Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.104 n.5-6 Pretoria May./Jun. 2008
COMMENTARY
Lessons from aloes in the Thicket Biome: reconstructing past elephant browsing to understand the present
Dan Parker; Ric Bernard
Wildlife and Reserve Management Research Group, Department of Zoology and Entomology, Rhodes University, P.O. Box 94, Grahamstown 6140, South Africa
It is very difficult to quantify elephant-induced change to vegetation in the absence of adequate historical benchmarks. In this commentary, we explore the historical distribution of aloes in the Thicket Biome of South Africa. We contend that the large stands of aesthetically pleasing aloes in the Thicket Biome can be likened to the even-aged stands of tall trees in the riparian forests of Botswana, both being artefacts of the loss of large herbivores through disease and hunting in the past. Elephant browsing on aloes may therefore be the first step in the vegetation reverting to a situation similar to the one prior to excessive hunting in the region.
The vehicle registration plates of the Eastern Cape province of South Africa have two seemingly unlikely partners emblazoned behind the text—an aloe and an elephant. Unlikely, because published data from the Addo Elephant National Park indicate that although aloes are commonly encountered in survey plots outside of the elephant enclosure, they are absent in the parts of the park where the elephants have access to these preferred food items.1 This has prompted some of the country's top ecologists (and members of the 'Elephant Science Roundtable'), as recently as 2006, to conclude: 'Plant species losses have been documented in the Addo Elephant National Park and are a cause for concern.' (ref. 2, p. 393). In this article we ask whether there is, in fact, a need to be concerned at the loss of plant species in the Thicket Biome of the Eastern Cape.
The key to determining whether plant species loss through the re-introduction of elephants is of concern requires an understanding of the conditions prior to the extirpation of elephants. Written records from the 18th and 19th centuries lack detail3 and it is not possible to establish how many elephants there were a century or more ago, what their movement patterns were and what the vegetation would have looked like.2,4 In the absence of these benchmarks against which change can be measured, it is not surprising that the opinions of researchers are mixed, with some reporting change as being negative5,6 and others adopting a more neutral standpoint.7,8 This is not a trivial issue, however, because if current elephant populations are interpreted as being high and change as undesirable, then elephant populations should be controlled. If the same elephant population is not seen to be high and the change is not interpreted negatively, then the urgency for population control is reduced. Fortunately, the ecological community has realised that a holistic approach is required to manage elephant populations effectively.2,4,9 As part of this approach, we need to try to reconstruct a picture of past elephant numbers and, more importantly, what the vegetation would have looked like prior to anthropogenic influences such as the introduction of fences, artificial water-points and elephant range contraction.
It is generally accepted that woodlands in Africa tend to establish during episodic windows of opportunity when biotic and abiotic factors are favourable.2,6,10,11 These periods give rise to aesthetically pleasing, even-aged stands of vegetation, such as the Acacia erioloba forests of Zimbabwe's Hwange National Park12 and the riparian forest of the Chobe National Park in Botswana.10 As Owen-Smith et al.2 point out, however, when large, generalist herbivores that have high energy requirements, like elephants, begin to alter the structure of these woodlands, there are no historical benchmarks with which to compare the resultant state.
Seminal work by Skarpe et al.11 demonstrated that there has been a profound change in the extent and composition of the riparian forest on the Chobe River in Botswana since 1960, which is presumed to be due to high numbers of elephants and other herbivores. Most of the mature trees are dead and the area has been largely replaced with shrubland.11 However, these authors show that, based on historical records, the current situation is probably similar to the one predating excessive hunting and the rinderpest in about 1880,11 raising the question of whether there was any need to be concerned about the effects of herbivores in the first place.
We believe that a similar situation exists with aloes and elephants in the Eastern Cape province. Stands of aloes, Aloe ferox in particular, have become synonymous with the province and can reach densities of 10 individuals per square kilometre or more (D. Parker, unpublished data). Using height data collected for living aloes at five sites with elephants (re-introduced between 1 and 13 years prior to the assessment) and five sites without elephants in the Eastern Cape, we estimated the ages of A. ferox individuals using a known growth rate (2.79 ± 0.20 cm/yr).13 An overwhelming proportion (81%) of the 459 aloes at sites without elephants were estimated to be younger than 83 years old. Furthermore, living aloes at sites with elephants (n = 348) were significantly younger than those at sites without elephants (Fig. 1; P < 0.05, t805 = 3.57). It is unlikely that the significant difference in the ages of aloes at sites with and without elephants simply reflects localised site effects (such as differing geologies, rainfall patterns, aspect, land use and herbivore densities), as aloes were measured at ten sites, some separated by as much as 60 km. It is more likely that elephants are selectively feeding on aloes of a particular height and killing these individuals, leaving behind only smaller/younger aloes (see ref. 14; D. Parker, pers. obs.).
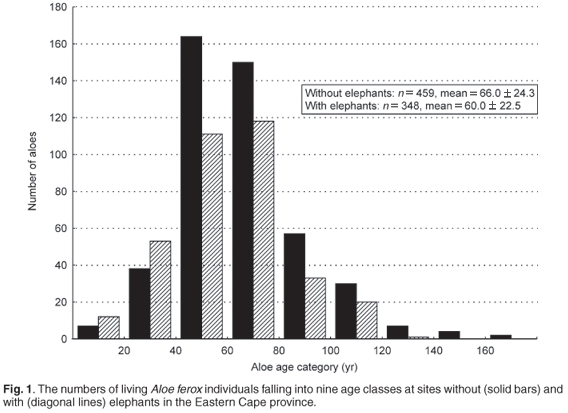
Interpreting the age structure of A. ferox populations is difficult, however, in the absence of a full understanding of natural mortality rates. The long, right-hand tail in Fig. 1 might simply imply that aloes over a certain age at sites without elephants are more likely to topple over. The shallow root systems of these plants certainly makes them more susceptible to being uprooted during strong winds (or by other large herbivores) when soil is moist (ref. 15; D. Parker, pers. obs.). Alternatively, the observed age structure may reflect the history of elephants and other herbivores in the Eastern Cape as the data show that at least some aloes can live for more than 150 years (Fig. 1). Thus, the large stands of (aesthetically pleasing) aloes currently found in the region may be derived from a cohort of seedlings that established some 100 years ago, during a period when elephant and herbivore numbers in general were low as a result of high levels of hunting.16 Whatever the case, the re-introduction of elephants to fenced reserves in the Eastern Cape can be expected to bring about a change in the height and age structure of aloes. Further, what is perceived to be damage may be the first step in the vegetation reverting to a situation similar to the one before excessive hunting sensu the results of Skarpe et al.11 When elephant and/or other herbivore numbers were much higher in the Eastern Cape,16 aloes may have depended on local refugia (such as steep gorges and slopes) to persist in the region.11,17,18 However, it is important to emphasise that a single factor is unlikely to explain the observed age structure of aloes. Previous land use and length of time under current land use (conservation or farming) may have resulted in a skewed aloe age-structure. For example, goats are known to browse the leaves of A. ferox, particularly of smaller individuals, causing extensive damage.17
It is clear that a definite answer to our original question—Is there a need to be concerned with plant species (aloe) losses in the Thicket Biome?—is elusive. We believe that, even 13 years after their re-introduction to fenced reserves, elephants have already brought about a change in the height structure of aloes and will continue to do so, leading to small-scale extinctions as seen at Addo Elephant National Park.1 Whether or not this is seen as damage depends on the time scale. Over 100 years, the loss of aloes could be interpreted as serious damage, whereas over a millennium it may well be a return to a more natural state. Let us not forget the observation by Lewin: 'On the larger scale of things, change within ecosystems should therefore be seen as natural and inevitable, even if it sometimes leads to local extinctions.'(ref. 19, p. 1071)
We hope that we have encouraged scientists in South Africa, particularly those involved in elephant management, to consider and seek alternative interpretations for observed changes in vegetation structure, rather than pigeonholing these changes as being undesirable.
This work was funded by Rhodes University and we thank Randall Hepburn and Christopher McQuaid for constructive comments on the manuscript.
1. Penzhorn B.L., Robbertse P.J. and Olivier M.C. (1974). The influence of the African elephant on the vegetation of the Addo Elephant National Park. Koedoe 17, 137–158. [ Links ]
2. Owen-Smith N., Kerley G.I.H., Page B., Slotow R. and Van Aarde R.J. (2006). A scientific perspective on the management of elephants in the Kruger National Park and elsewhere. S. Afr. J. Sci. 102, 389–394. [ Links ]
3. Bernard R.T.F. and Parker D.M. (2006). The use of archaeological and ethnographical information to supplement the historical record of the distribution of large mammalian herbivores. S. Afr. J. Sci. 102, 117–119. [ Links ]
4. Van Aarde R.J., Jackson T.P. and Ferreira S.M. (2006). Conservation science and elephant management in southern Africa. S. Afr. J. Sci. 102, 385– 388. [ Links ]
5. Barnes R.F.W. (1980). The decline of the baobab tree in Ruaha National Park, Tanzania. Afr. J. Ecol. 18, 243–252. [ Links ]
6. Western D. and Maitumo D. (2004). Woodland loss and restoration in a savanna park: a 20-year experiment. Afr. J. Ecol. 42, 111–121. [ Links ]
7. Leuthold W. (1996). Recovery of woody vegetation in Tsavo National Park, Kenya, 1970–94. Afr. J. Ecol. 34, 101–112. [ Links ]
8. Guldemond R. and Van Aarde R. (2007). The impact of elephants on plants and their community variables in South Africa's Maputaland. Afr. J. Ecol. 45, 327–335. [ Links ]
9. Van Aarde R.J. and Jackson T.P. (2007). Megaparks for metapopulations: addressing the causes of locally high elephant numbers in southern Africa. Biol. Conserv. 134, 289–297. [ Links ]
10. Mosugelo D.K., Moe S.R., Ringrose S. and Nelleman C. (2002). Vegetation changes during a 36-year period in northern Chobe National Park, Botswana. Afr. J. Ecol. 40, 232–240. [ Links ]
11. Skarpe C., Aarrestad P.A., Andreassen H.P., Dhillion S.S., Dimakatso T., du Toit J.T., Halley D.J., Hytteborn H., Makhabu S., Mari M., Marokane W., Masunga G., Modise D., Moe S.R., Mojaphoko R., Mosugelo D., Motsumi S., Neo-Mahupeleng G., Ramotadima M., Rutina L., Sechele L., Sejoe T.B., Stokke S., Swenson J.E., Taolo C., Vandewalle M. and Wegge P. (2004). The return of the giants: ecological effects of an increasing elephant population. Ambio 33, 276–282. [ Links ]
12. Seymour C. and Milton S. (2003). A collation and overview of research information on Acacia erioloba (camelthorn) and identification of relevant research gaps to inform protection of the species. Department of Water Affairs and Forestry, Pretoria. [ Links ]
13. Shackleton C.M. and Gambiza J. (2007). Growth of Aloe ferox Mill. at selected sites in the Makana region of the Eastern Cape. S. Afr. J. Bot. 73, 266–269. [ Links ]
14. Steyn A. and Stalmans M. (2001). Selective habitat utilisation and impact on vegetation by African elephant within a heterogeneous landscape. Koedoe 44, 95–103. [ Links ]
15. Prasad M.N.V. (1997). Plant Ecophysiology. Wiley-Interscience, New Jersey. [ Links ]
16. Skead C.J. (2007). Historical Incidence of the Larger Land Mammals in the Broader Eastern Cape, 2nd edn. Nelson Mandela Metropolitan University, Port Elizabeth. [ Links ]
17. Breebaart L., Bhikraj R. and O'Connor T.G. (2002). Impact of goat browsing on Aloe ferox in a South African savanna. Afr. J. Range For. Sci. 19, 77–78. [ Links ]
18. Edkins M.T., Kruger L.M., Harris K. and Midgley J.J. (2007). Baobabs and elephants in Kruger National Park: nowhere to hide. Afr. J. Ecol. doi: 10.1111/j.1365-2028.2007.00798.x [ Links ]
19. Lewin R. (1986). In ecology, change brings stability. Science 234, 1071–1073. [ Links ]
Author for correspondence. E-mail: d.parker@ru.ac.za














