Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.104 n.5-6 Pretoria May./Jun. 2008
RESEARCH LETTERS
Palaeobiological implications of the bone microstructure of South American traversodontids (Therapsida: Cynodontia)
Anusuya ChinsamyI, *; Fernando AbdalaII
IZoology Department, University of Cape Town, Private Bag, Rondebosch 7701, South Africa
IIBernard Price Institute for Palaeontological Research, University of the Witwatersrand, Private Bag 3, WITS 2050, South Africa
ABSTRACT
We document the bone microstructure of traversodontids from Argentina and Brazil and make detailed comparisons of the bone microstructure of these cynodonts with that of other traversodontids, nonmammaliaform therapsids (including cynodonts), and Mesozoic mammalian taxa. Our analysis provides information on traversodontid ontogenetic age, growth patterns and life-style adaptations. The osteohistological data derived from the current study, as well as data on other nonmammaliaform cynodonts, provide an understanding of the evolution of life-history patterns among the therapsids. We show that many of the nonmammaliaform cynodonts (including some traversodontids) formed bone at rapid rates but retained flexible developmental growth patterns. In addition, although we observe a trend of increasingly rapid osteogenesis among the traversodontids, our findings do not discern a particular bone tissue type as a synapomorphy of the clade. This reinforces earlier hypotheses that the microscopic structure of bone is influenced by several intrinsic and extrinsic factors, and is not constrained by phylogeny alone.
Introduction
Over the past several years, there has been a growing interest in using bone microstructure to interpret aspects of the biology of extinct animals.1–4 Although investigations of the bone microstructure of nonmammaliaform therapsids began fairly early,5–9 in recent years there has been a surge of such studies on nonmammaliaform therapsids from the Karoo Basin of South Africa (dicynodonts,10–12 cynodonts,13–16 and gorgonopsians and therecephalians16). Even more recently, studies of fossil bone microstructure have been undertaken in five Mesozoic mammaliaform taxa.17 However, it is noteworthy that except for an earlier study by De Ricqlès,8 the bone microstructure of traversodontid cynodonts, which are the most diverse group of nonmammaliaform cynodonts, has been poorly sampled.
The study reported here documents the bone microstructure of three South American traversodontid cynodonts from Argentina (Table 1): Andescynodon mendozensis Bonaparte;18 Masseto- gnathus pascu ali Romer;19 and Exaeretodon frenguellii Cabrera.20 Material of Exaeretodon riograndensis Abdala, Barberena and Dornelles21 from Brazil was also prepared for this study, but unfortunately the material was diagenetically altered during fossilization, and its histological details were obliterated. Detailed comparisons of our study material are made with earlier osteohistological findings of nonmammalian therapsids, including other traversodontids,8 as well as with that of Mesozoic mammaliaform taxa.17 Thus, the current study of these three species expands our understanding of nonmammaliaform cynodont biology, and provides valuable palaeobiological insight into the Traversodontidae.

Materials and methods
Each of the traversodontid taxa studied is described separately below, and a description of the histological method follows thereafter.
Andescynodon mendozensis Bonaparte, 1969
Andescynodon is the most abundant taxon from the Cerro de las Cabras Formation.22 The largest skulls of Andescynodon range from 80 to 90 mm and its body length was probably between 400 and 500 mm. On the basis of its dental morphology, Andescynodon is considered to be an omnivorous cynodont.23,24 The femur (PVL 3836, Table 1) is the smallest femur known of this taxon and was associated with a skull of A. mendozensis that had a basal skull length of 65 mm (Table 2). Considering its skull length, the individual that we studied is estimated to be about 72% of adult body size, and therefore of subadult ontogenetic status. Indeed, the Andescynodon femur studied here is 80% of the length of the largest traversodontid femur known from Cerro de las Cabras Formation (since this bone was not associated with a skull, however, it could be either from Andescynodon or another traversodontid, such as Rusconiodon, which is also known from these deposits).
Massetognathus pascuali Romer, 1967
Abundant remains of Massetognathus are known from the Chañares Formation of Argentina.19,25,26 The maximum skull length of this taxon is about 205 mm and its estimated body length is 1.3 m.25 The femur included in this study (PVL 4613, Table 1) is part of a skeleton of a diagnostic Massetognathus specimen. The estimated length of PVL 4613 (see Table 2) is 66% of the largest femur known, and it is similar in size to that of MCZ 3691 R, which is a virtually complete and articulated skeleton of Massetognathus that Abdala and Giannini25 have proposed is subadult in age.
Exaeretodon frenguellii Cabrera, 1943
Exaeretodon is one of the best representatives of the Traversodontidae, and one of the common forms in the Ischigualasto Formation.27 The species is well known and it has been well-studied anatomically.28–30 The skull length of the species reaches 400 mm and an estimated body length of 2.5 m. Exaeretodon were omnivores, showing the expanded upper postcanines with a high and sharp sectorial crest. A distal portion of the fibula was included in the analysis (PVL 2162, Table 1). Considering the size of the element, the specimen is probably from an adult.
Exaeretodon riograndensis Abdala, Barberena and Dornelles 2002
Exaeretodon riograndensis is represented by a few specimens recovered from the Santa Maria Formation in southern Brazil.21 A complete tibia (MCNPV 10001, Table 1) was included in the current histological analysis.
Histological methods
As a consequence of limited diagnostic material available for histological analyses, we were unable to study the same skeletal element for each taxon. However, since several studies have found that long bones (such femora, tibiae and humeri) are especially well suited for histological analysis as they reliably preserve the histological record of growth processes,3,4 we limited this analysis by selecting representative long bones of each taxon. The sampled traversodontid bones were embedded in a clear epoxy resin and thin sections were prepared according to the method outlined in Chinsamy and Raath.31 Transverse sections were made in the diaphyseal regions of all the sampled bones, and in addition we were able to make longitudinal sections of the femora of Andescynodon, Massetognathus, and the tibia of Exaeretodon riograndensis. The thin sections were examined under ordinary light and polarized microscopes. Image analysis and measurements were conducted using NIS-Elements (version 2.2).
Results
Andescynodon mendozensis
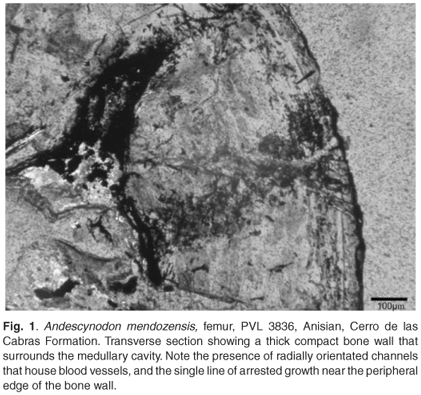
Transverse sections of the femur show a central medullary cavity that is surrounded by a fairly thick compact bone wall that measures on average about 1239 µm (Fig. 1). Although the bone is somewhat fragmented, the bone wall thickness is deducible and is about 29% of the diameter. A large number of radially organized blood vessels are visible in the compacta (Fig. 1). These tend to be simple blood vessels, without any osteonal development around them. The overall bone tissue has a lamellar texture. In the peripheral region, one line of arrested growth (LAG) is visible. In longitudinal sections of the bone, columns of calcified cartilage are present at the distal ends, and islands of calcified cartilage are present in some bony struts.
Massetognathus pascuali
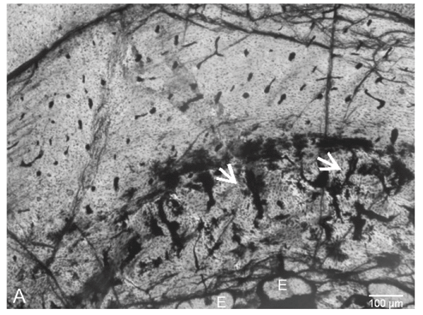
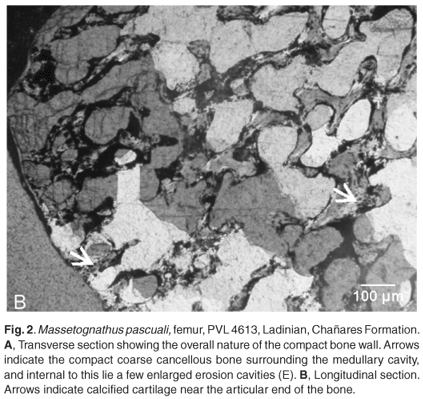
Transverse sections of the femur reveals a fairly thick bone wall (980 µm), of which about 47% consists of compacted coarse cancellous bone tissue (Fig. 2A). The periosteal bone tissue comprises parallel-fibred bone with a predominance of simple blood vessels that are mainly longitudinally arranged. Some circumferentially orientated blood vessels occur. A large number of erosion cavities are present around the medullary cavity (Fig. 2A). Compacted coarse cancellous bone is present in more distally located sections (Fig. 2A). Longitudinal thin sections of the femur showed struts of endochondral bone, and a narrow layer of calcified cartilage at the 'articular' end of the bone (Fig. 2B).
Exaeretodon riograndensis
Unfortunately, even though the gross morphology of the tibia is well preserved, the thin sections of the bone reveal that the bone has been diagenetically altered,3 and no histological detail is preserved.
Exaeretodon frenguellii
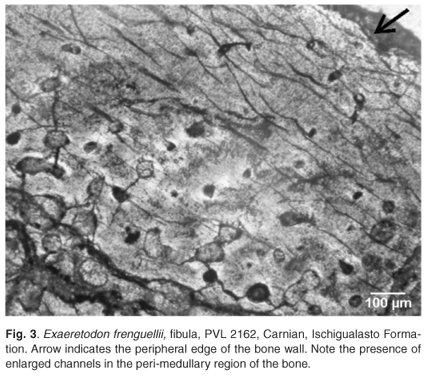
Thin sections of the fibula revealed that although the bone wall is quite fragmented, the histology is still discernible. The bone wall is fairly thick and consists of a compact bone region (measures about 880 µm), which is well vascularized by predominantly longitudinally orientated vascular canals (Fig. 3). Under polarized light, the osteonal structure around the vascular channels is clearly visible. There appear to be 1–2 rather faint growth rings visible, but these cannot be followed around the entire compacta. Bone around the medullary cavity comprises fibrolamellar bone tissue, whereas bone nearer the periosteal surface is more lamellar in structure. More distally located sections have much more cancellous bone present. A large number of erosion cavities are present around the medullary cavity, but few show secondary deposits of bone.
Discussion
The microscopic structure of the fossilized bone (bone histology/osteohistology) provides a host of information about various aspects of the life history of extinct animals.1–4,16 For example, deductions about an ontogenetic age, developmental patterns and growth strategies can be assessed directly from the histology of fossil bones. More specifically, different types of bone tissue permit deductions about the relative rate at which the bone was formed.32 Thus, a fast rate of bone formation results in fibrolamellar bone tissue in which the collagen fibres are haphazardly arranged and form a woven bone matrix wherein numerous primary osteons occur.1–3 A slower rate of bone formation results in a more lamellar type of bone tissue with a more organized arrangement of collagen fibres, and distinctly flattened osteocytes. Intermediates between these two bone tissue types occur and result in different, easily distinguishable types of tissues.1–3 In recent years, bone depositional rates of different tissue types have been quantified using fluorochrome staining techniques in a variety of modern vertebrates.33–35 From these analyses it appears that bone depositional rates of particular tissue types are quite variable and depend on several factors, including ontogeny, location in the skeleton, and environment. As a result, extrapolating actual bone depositional rates directly from modern vertebrates to extinct animals is questionable.3,4,34 In the current study of traversodontid osteohistology, therefore, we use qualitative, relative rates of bone deposition for comparisons between the taxa.
The palaeobiological implications of the bone microstructure for each of the study animals are discussed separately below. This is followed by a general description of traversodontid bone microstructure considering both previously published material and specimens in the current study, and a then by comparison of traversodontid bone microstructure with nonmammaliaform cynodonts, and Mesozoic mammaliaforms.
Bone microstructure of the South American traversodontids
Andescynodon mendozensis. The Andescynodon femoral microstructure is distinctive from that of the other traversodontids studied in that it has the thickest bone wall (almost 30% of the cross-sectional diameter of the bone), and also in having a number of radially orientated 'vascular canals' in a lamellar matrix of bone. The latter characteristic suggests that bone formation occurred in this animal at a much slower rate when compared with the other traversodontids studied. Interestingly, thick bone walls in terrestrial animals have been documented in the nonmammaliaform cynodonts, Thrinaxodon15 and Trirachodon,14 and have been interpreted as suggesting a fossorial lifestyle – a deduction additionally supported by the fact that both these cynodonts have been found associated with burrows.36,37 Furthermore, modern burrowing vertebrates (e.g. the naked mole rat, Heterocephalus glaber, the porcupine, Erethizon; as well as burrowing or digging lizards: Gerrhonotus grantis, Heloderma suspectum and Phyrynosoma doublassi) are also known to have thick bone walls.38,14,17 It is possible that Andescynodon, with its small body size, may also have been fossorial, but it should be noted that its skeletal remains have not been associated with burrows, and as in the majority of nonmammaliaform cynodonts (including possibly 'burrowing forms', Trirachodon and Thrinaxodon), there are no anatomical features in its skeleton suggestive of this type of lifestyle.
The columns of calcified cartilage near the articular surfaces, as well as islands of calcified cartilage within bony struts near the ends of the bone in the longitudinal sections of the femur, suggest that the specimen examined is an individual that was still growing at the time of its death, and agrees with the size-based deduction that it is a subadult individual.
Massetognathus pascuali. The Massetognathus femoral bone microstructure suggests a faster rate of osteogenesis than that of Andescynodon, and a slower rate of bone formation compared with Exaeretodon. The large number of peri-medullary erosion cavities also suggests that secondary resorption of bone was well under way. Longitudinal sections of the femur revealed the presence of calcified cartilage nearest the 'articular surface' of the bone, which suggest that maximum skeletal size had not yet been attained. The sparse occurrence of calcified cartilage and its restriction to a narrow region at the ends of the bone suggests that the individual was not very young and, more likely, subadult in ontogenetic status. These histological findings support the assumption that PVL 4613, like MCZ 3691 R, is a subadult.25
Exaeretodon frenguellii. The bone microstructure of the fibula of Exaeretodon frenguellii provides information regarding its growth dynamics. The initial deposition of a well-vascularized bone tissue in a woven bone matrix indicates a rapid growth phase, which was followed by a slower rate of growth as indicated by the parallel-fibred bone tissue. During the latter phase of growth, it is not certain but it seems that periodic pauses in the rate of bone deposition may have occurred. This type of growth pattern, with a rapid initial growth and subsequent slower growth, has been observed in several nonmammaliaform therapsids,16 as well as in mammaliaforms such as Morganucodon and the multituberculates Kryptobaatar and Nemegtbaatar.17 Resorption of bone is indicated by the presence of a large number of erosion cavities. The sparse occurrence of secondary osteons is indicative of a slower rate of re-deposition of bone in the erosion cavities.
Traversodontid bone microstructure and developmental growth patterns
The analysis of diagnostic traversodontid taxa permits some overall deductions about traversodontid bone microstructure and developmental patterns. A distinctive feature of the bone microstructure of the traversodontid taxa is that they are comprised essentially of primary bone tissue. Some peri-medullary erosion cavities are evident in Exaeretodon and Massetognathus, but secondary osteons tend to be sparse. These findings with regard to the compact bone tissues are consistent with that described by De Ricqlès8 for Gomphodontosuchus and Traversodon, suggesting that calcium metabolism among the traversodontids was not intensive, and appears not to have imposed heavy demands on their skeletons.1,2 In addition, because Andescynodon and Massetognathus appear to be subadults, we would not expect extensive development of secondary reconstructed bone (which tends to increase with increasing maturity). The Exaeretodon sample, on the other hand, is from an older individual—this is evident from the change in texture of the bone, that is, from a fibrolamellar tissue type to a lamellar type. Interestingly, in this individual, large peri-medullary erosion cavities occur, but secondary deposits therein have not yet formed.
The osteohistology of the traversodontids suggest that the different taxa experienced different growth strategies—Andescynodon mendozensis with its characteristic lamellar textured bone and the absence of osteonal development around the channels in the bone that housed blood vessels, suggests that bone formation and hence growth occurred at a slower rate than in both Massetognathus pascuali and Exaeretodon frenguellii. The compacta of M. pascuali tends to be uninterrupted, and consists of parallel-fibred bone tissue with simple blood vessels—these features suggest a faster rate of bone formation than in A. mendozensis. It is possible that an earlier, more rapid (and perhaps cyclical) phase of growth has been eroded away by secondary reconstruction, but this cannot be ascertained from the material at hand. If additional juvenile samples become available for histological study, it would be worth verifying whether indeed this was the case. If the initial phase of rapid growth were absent, then it would appear that Massetognathus has the distinctive characteristic of intermediate, but sustained rate of bone formation (and hence growth). The presence of fibrolamellar bone and primary osteons in the compacta of Exaeretodon frenguellii suggests that it had the fastest rate of growth as compared with the traversodontids in this study. The initial fast rate of bone growth is followed by more slowly formed lamellar bone tissue (and as mentioned above, it is uncertain whether growth was interrupted). These histological features of Exaeretodon suggest that the individual had passed its rapid phase of growth, and that its growth was slowing down. The fact that closely spaced peripheral rest lines (indicative of the attainment of maximum body size)2–4 are not visible in the compacta imply that the Exaeretodon fibula had not yet reached its maximum size.
The longitudinal sections of the bones provided an assessment of growth in length, and permitted deductions regarding relative ages of individuals: the calcified cartilage in the Andescynodon and Massetognathus specimens examined suggests that they were still growing at the time of death, being more likely subadults. However, the more extensive development of the calcified cartilage at the extremity suggests that the Andescynodon femur was growing more rapidly in length than the Massetognathus specimen. This is particularly noteworthy as the Andescynodon femur is 80% as long as the largest femur from the deposit whereas the Massetognathus bone is 66% of the largest known. This raises the intriguing possibility that perhaps the largest traversodontid femur known from the Cerro de las Cabras Formation is not representative of the largest, adult size of Andescynodon, as the histological features in the femur studied clearly suggest that it was still rapidly growing in length.
Thus, our analysis of the bone microstructure of these Argentinian traversodontids showed that although Andescynodon and Massetognathus clearly passed their most rapid phases of growth, they had not yet reached skeletal maturity. The Exaeretodon fibula suggests that this individual was more mature, and appositional growth had visibly slowed. Curiously, it is also apparent that there seems to be a trend of increasing the rate at which bone is deposited, i.e. from the most basal traversodontid to the more derived forms such as Exaeretodon and Gomphodontosuchus.8 This seems to parallel the trend found among the more basal nonmammaliaform cynodonts.39
It is also worth noting that palaeoenvironmental analyses suggest that both Andescynodon and Exaeretodon lived in humid, warm climates.40,41 However, the alluvial plain, with lakes, swamps and marshes interpreted for the Cerro de las Cabras Formation40 from which Andescynodon was recovered, represents a more equable humid environment than the predominantly fluvial Ischigualasto Formation (from which Exaeretodon was obtained) that shows seasonal variations in water availability.26,41 Differences observed in the bone microstructure of these taxa, and hence bone growth, therefore possibly reflect innate biological differences between the taxa.
Growth patterns of the traversodontids within the context of the nonmammaliaform cynodonts and mammaliaforms
Earlier studies of the bone microstructure of nonmammaliaform cynodonts have shown that they have variable patterns of bone microstructure, ranging from a lamellar-zonal bone in Procynosuchus16,39 to zonal bone with fibrolamellar bone tissue (e.g. Diademodon13 and Trirachodon14) and to uninterrupted fibrolamellar bone tissue (e.g. Cynognathus;13 and the tritylodontids,Tritylodon,16 Bienotherium and Oligokyphus).8 These findings suggest an increasing trend towards more rapid growth as reflected by uninterrupted fibrolamellar bone tissues. However, it is noteworthy that the ability to form fibrolamellar bone (albeit periodically) seems to have been an early innovation among the nonmammaliaform therapsids, and is already present in Scylacops, a Late Permian gorgonopsian,16 as well as among the dicynodonts10–12 (Fig. 4).
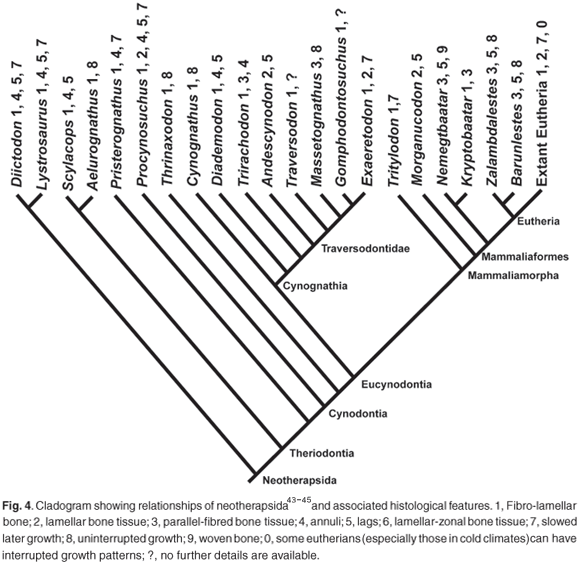
Our results on the bone microstructure of the traversodontids Exaeretodon frenguellii, Massetognathus pascuali and Andescynodon mendozensis, as well as those of Gomphodontosuchus brasiliensis and Traversodon stahleckeri, previously studied by De Ricqlès,8 show heterogeneous patterns of bone microstructure. This concurs with findings in several other nonmammaliaform cynodonts and therapsids,11–16,39 and appears to be a trend also in the basal mammaliaforms17 (Fig. 4).
Conclusions
Examined within a phylogenetic framework, the osteohistological data of traversodontids derived from the current study, as well as those of other nonmammaliaform cynodonts provided by previous contributions,8,13–16 permits comparisons of growth dynamics within these clades and provides an understanding of the evolution of life-history patterns among the therapsids. As a result of these studies, it appears that the ability to grow rapidly evolved fairly early in the Therapsida. Indeed, the widespread occurrence of fibrolamellar bone in Permian dicynodonts10,11 and gorgonopsians16 suggests that this 'innovation' extends deep into the therapsid lineage.
However, it appears that many of the nonmammalian cynodonts (including some traversodontids), although having the ability to form bone at rapid rates, retained flexible growth trajectories (developmental plasticity), which may have been dictated by prevailing environmental conditions. This plasticity also appears to have been present among some Mesozoic eutherians (Barunlestes and Zalambdalestes17). Although most modern mammals grow rapidly without periodic interruptions, some living mammals that experience stressful environments such as harsh seasonal conditions (e.g. polar bears and small rodents), can have periodic interruptions in bone growth.3,42
Thus, although there appears to be an increase in the rate of bone formation from basal nonmammaliaform cynodonts to more derived forms,16,39 and also among the traversodontids studied here, the plesiomorphic pattern of bone microstructure among the Therapsida is one of developmental plasticity.16,34 Within this general pattern, however, there are variations in the rate at which bone is deposited within the zonal region (i.e. whether fibrolamellar bone or lamellar bone forms) and whether the growth cycles are marked by abrupt pauses (indicated by lines of arrested growth) or simply a decrease in the rate of bone formation. It is also apparent that some therapsids (and particularly some nonmammaliaform cynodonts) have independently acquired the ability to grow along inflexible growth trajectories.13,16 Given the variability that occurs in the Cynodontia where the bone histology is known, it is apparent that there is not one particular type of bone tissue that can be regarded as a synapomorphy for the clade. Thus, the microscopic structure of bone appears to be highly variable even among closely related forms, and therefore not constrained by phylogeny alone. On the contrary, the overall texture and nature of bone appears to be dependent on a host of extrinsic and intrinsic factors.3,4
Access to materials for this study was permitted by Ana Maria Ribeiro (Fundação Zoobotânica de Rio Grande do Sul, Brazil), and Jaime Powell (Universidad Nacional de Tucumán, Argentina). Charles Schaff (Museum of Comparative Zoology, Harvard University) provided information and measurements of material of Massetognathus housed in the MCZ. Sarah Werning provided comments on an earlier draft of this manuscript. This research was funded by the National Research Foundation (to A.C.) and by PAST and the Oppenheimer Foundation (to F.A.).
1. Francillon-Vieillot H., Buffrénil V., de Castanet J., Géraudie J., Meunier F.J., Sire J.Y., Zylberberg L. and Ricqlès A.de (1990). Microstructure and mineralization of vertebrate skeletal tissues. In Skeletal Biomineralisation: Patterns, Processes and Evolutionary Trends, ed. J.G. Carter, pp. 471–530. Van Nostrand Reinhold, New York. [ Links ]
2. Chinsamy A. (1997). Assessing the biology of fossil vertebrates through bone histology. Palaeont. afr. 33, 29–35. [ Links ]
3. Chinsamy-Turan A. (2005). The Microstructure of Dinosaur Bone: Deciphering Biology with Fine Scale Techniques. Johns Hopkins University Press, Baltimore. [ Links ]
4. Erickson G.M. (2005). Assessing dinosaur growth patterns: a microscopic revolution. TREE 20, 677–684. [ Links ]
5. Gross W. (1934). Die Typen des mikroskopischen Knochenbaues bei fossilen Stegocephalen und Reptilien. Zeit. Anat. 103, 731–764. [ Links ]
6. Enlow D.H. and Brown S.O. (1956). A comparative histological study of fossil and recent bone tissues. Part I. Tex. J. Sci. 8, 405–443. [ Links ]
7. Enlow D.H. and Brown S.O. (1957). A comparative histological study of fossil and recent bone tissues. Part II. Tex. J. Sci. 9, 136–214. [ Links ]
8. Ricqlès de A. (1969). Recherches paléohistologiques sur les os longs des tétrapodes. II. Quelques observations sur la structure des os logns des thériodontes. Ann. Paléont. 40, 1–52. [ Links ]
9. Ricqlès de A. (1972). Recherches paléohistologiques sur les os longs des tétrapodes. III. Titanosuchiens, Dinocéphales et Dicynodontes. Ann. Paléont. 58, 17–60. [ Links ]
10. Chinsamy A. and Rubidge B. (1993). Dicynodont (Therapsida) bone histology: phylogenetic and physiological implications. Palaeont. afr. 30, 97–106. [ Links ]
11. Ray S. and Chinsamy A. (2004). Diictodon feliceps (Therapsida, Dicynodontia): bone histology, growth and biomechanics. J. Vert. Paleontol. 24, 180–194. [ Links ]
12. Ray S., Chinsamy A. and Bandyopathy S. (2005). Lystrosaurus murrayi (Therapsida, Dicynodontia): bone histology, growth and palaeoecological implications. Palaeontology 48, 1169–1185. [ Links ]
13. Botha J. and Chinsamy A. (2000). Growth patterns deduced from the bone histology of the Cynodonts Diademodon and Cynognathus. J. Vert. Paleontol. 20, 705–711. [ Links ]
14. Botha J. and Chinsamy A. (2004). Growth and life habits of the Triassic non-mammalian cynodont Trirachodon inferred from bone histology. Acta Paleontol. Pol. 49, 619–627. [ Links ]
15. Botha J. and Chinsamy A. (2005). Growth patterns of Thrinaxodon liorhinus, a non-mammalian cynodont from the Lower Triassic of South Africa. Palaeontology 48, 385–394. [ Links ]
16. Ray S., Botha J. and Chinsamy A. (2004). Bone histology and growth patterns of some non mammalian therapsids. J. Vert. Paleontol. 24, 634–648. [ Links ]
17. Chinsamy A. and Hurum J.H. (2006). Bone microstructure and growth patterns of early mammals. Acta Paleontol. Pol. 51, 325–338. [ Links ]
18. Bonaparte J.F. (1969). Dos nuevas 'faunas' de reptiles triásicos de Argentina. Gondwana Stratigraphy, I.U.G.S., Mar del Plata, 283–306. [ Links ]
19. Romer A.S. (1967). The Chañares (Argentina) Triassic reptile fauna. III. Two new gomphodonts, Massetognathus pascuali and Massetognathus teruggii. Breviora 264, 1–25. [ Links ]
20. Cabrera A. (1943). El primer hallazgo de terápsidos en la Argentina. Notas Museo de La Plata 8, 317–331. [ Links ]
21. Abdala F., Barberena M.C. and Dornelles J. (2002). A new species of the traversodontid cynodont Exaeretodon from the Santa Maria Formation (Middle/Late Triassic) of southern Brazil. J. Vert. Paleontol. 22, 313–325. [ Links ]
22. Zavattieri A.M. and Arcucci A.B. (2003). Edad y posición estratigráfica de los tetrápodos de la localidad Cerro Bayo de Potrerillos (Triásico), Mendoza, Argentina. Ameghiniana 40, 75R. [ Links ]
23. Goñi R.G. (1986). Reemplazo de dientes postcaninos en Andescynodon mendozensis Bonaparte (Cynodontia, Traversodontidae). Actas IV Congreso Argentino de Paleontología y Bioestratigrafia 2, 7–14. [ Links ]
24. Goñi R.G. and Goin F.J. (1988). Morfología dentaria y biomecánica masticatoria de los cinodontes (Reptilia-Therapsida) del Triásico argentino. I. Andescynodon mendozensis Bonaparte (Cynodontia, Traversodontidae). Ameghiniana 25, 139–148. [ Links ]
25. Abdala F. and Giannini N.P. (2000). Gomphodont cynodonts of the Chañares Formation: the analysis of an ontogenetic sequence. J. Vert. Paleontol. 20, 501–506. [ Links ]
26. Rogers R.R., Arcucci A.B., Abdala F., Sereno P.C., Forster C.A. and May C.L. (2001). Paleoenviroment and taphonomy of the Chanares Formation tetrapod assemblage (Middle Triassic), northwestern Argentina, spectacular preservation in volcanogenic concretions. Palaios 16, 461–481. [ Links ]
27. Rogers R.R., Swisher C.C. III, Sereno P.C., Monetta A.M., Forster C.A.and Martínez R.N. (1993). The Ischigualasto tetrapod assemblage (Late Triassic, Argentina) and 40Ar/39Ar dating of dinosaur origins. Science 260, 794–797. [ Links ]
28. Bonaparte J.F. (1962). Descripción del cráneo y mandíbula de Exaeretodon frenguellii, Cabrera, y su comparación con Diademodontidae, Tritylodontidae y los cinodontes sudamericanos. Publicaciones Museo Municipal de Ciencias Naturales y Tradición, Mar del Plata 1, 135–202. [ Links ]
29. Bonaparte J.F. (1963). Descripción del esqueleto postcraneano de Exaeretodon (Cynodontia-Traversodontidae). Acta Geol. Lilloana 4, 5–52. [ Links ]
30. Bonaparte J.F. (1966). Sobre las cavidades nasal, cerebral y otras estructuras del craneo de Exaeretodon sp. (Cynodontia-Traversodontidae). Acta Geol. Lilloana 8, 5–29. [ Links ]
31. Chinsamy A. and Raath M.A. (1992). Preparation of fossil bone for histological examination. Palaeont. afr. 29, 39–44. [ Links ]
32. Amprino R. (1947). La structure du tissue osseux envisage comme expression de différences dans la vitesse de l'accroissement. Arch. Biol. 58, 315–330. [ Links ]
33. Castanet J., Rogers K.R., Cubo J. and Boisard J. (2000). Periosteal bone growth rates in extant ratites (ostrich and emu). Implicaitons for assessing growth dynamics in dinosaurs. Life Sci. 323, 543–550. [ Links ]
34. Starck J.M. and Chinsamy A. (2004). Bone microstructure and developmental plasticity in birds and other dinosaurs. J. Morphol. 254, 232–246. [ Links ]
35. De Margerie E., Cubo J. and Castanet J. (2002). Bone typology and growth rate: testing and quantifying 'Amprino's Rule' in the mallard (Anas platyrhynchos). C. R. Biol. 325, 221– 230. [ Links ]
36. Groenewald G.H., Welman J. and MacEachern J.A. (2001). Vertebrate burrows complexes from the Early Triassic Cynognathus Zone (Driekoppen Formation, Beaufort Group) of the Karoo Basin, South Africa. Palaios 16, 148–160. [ Links ]
37. Damiani R., Modesto S., Yates A. and Neveling J. (2003). Earliest evidence of cynodont burrowing. Proc. R. Soc. Lond. B 270, 1747–1751. [ Links ]
38. Magwene G. (1993). What's bred in the bone: histology and cross-sectional geometry of mammal-like reptile long bones – evidence of changing physiological and biomechanical demands. M.Sc. dissertation, Harvard University, Cambridge, [ Links ]
39. Botha J. (2002). The palaeobiology of the non-mammalian cynodonts deduced from bone microstructure and stable isotopes. Ph.D. dissertation, University of Cape Town, South Africa. [ Links ]
40. Zavattieri A.M. (1990). Stratigraphic and paleoecologic evaluation of the palynofloras of the Triassic Las Cabras Formation at the type locality Cerro de Las Cabras, Mendoza, Argentina. N. Jb. Geol. Paläont., Abh. 181, 117–142. [ Links ]
41. Milana J.P. (2002). Ischigualasto (Secuencia). In Léxico estratigráfico de la Argentina, Volumen VIII, Triásico, eds P.N. Stipanicic and C.A. Marsicano, pp. 149, Asociación Geológica Argentina, Serie B (Didáctica y Complementaria) 26, Buenos Aires. [ Links ]
42. Klevezal G.A. and Kleinenberg S.E. (1969). Age determination of mammals from annual layers in teeth and bone. Translated from Russian by J. Salkind: Israel Program for Scientific Translations Press, Jerusalem. [ Links ]
43. Abdala F.J. Neveling and Welman J. (2006). A new trirachodontid cynodont from the lower levels of the Burgersdorp Formation (Lower Triassic) of the Beaufort Group, South Africa and the cladistic relationships of Gondwanan gomphodonts. Zool. J. Linn. Soc. 147, 383–413. [ Links ]
44. Abdala F. (2007). Redescription of Platycraniellus elegans (Therapsida, Cynodontia) from the Lower Triassic of South Africa, and the cladistic relationships of eutheriodonts. Palaeontology 50, 591–618. [ Links ]
45. Rubidge B.S. and Sidor C.A. (2001). Evolutionary patterns among Permo-Triassic therapsids. Annu. Rev. Ecol. Syst. 32, 449–480. [ Links ]
Received 10 December 2007. Accepted 18 May 2008.
Institutional abbreviations: MCN-PV, Museu de Ciências Naturais, Fundação Zoobotânica do Rio Grande do Sul, Porto Alegre, Brazil; MCZ, Museum of Comparative Zoology, Harvard University, Cambridge, Massachusetts; PVL, Colección Paleontología de Vertebrados Lillo, Universidad Nacional de Tucumán, Argentina.
* Author for correspondence. E-mail: anusuya.chinsamy-turan@uct.ac.za