), a handful in East Africa (Kipsaraman, Maboko, Moruorot, Nachola and Kalodirr in Kenya (17.2–14.5 Mya);28 Moroto in Uganda (c. 17.5 Mya);29 Burji-Somaya in Ethiopia);30 and North Africa (Wadi Moghara, Egypt;32 Gebel Zelten, Libya;12 Gebel Cherichera, Tunisia25).
Second, the discovery greatly extends the known geographic range of the suid species Libycochoerus massai, hitherto known only from the northern parts of the African continent. The island of Pemba is postulated to have separated from Africa during the Late Miocene3 or Pliocene;4 if this is so, then it was presumably close to, or even part of, Africa at the time of deposition of the Middle Miocene Chake-Chake Beds (Lower Pemba Series) and the Weti Beds (Upper Pemba Series).
Third, the Pemba vertebrate assemblage is important in that it provides a rare opportunity to compare directly the terrestrial African biochronological scale25,31 with the marine one33 (Fig. 5). On the basis of the marine microfauna, Eames and Kent1 determined the age of the Pemba Series as Lower–Middle Miocene (Burdigalian–Aquitanian), and this is confirmed by the recognition that the suid from Chwaka, and possibly the ruminant from Weti (now known as Wete), belong to species typical of the base of the Middle Miocene in mainland Africa. Clearly, there is potential in the Pemba deposits for improving the accuracy of correlations between marine and continental time scales, about which there is a great deal of uncertainty, especially for the period MN 03 to MN 06 (Burdigalian to Helvetian).25 Allied to biochronology is the potential of the deposits for throwing light on Early to Middle Miocene eustatic history (Fig. 6).
Fourth, in view of the phenomenon of insularity that greatly affected evolution of terrestrial faunas on Mediterranean and other islands during the Neogene,34 it would be interesting to determine whether similar evidence occurs on Pemba. Available fossils from the island do not currently demonstrate any peculiarities, but this could be because of the restricted nature of the Middle Miocene palaeontological sample and the fact that the island was possibly close to, or part of, the African continent at the time of deposition.
I thank Andy Currant for providing access to the fossil mammal collections at the Natural History Museum, London, and Andrei Belopolsky of British Petroleum for locating the archives that indicate the precise localities of the two faunal sites on Pemba.
1. Eames F.E. and Kent P.E. (1955). Miocene beds of the East African coast. Geol. Mag. 92, 338–344. [ Links ]
2. Davies A.M., Cox L.R., Stockley G.M., Stubblefield C.J. and White E.I. (1927). Report on the Palaeontology of the Zanzibar Protectorate, based mainly on the Collection made by G.M. Stockley, Government Geologist, 1925-1926. Zanzibar Government Report. [ Links ]
3. Stockley G.M. (1942). The geology of the Zanzibar Protectorate and its relation to the East African mainland. Geol. Mag. 79(4), 233–240. [ Links ]
4. Kent P.E., Hunt J.A. and Johnstone D.W.( 1971). The geology and geophysics of coastal Tanzania. Institute of Geological Sciences (London), Geophysical Paper 6, 1–101. [ Links ]
5. Pickford M. (1986). A revision of the Miocene Suidae and Tayassuidae of Africa. Tertiary Res. Spec. Pap. 7, 1–83. [ Links ]
6. Van der Made J. (1996). Listriodontinae (Suidae, Mammalia), their evolution, systematics and distribution in time and space. Contrib. Tert. Quatern. Geol. 33, 1–254. [ Links ]
7. Arambourg C. and Magnier P. (1961). Gisements de vertébrés dans le bassin tertiaire de Syrte (Libye). C. R. Acad. Sci. Paris 252, 1181–1183. [ Links ]
8. Arambourg C. (1961). Note preliminaire sur quelques vertébrés nouveaux du Burdigalien de Libye. C. R. Somm. Soc. géol. Fr. 4, 107–108. [ Links ]
9. Arambourg C. (1963). Le genre Bunolistriodon. Bull. Soc. géol. Fr. 5, 903–911. [ Links ]
10. Hamilton W.R. (1973). The lower Miocene ruminants of Gebel Zelten, Libya. Bull. Br. Mus. (Nat. Hist.) Geol. 21, 75–150. [ Links ]
11. Hamilton W.R. (1978). Fossil giraffes from the Miocene of Africa and a revision of the phylogeny of the Giraffoidea. Phil. Trans. R. Soc. Lond. B283, 165–229. [ Links ]
12. Pickford M. (1991). Biostratigraphic correlation of the middle Miocene mammal locality of Jabal Zaltan, Libya. In The Geology of Libya, ed. M.J. Salem, 4, 1483–1490. Elsevier, Amsterdam. [ Links ]
13. Wilkinson A. (1976). The lower Miocene Suidae of Africa. Foss. Vert. Afr. 4, 173–282. [ Links ]
14. Savage R. and White M.E. (1965). Two mammal faunas from the early Tertiary of central Libya. Proc. Geol. Soc. Lond. 1623, 89–91. [ Links ]
15. Savage R. and Hamilton W.R. (1973). Introduction to the Miocene mammal faunas of Gebel Zelten, Libya. Bull. Br. Mus. (Nat. Hist.) Geol. 22, 514–527. [ Links ]
16. Pickford M. (2001). The biochronological importance of Kubanochoerinae (Mammalia, Suidae) together with a description of new material of Megalochoerus khinzikebirus and Libycochoerus massai from Kenya. C. R. Acad. Sci. Paris 332, 193–201. [ Links ]
17. Pickford M. (1988). Revision of the Miocene Suidae of the Indian subcontinent. Münchner Geowiss. Abh. 12, 1–91. [ Links ]
18. Pickford M. (1993). Old World suoid systematics, phylogeny, biogeography and biostratigraphy. Paleontologia i Evolucio 26–27, 237–269. [ Links ]
19. Rögl F. and Steininger F. (1983). Vom Zerfall des Tethys zu Mediterran und Paratethys. Annal. Naturhist. Mus., Wien, 85/A, 135–163. [ Links ]
20. Pickford M.(1988). Geology and fauna of the Mid-Miocene Muruyur Beds, Baringo District, Kenya. Hum. Evol. 3, 381–390. [ Links ]
21. Pickford M., Miller E. and El-Barkooky A.N. (in press). Early Miocene Suidae and Sanitheriidae from Wadi Moghra, Egypt. Journal of Vertebrate Paleontology. [ Links ]
22. Drake R., Van Couvering J.A., Pickford M., Curtis G. and Harris J.A. (1988). New chronology for the early Miocene mammalian faunas of Kisingiri, Western Kenya. J. Geol. Soc. (London) 145, 479–491. [ Links ]
23. Pickford M. (1981). Preliminary Miocene mammalian biostratigraphy for western Kenya. J. Hum. Evol. 10, 73–97. [ Links ]
24. Pickford M. (1983). Sequence and environments of the lower and middle Miocene hominoids of Western Kenya. In New Interpretations of Ape and Human Ancestry, eds R. Ciochon and R. Corruccini, pp. 421–439. Plenum, New York. [ Links ]
25. Pickford M. (1998). Dating of the Neogene Old World anthropoid fossil record: essential base for phylogenetic analysis, biogeography and palaeoecology. Primatologie 1, 27–92. [ Links ]
26. Moreau R.E. and Pakenham R.H.W. (1941). The land vertebrates of Pemba, Zanzibar and Mafia: a zoo-geographical study. Proc. Zool. Soc. London, Series A, 110, parts 3 and 4, 1940. [ Links ]
27. Bartholomew J. (1957). The Advanced Atlas of Modern Geography. Meiklejohn, London. [ Links ]
28. Pickford M., Ishida H., Nakano Y. and Yasui Y. (1999) (cover mis-dated 1987). The Middle Miocene Fauna from the Nachola and Aka Aiteputh Formations, northern Kenya. Afr. Stud. Monogr. Suppl. Issue, 5, 141–154. [ Links ]
29. Pickford M. and Mein P. (2006). Early Middle Miocene mammals from Moroto II, Uganda. Beitr. Paläont. (Wien), 30, 361–386. [ Links ]
30. Suwa G., White T.D., Asfaw B., WoldeGabriel G. and Yemane T. (1991). Miocene faunal remains from the Burji-Somaya area, Amaro Horst, southern sector of the main Ethiopian Rift. Palaeont. afr. 28, 23–28. [ Links ]
31. Pickford M. and Morales J. (1994). Biostratigraphy and palaeobiogeography of East Africa and the Iberian Peninsula. Palaeogeogr. Palaeoclimatol. Palaeoecol. 112, 297–322. [ Links ]
32. Miller E. (1999). Faunal correlation of Wadi Moghara, Egypt: implications for the age of Prohylobates tandyi. J. Hum. Evol. 36, 519–533. [ Links ]
33. Harzhauser M., Kroh A., Mandic O., Piller W.E., Göhlich U., Reuter, M. and Berning B. (2007). Biogeographic responses to geodynamics: a key study all around the Oligo-Miocene Tethyan Seaway. Zoologischer Anzeiger 246, 241–256. [ Links ]
34. Reumer J.W.F. and de Vos, J. (eds) (1999). Elephants have a snorkel! Papers in honour of Paul Y. Sondaar. Deinsea 7, 1–420. [ Links ]
Received 4 March. Accepted 20 May 2008.