Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.104 n.3-4 Pretoria Mar./Apr. 2008
RESEARCH LETTERS
A comparison of drought stress and heat stress in the leaves and tubers of 12 potato cultivars
A. MienieI, *; J.A. De RondeII
IMangosuthu University of Technology, P.O.Box 12363, Jacobs, Durban 4026, South Africa
IIARC-VOPI, Private Bag X293, Pretoria 0001, South Africa
ABSTRACT
Potato yield is extremely sensitive to drought and heat stress, but there are variations in the degree to which cultivars are affected by these stresses. Strategies for the selection of stress-tolerant cultivars in a breeding programme can therefore be developed. The 2,3,5-triphenyltetrazolium chloride viability assay was evaluated to estimate drought and heat tolerance of leaves and tubers of 12 potato cultivars which differ in their response to drought. Floating leaf discs and tuber slices in 0.5 M mannitol (–1.24 MPa) simulated drought. After the drought acclimation treatment, the leaf discs were subjected to a lethal drought stress by exposure to an osmotic potential of –2.48 MPa. The viability of the leaf discs after the drought treatment was estimated spectrophotometrically by measuring the formazan concentration at 485 nm. Lower absorbance values in the control treatment, compared with the stress treatment, indicated a tolerant reaction. As drought simulation in the laboratory can differ from field conditions due to the effect of heat stress, the cultivars were also evaluated for heat tolerance. A stress index was established which can assist breeders to distinguish between plant responses to heat and drought.
Introduction
Potato yield is optimal under growing conditions with adequate light, water and cool temperatures. According to Vayda,1 heat and drought stress greatly influence potato yield. Total and marketable yield substantially decrease after short periods of severe stress. There are, however, variations in the degree to which cultivars are affected by these stresses. Thus, strategies for the selection of tolerant cultivars in a breeding programme are required.
Several physiological methods have been devised to measure abiotic stress tolerance in crop plants. These include: regrowth,2 triphenyltetrazolium chloride reduction (TTC),3 vital staining,4 protoplasmic streaming,5 plasmolysis,6 leakage of ions,7 and measurement of ultraviolet absorbing compounds.8 Ishikawa et al.2 compared viability assays in an attempt to identify the best for estimating freezing, heat and salt tolerance in brome grass. They found that TTC reduction was the most convenient assay, whereas regrowth, although time consuming and labour intensive, was the most sensitive and reliable assay. Although membrane injury as estimated by electrolyte leakage has been found to be unreliable in some cases,9 TTC tests and conductivity have been successfully used for evaluating heat and cold tolerance in potato. However, TTC reduction was found to be more sensitive than the conductivity test for evaluating heat tolerance.10 Vratsanos and Rossouw11 demonstrated a positive correlation between TTC viability assays and heat tolerance in three potato cultivars. The TTC viability assay also proved valuable in evaluation of soybean seedlings for thermotolerance,12 senescence in rice13 and cryopreservation.14 De Ronde and Van der Mescht15 expanded the TTC assay to measure drought stress in cotton. A positive correlation between drought tolerance and TTC reduction was shown in six cotton cultivars.
The TTC assay is based on the ability of viable cells to reduce tetrazolium salts metabolically into soluble formazans.16 Earlier work of ours showed that formazan production was relatively less in stressed leaves of drought-sensitive cotton cultivars than in the leaves of controls. However, for tolerant cultivars the opposite reaction was observed. Formazan levels were higher in the stress treatment than in the controls. Similar tendencies were found for drought as well as heat stress.15 This is possibly because the sensitive genotypes had inefficient tolerance mechanisms to survive a moderate stress and the plant could not adapt to the severe stress. The tolerant genotype has the potential to activate the stress response during moderate stress, thus the plant can tolerate a severe stress. This resulted in enhanced formazan production in the stress treatment. Thus, the method measures the ability of the plant tissue to adapt to increasing stress conditions.3 De Ronde and Van der Mescht15 hypothesized that a lower formazan level in the control treatment compared to a stress treatment indicates a drought-tolerant reaction. This was also used to predict the yield vigour of hybrids and open-pollinating varieties of Brassica napus, with the vigorous lines having a higher capacity to reduce TTC than less performing lines.17
Van der Mescht, De Ronde and Rossouw18 showed that drought-related protein synthesis is cultivar and organ specific in potato. It is thus important to evaluate the effect of drought stress on different organs. Additionally, De Ronde and Van der Mescht15 showed that drought and heat tolerances were negatively correlated in some cotton cultivars. The aim of the work reported here was to evaluate TTC reduction as a metabolic indicator of drought and heat tolerance in potato leaves and tubers.
Materials and methods
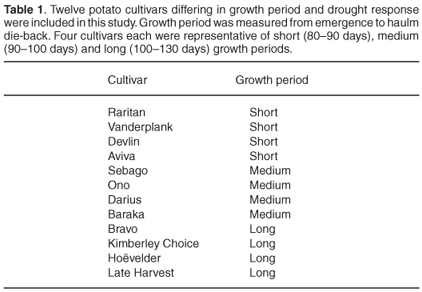
Plant material. Twelve potato cultivars (Table 1) were grown in a greenhouse under conditions as previously described by Van der Mescht et al.19 Leaves from the third apical node were harvested four weeks after emergence. Fresh tubers were harvested for the TTC tests.
Induction of drought and heat stress. Leaf discs (7 mm in diameter) or tuber slices (4 mm in diameter and 2 mm wide) of the control treatments were incubated in a sodium hydrogen maleate buffer (pH 6.0) at 23°C for 3 hours.20 The drought stress was induced by incubating the leaf discs or tuber slices in a sodium hydrogen maleate buffer containing 0.5 M mannitol (–1.24 MPa) as an osmoticum.15 After 3 hours, the stress and control treatments were incubated in 1.0 M mannitol (–2.48 MPa) and sampled over a period of 120 min. The moderate heat stress was induced by incubating the stress treatment at 37°C for 150 min before the lethal stress was induced at 45°C.9
TTC assay. The triphenyl formazan accumulation was measured spectrophotometrically at 485 nm as described by De Ronde et al.3 Five replicates were measured.
Results
Drought stress in leaves
Viability was measured every 30 min over 150 min after the lethal stress was induced. The formazan concentrations were higher in the stress treatment than in the control treatment in the cultivars 'Darius' and 'Ono', indicating a tolerant reaction (see Fig. A in supplementary material online). In the sensitive reaction, the formazan concentrations over time were lower in the stress treatment compared to the control treatment. This was true for all the cultivars except 'Darius' and 'Ono' (see Fig. B in supplementary material online). The area between the graphs was estimated as the difference between the respective means of the stress and control treatments over time (Fig. 1).3 With this information a stress index was developed ranking the cultivars according to their viability during osmotic stress (Table 2).


Drought stress in tubers
Tubers were subjected to osmotic stress as described for leaves. In contrast to the leaves, all the cultivars showed a sensitive reaction when tubers were used. The stress index was calculated (Fig. 2), ranking the cultivars from least sensitive to most sensitive. The cultivar 'Kimberley Choice' was least sensitive whereas 'Aviva' was most sensitive (Table 2). Additionally, the areas between the graphs were ten times larger than for leaves, an indication of tubers' hypersensitive reaction to drought.

Heat stress in leaves
Leaf discs were subjected to a moderate stress of 37°C for 3 hours before a lethal stress of 45°C was induced. Viability was measured every 30 min for 3 hours after induction of the lethal stress. Heat-tolerant reactions were found in the cultivars 'Bravo', 'Baraka' and 'Late Harvest', whereas sensitive reactions were observed in the rest of the cultivars (Fig. 3). The stress index was calculated and indicated that 'Bravo' was most heat tolerant and 'Vanderplank' the most heat sensitive (Table 2).

The cultivars were ranked according to the values of the difference between the mean of the stress treatments and the mean of the control treatments over time for each cultivar. The cultivar with the highest value was designated 1 and the cultivar with the lowest value 12.
Heat stress in tubers
Potato tubers were subjected to heat stress as described for leaves. According to the histogram, the cultivars 'Bravo', 'Vanderplank', 'Hoëvelder', 'Late Harvest', 'Kimberley Choice', 'Baraka' and 'Sebago' showed tolerant reactions whereas cultivars 'Devlin', 'Raritan', 'Ono', 'Darius' and 'Aviva' showed sensitive reactions (Fig. 4). A stress index with 'Bravo' as most tolerant and 'Aviva' as most sensitive was calculated and presented in Table 2. As for tubers and leaves subjected to osmotic stress, the areas between the graphs of tubers subjected to heat stress were ten times higher than for leaves.

Discussion
The TTC assay measures the ability of plant tissue to carry out electron transport. Additionally, inhibition of TTC-reduction indicates dehydrogenase inactivation resulting in a decrease in formazan production.21 TTC is reduced by the mitochondrial dehydrogenases and the efficiency of the formazan formation depends on the activity of cytochrome oxidase,17 particularly complex I, probably by accepting electrons directly from low potential cofactors.22 Results from leaves and tubers subjected to an osmotic stress of 0.5 M mannitol (–1.24 MPa) showed a sensitive response with the exception of leaves from the cultivars 'Darius' and 'Ono'. The difference in absorbance values between stress and control treatments in leaves subjected to osmotic stress is generally ten times higher than for tubers exposed to the same stress. This may indicate that potato leaves have a greater ability to adapt to drought stress than tubers. A stress ranking was established by listing the cultivars from the most viable to the least viable during osmotic stress. The difference between the ranking of leaves and tubers showed that the drought response was organ specific with the exception of 'Raritan' and 'Vanderplank' (Table 2).
Potato leaves were more heat sensitive than tubers. According to Van der Mescht et al.,23 cultivars are selected for drought avoidance rather than tolerance because drought tolerance is defined in terms of yield reduction. Thus, we are interested in plants that avoid drought by tuberization. This may also be true for heat tolerance. The difference in absorbance values between stress and control treatment was less in heat-stressed leaves (less than 0.06) than in tubers (less than 0.15). These values are similar to those recorded for drought and heat-stressed Eucalyptus grandis clones24 and cotton cultivars.15 The only exception was that these genotypes had more positive values than potato genotypes, which had more negative values, indicating sensitivity. The cultivar 'Bravo' was most heat tolerant when both leaves and tubers were tested compared to 'Aviva', which tested most sensitive to heat. According to the stress ranking (Table 2), the relative rankings between leaves and tubers were similar in the cultivars 'Devlin', 'Darius', 'Bravo', 'Kimberley Choice' and 'Late Harvest'. Tubers were more sensitive to heat than leaves for the cultivars 'Raritan', 'Aviva', 'Ono' and 'Baraka', whereas leaves were more heat sensitive than tubers in the cultivars 'Vanderplank', 'Sebago' and 'Hoëvelder'.
Information about the correlation between heat and drought tolerance is of great economic value. It may be used to determine the best locality for a specific cultivar as well as for cultivar improvement in a breeding programme. The data in Table 2 are also an indication of the correlation between heat and drought tolerance. When leaves were evaluated, a negative correlation was found in the cultivars 'Vanderplank', 'Darius', 'Baraka' and 'Late Harvest'. The cultivars 'Vanderplank' and 'Darius' were relatively drought tolerant and heat sensitive whereas 'the cultivars 'Baraka' and 'Late Harvest' were relatively heat tolerant and drought sensitive. In the case of tubers, it was found that the cultivar 'Darius' was relatively drought tolerant and heat sensitive whereas 'Late Harvest' and 'Hoëvelder' were relatively heat tolerant and drought sensitive. Li et al.10 reported a negative correlation between heat and cold tolerance in potato using TTC-reduction. They concluded that heat and cold resistance are mutually exclusive. It is possible that heat and drought tolerance are mutually exclusive in potato.
Steyn et al.25 subjected three of the cultivars used in this study ('Vanderplank', 'Hoëvelder' and 'Late Harvest') to differential water treatments in rain shelter trials in both autumn and spring. They showed that the effect of drought on yield was more detrimental in spring plantings than in autumn plantings. According to Steyn et al.,25 higher temperatures during spring trials may aggravate the effect of water stress. When our data were compared with those from the rain shelter trials, a negative correlation was found between results from the TTC-reduction and the rain shelter trials. For example, 'Late Harvest' was drought tolerant according to the results from the rain shelter trials but tested sensitive with the viability assay, while 'Vanderplank' was drought sensitive according to the results from the rain shelter trials and tolerant according to the TTC-reduction experiments. 'Hoëvelder' was sensitive to drought but had a high yield potential according to the rain shelter trials; with the viability assay, however, it tested more tolerant than 'Late Harvest'. In field studies, cultivar assessment is complicated by the interaction between heat and drought stress,1 thus the results from the rain shelter trials were compared to heat tolerance as estimated by TTC-reduction in potato leaves. The comparison between heat tolerance, as measured by TTC-reduction, and drought tolerance in rain shelter trials resulted in a positive correlation with 'Vanderplank' and 'Hoëvelder' testing sensitive and 'Late Harvest' tolerant.
We conclude that heat and drought tolerance are organ and cultivar specific. In addition to our results on tubers that showed a hypersensitive reaction to drought, Shimshi and Susnoschi26 found a linear relationship between reduction in tuber yield and soil moisture during drought. Vayda1 suggests that this relationship disguises a complex set of responses. A reduction in photosynthetic efficiency may play a role in yield loss during water stress as it influences carbon assimilation.27 Thus, we recommend using leaves in all subsequent experiments. According to Walter,28 stress responses during phenylpropanoid biosynthesis are preferentially expressed in order of heat shock, fungal elicitor and ultraviolet light. It is thus possible that heat plays a dominant role when plants are subjected to both heat and drought stress. This agrees with the visual observations of breeders and this interaction complicates the selection for drought tolerance (H.J. Vorster, pers. comm.).
We are grateful to the Water Research Commission for financial support.
1. Vayda M.E. (1994). Environmental stress and its impact on potato yield. In Potato Genetics, eds J.E. Bradshaw and G.R. Mackay, pp. 239–261. CAB International and Cambridge University Press, Cambridge. [ Links ]
2. Ishikawa M., Robertson A.J. and Gusta L.V. (1995). Comparison of viability tests for assessing cross-adaptation to freezing, heat and salt stresses induced by abscisic acid in brome grass (Bromus inermis Legss) suspension cultured cells. Plant Sci. 107, 83–93. [ Links ]
3. De Ronde J.A., Van der Mescht A. and Cress W.A. (1995). The biochemical responses of six cotton cultivars to heat stress. S. Afr. J. Sci. 91, 363–366. [ Links ]
4. Chen P.M. and Gusta L.V. (1982). Cold acclimation of wheat and smooth bromegrass cell suspension. Can. J. Bot. 60, 1207–1211. [ Links ]
5. Larcher W. (1980). Physiological Plant Ecology, 2nd edn. Springer-Verlag, Berlin. [ Links ]
6. Palta J.P., Levitt J.L. and Stadelmann E.J. (1977). Freezing tolerance of onion bulbs and significance of freeze induced tissue infiltration. Cryobiology 14, 614–619. [ Links ]
7. Sukumaran V.P. and Weiser C.J. (1972). An excised leaflet test for evaluating potato frost tolerance. Hort. Sci. 7, 467–468. [ Links ]
8. Wiest S.C., Good G.L. and Steponkus P.L. (1976). Evaluation of root viability following freezing by the release of ninhydrin reactive compounds. Hort. Sci. 11, 197–199. [ Links ]
9. Zhang M.I.N., Willison J.H.M. and Hall S.A, (1993). Measurement of heat injury in plant tissue by using electrical impedance analysis. Can. J. Bot. 71, 1605–1611. [ Links ]
10. Li P.H., Huner N.P.A., Toivio-Kinnucan M., Chen H.H. and Palta J.P. (1981). Potato freezing injury and survival, and their relationships to other stress. Am. Potato J. 58, 15–29. [ Links ]
11. Vratsons D. and Rossouw F.T. (1991). Heat shock protein synthesis in Solanum tuberosum, an inter-cultivar comparison. S. Afr. J. Sci. 87, 442–446. [ Links ]
12. Jinn T-L., Chiu C-C., Song W-W., Chen Y-M. and Lin C-Y. (2004). Azetidine-induced accumulation of class i small heat shock proteins in the soluble fraction provides thermotolerance in soybean seedlings. Plant and Cell Physiol. 45(12), 1759–1767. [ Links ]
13. Hosseini R and Mulligan B.J. (2002). Application of rice (Oryza sativa L.) suspension culture in studying senescence in vitro (I). Single strand preferring nuclease activity. EJB Electronic Journal of Biotechnology 5(1), online at www.ejb. org/content/vol5/issue1/full/8 [ Links ]
14. Verleysen H., Samyn G., Van Bockstaele E. and Debergh P. (2004). Evaluation of analytical techniques to predict viability after cryopreservation. Plant Cell, Tissue and Organ Culture 77(1), 11–21. [ Links ]
15. De Ronde J.A. and Van der Mescht A. (1997). Utilization of 2,3,5-triphenyltetrazolium chloride reduction as a measure of the interaction between drought tolerance simulation and heat tolerance in cotton. S. Afr. J. Sci. 93, 431–433. [ Links ]
16. Berridge M.V., Tan A.S., McCoy K.D. and Wang R. (1967). The biochemical and cellular basis of cell proliferation assays that use tetrazolium salts. Plant Physiol. 42, 1423–1426. [ Links ]
17. De Block M. and De Brouwer D. (2002). A simple and robust in vitro assay to quantify the vigour of oilseed rape lines and hybrids Plant Physiol, Biochem. 40(10), 845–852. [ Links ]
18. Van der Mescht A., De Ronde J.A. and Rossouw F.T. (1993). Drought related protein synthesis is cultivar and organ specific in potato. J. S. Afr. Soc. Hort. Sci. 3(2), 97–101. [ Links ]
19. Van der Mescht A., Visser A.F., De Ronde J.A. and Vorster H.J. (1992). Protein profiles during drought stress in potato. J. S. Afr. Soc. Hort. Sci 2(1), 55–57. [ Links ]
20. McKenzie H.A., (1969). pH, buffers and physiological media. In Data for Biochemical Research, eds R.M.C. Dawson, D.C. Elliot, W.H. Elliot and K.M. Jones, 2nd edn. Clarendon Press, Oxford. [ Links ]
21. Chen H.H., Shen Z.Y. and Li P.H. (1982). Adaptability of crop plants to high temperature stress. Crop Sci. 22, 719–725. [ Links ]
22. Rich P.R., Mischis L.A., Purton S. and Wiskich J.T. (2001). The sites of interaction of triphenyltetrazolium chloride with mitochondrial respiratory chains. FEMS Microbiol. Lett. 202(2), 181–187. [ Links ]
23. Van der Mescht A., De Ronde J.A. and Rossouw F. T. (1998). Superoxide dismutase, glutathione reductase and ascorbate peroxidase levels during drought stress in potato. S. Afr. J. Sci. 94, 496–498. [ Links ]
24. Van der Merwe T., Van Staden L., Van der Mescht A. and Laurie R. (1997). Evaluation of 2,3,5-triphenyltetrazolium chloride reduction as a measure of heat- and drought tolerance in Eucalyptus grandis. IUFRO Conference on Silviculture and Improvement of Eucalypts 4, 112–116. [ Links ]
25. Steyn J.M., Du Plessis H.F., Fourie P. and Hammes P.S. (1998). Yield response of potato genotypes to different soil water regimes in contrasting seasons of a subtropical climate. Potato Res. 41, 239–254 [ Links ]
26. Shimshi D. and Susnoschi M. (1985). Growth and yield studies of potato development in a semi-arid region. 3. Effect of water stress and amounts of nitrogen top dressing on physiological indices and on tuber yield and quality of several cultivars. Potato Res. 28, 177–191. [ Links ]
27. Ogren E. (1990). Evaluation of chlorophyll fluorescence as a probe for drought stress in willow leaves. Plant Physiol. 93, 1280–1285. [ Links ]
28. Walter M.H. (1989). The induction of phenylpropanoid biosynthetic enzymes by ultraviolet light or fungal elicitor in cultured parsley cells is overridden by a heat-shock treatment. Planta 177, 1–8. [ Links ]
Received 7 May 2006. Accepted 15 April 2008.
This article is accompanied by supplementary figures online at www.sajs.co.za
* Author for correspondence. E-mail: anette@mut.ac.za
Mienie A. and De Ronde J.A. (2008). A comparison of drought stress and heat stress in the leaves and tubers of 12 potato cultivars S. Afr. J. Sci. 104, 156–159.
















