Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.104 n.3-4 Pretoria Mar./Apr. 2008
RESEARCH LETTERS
Beaked whale mysteries revealed by seafloor fossils trawled off South Africa
Giovanni BianucciI, *; Klaas PostII; Olivier LambertIII
IDipartimento di Scienze della Terra, Università di Pisa, via S. Maria, 53, I-56126 Pisa, Italy
IINatuurhistorisch Museum Rotterdam, P.O. Box 23452, N-3001 Rotterdam, the Netherlands
IIIInstitut royal des Sciences naturelles de Belgique, Département de Paléontologie, rue Vautier, 29, B-1000 Brussels, Belgium
ABSTRACT
An unexpectedly large number of well-preserved fossil ziphiid (beaked whale) skulls trawled from the seafloor off South Africa significantly increases our knowledge of this cetacean family. The eight new genera and ten new species more than double the known diversity of fossil beaked whales and represent more than one-third of this family (fossil and extant). A cladistic parsimony analysis based on 18 cranial characters suggested that some of these fossil taxa belong to the three extant ziphiid subfamilies, whereas others might represent extinct ziphiid lineages. Such high fossil ziphiid diversity might be linked to the upwelling system and the resulting high productivity of the Benguela Current, which has been in place and influenced conditions of the shallower waters along the southwest coast of South Africa and Namibia since the Middle Miocene. Both fossil and extant South African beaked whale faunas show a wide range in body size, which is probably related to different dietary niches and to wide exploration of the water column. Moreover, most South African fossil ziphiids share two morphological traits with extant species, which indicates that some of the behaviours associated with these traits had likely already developed during the Neogene: 1) the absence of functional maxillary teeth—providing clear evidence of suction feeding; and 2) the heavy ossification of the rostrum in specimens assumed to represent adult males—a feature which likely helps prevent injury and damage on impact during male–male fighting.
Beaked whales (Cetacea, Ziphiidae), with 21 recognized extant species, are a highly diverse family of marine mammals. These medium- to large-sized cetaceans (4–12 m in length) feed mostly on deep-water squid. Because of their deep-sea habitat, elusive habits, and apparent low abundance, and despite their species' high diversity and large size, beaked whales are one of the least known groups of mammals. This is one of the few families of large mammals for which new species have been recently discovered both from morphological evaluations of stranded animals and genetic analyses to confirm species distinctiveness.1–4
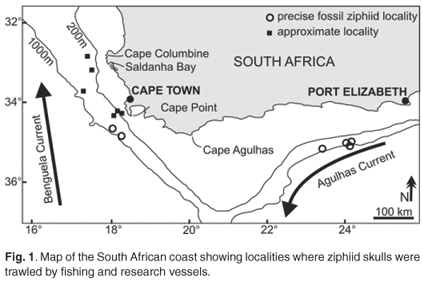
The fossil record of this family is relatively scarce compared to other whale groups.5–7 This could be at least partly due to the relatively rare inland outcropping of sediments originally deposited in deep marine environments in which ziphiids live. An alternative to traditional methods of fossil discovery for beaked whales and other deep-sea vertebrates is to trawl the deep-sea floor in search of outcropping fossils. Along the South African coast an exceptionally abundant and diversified association of fossil beaked whales was discovered at depths between 100 and 1000 m, and recently described in detail7 (Fig. 1). Two concomitant factors have contributed to the positive results of this research: 1) the intensive commercial and scientific deep-sea fishing activities aimed at exploring the seafloor in this area; and 2) the locally abundant outcrops of phosphorites (rocks containing over 5% P2O5), which are rich in Neogene fossil remains.8
Although the worldwide phenomenon of phosphatized, glauconized, and/or cemented fossil ziphiid remains dredged from sea beds has been noted before,9 the incredible abundance of relatively well-preserved fossil ziphiid remains off South Africa is both unique and exceptional. Based on the most significant specimens, no less than eight new genera and ten new species of fossil beaked whales have been described, more than doubling the previously known diversity of fossil beaked whales, and increasing by more than one-third the total diversity of this family, fossil and extant.
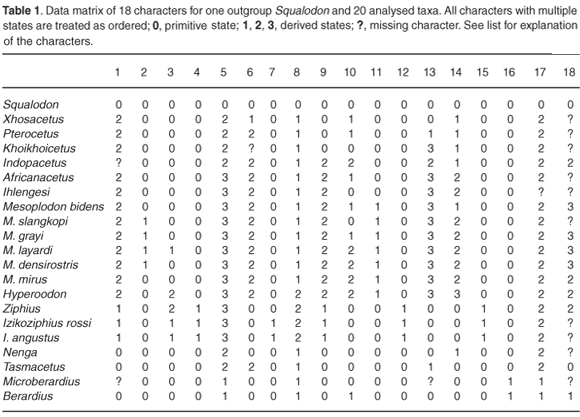
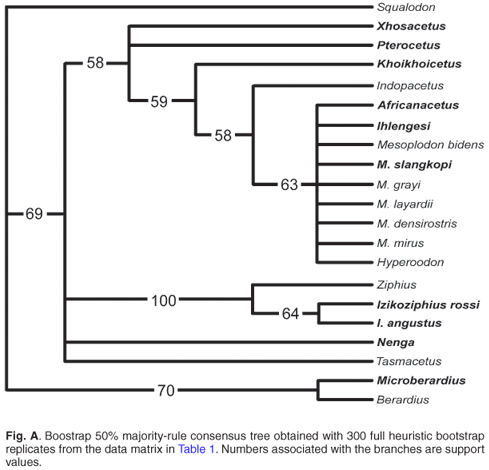
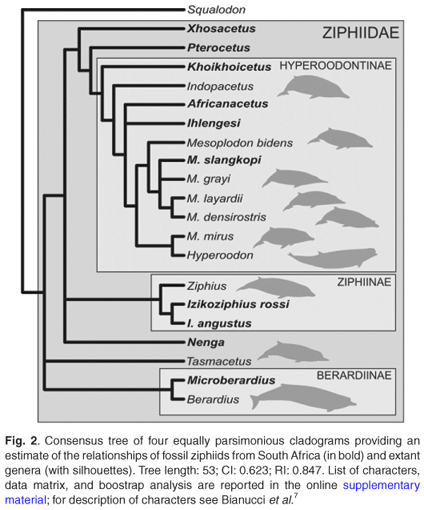
A phylogenetic parsimony analysis based on 18 cranial characters, including both fossil and extant ziphiid taxa, suggests that some of the South African fossil species belong to the three extant subfamilies, Berardiinae, Hyperoodontinae and Ziphiinae (as redefined by Bianucci et al.7), whereas others might represent hitherto unknown extinct ziphiid lineages (Fig. 2). Although exact stratigraphic information on these fossils is not available, a Middle–Late Miocene age may be proposed for most of them based on the main phosphogenic episodes known to have occurred in South African coastal waters since the latest Oligocene.8
To explain this past high ziphiid concentration and diversity off the South African coast, a direct link with the genesis of the Benguela Current and its related coastal upwelling area may be postulated. Nannofossil and sedimentary studies indicate that this upwelling system, which is driven by the entry of the northward-running cool oceanic Benguela Current into the shallower waters off the southwest coast of South Africa and Namibia, has been established at least intermittently since the Middle Miocene (c. 15 Myr ago),10 and was more consistently present during the Late Miocene (10 Myr ago).11 Furthermore, periods of intensified upwelling are recognized during the Late Miocene and the Early to early Late Pliocene.12
Recent studies of extant ziphiid habitats indicate strong links between feeding areas and the interaction of submarine topography with marine currents.13,14 Conditions along the South African coast favour such habitats and, indeed, at least nine ziphiid species are resident or occasional visitors in these waters.15–17 Ross15 divided the extant ziphiids of the South African southeast coast into different ecological groups based on body size and water-temperature preferences: (1) tropical/subtropical: Mesoplodon densirostris, associated with the Agulhas Current; (2) temperate/subantarctic: M. layardii, Hyperoodon planifrons, Berardius arnuxii (with probably more accidental records of M. hectori and M. grayi), in cool inshore water of the Agulhas Current; (3) cosmopolitan: Ziphius cavirostris; and (4) mixed cool and warm waters: M. mirus. Indopacetus pacificus, a recent addition to South Africa's extant ziphiid fauna,17 likely belongs to the first of these guilds.
The high diversity of fossil Neogene forms—even if we consider that it is not clear whether all species were contemporary— might suggest a similar division among several ecological niches. Among extant ziphiids, local coexistence of several species appears to be possible because of dietary (different prey size), temporal, or fine-scale geographic segregation.13,18 Taking into account the variation in general size, different dietary niches may logically be proposed for the fossil South African ziphiid fauna as well. Further supporting this hypothesis, the suction feeding adaptation, probably already realized in these beaked whales (see below), could have restricted the size of the preys that can be ingested by each species, depending on its own size in a way similar to what is observed in extant ziphiids.19
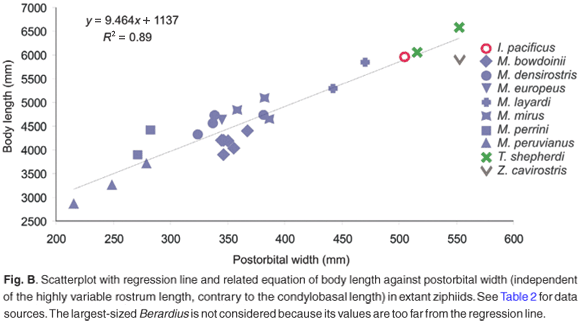
As the postcranial material is not held from any fossil ziphiid of South Africa, this leaves extrapolation from skull size as the only way to estimate the original body length of the animal. Using the equation given in the online additional material (see www.sajs.co.za), we estimated the body length for the South African fossil specimens and present the results in Fig. 3, where fossil and extant South African beaked whales are grouped into four size classes. Although smaller taxa are more numerous in the fossil sample than among extant species, both fossil and extant faunas show a wide range in size.

Considering the relationship between body size and diving capacity in cetaceans and other air-breathing vertebrates,20 we may postulate that the wide spectrum of sizes observed in South African fossil ziphiids also corresponds to a wide exploration of the water column by these whales for foraging. Although studies of the diving behaviour of extant beaked whales are scarce, some results are evidence that routinely very deep dives (up to 1000 m) are performed not only by the largest ziphiids such as Berardius, Hyperoodon, and Ziphius, but also by medium-sized ziphiids such as Mesoplodon.14,21–23 Nevertheless, considering that the aerobic dive limit is directly related to the body mass,23 we suggest that the largest South African fossil beaked whales may have been capable of diving deep for a longer time.
Although the South African fossil ziphiids clearly differ from each other in body size, most share two obvious common features: the strong reduction and/or absence of maxillary teeth and ossification of the rostrum. The extremely reduced dentition shared by all extant beaked whales, apart from Tasmacetus, is a morphological trait thought to be related to suction feeding, the process by which the prey is attracted to the mouth of a predator by rapid retraction and depression of the large piston-like tongue.24,25 The absence of functional maxillary teeth in all South African fossil ziphiids is clear evidence that efficient suction feeding had already developed in several beaked whale lineages during the Miocene. The strong mesorostral ossification of the rostrum is caused by the filling of the mesorostral canal with a pachyostosed vomer and/or mesethmoid. A similarly dense ossification occurs in the adult males of the extant Ziphius cavirostris and in most extant species of Mesoplodon. Three hypotheses have been proposed for the function of the mesorostral bones: assistance for deep diving (ballast),26 sound transmission,27 or prevention of damage during male–male fighting (that is, strengthening of the rostrum).28,29 McLeod30 re-discussed these three hypotheses, adding new observations on the already described28,29 intense scarring on the bodies of tusk-bearing adult males, and concluded that the hypothesis related to male–male interactions is most plausible. The presence of a similarly dense rostrum in most South African fossil ziphiids indicates that this behaviour was already present in several Miocene beaked whale lineages. It is therefore important to note that the relative abundance of pachyostosed rostra among the South African fossil ziphiids might have been artificially increased by selective fossilization favouring the preservation of the most robust skulls, probably belonging prevalently to adult males. Among extant ziphiids the males do not reach a significantly larger size than the females (except in Hyperoodon ampullatus); the preferential preservation of male skulls therefore probably did not produce a systematic bias towards larger sizes.31
In summary, the striking abundance of fossils on the seafloor off South Africa reveals an unusually high ziphiid diversity, which significantly unravels further the evolutionary history of these mysterious whales. Not only do some peculiar adaptations of the ziphiids prove to be of ancient origin, but for the first time we can provide an important amount of systematic information on the poorly known southern hemisphere fossil ziphiid faunas, especially those of South African waters. The large fossil ziphiid sample from the South African coast confirms that the extant ziphiid diversity might just be a remnant of a higher past diversity.16
We thank G. Avery, M. Avery and K. van Willingh (Iziko South African Museum, Cape Town), M. Smale, S. Ploen, and G. Watson (Port Elizabeth Museum, Port Elizabeth), and H. van der Es (Natuurhistorisch Museum Rotterdam, the Netherlands) for the access to the collections of South African fossil ziphiids. R. Leslie and J. Augustyn (Marine and Coastal Management, Cape Town) provided information on research vessel activities, and Kees Hazevoet (National Museum of Natural History, Lisbon) provided useful suggestions and comments on the text. We also thank two anonymous reviewers for their comments that greatly improved this manuscript. Thanks are also due to J.W.F. Reumer (NMR) for financial support enabling our study in South Africa. The work of O.L. at the IRSNB is financially supported by Research Project MO/36/016 of the Belgian Federal Science Policy Office.
1. Reyes J.C., Mead J.G. and Van Waerebeek. K. (1991). A new species of beaked whale Mesoplodon peruvianus sp. n. (Cetacea: Ziphiidae) from Peru. Mar. Mamm. Sci. 7, 1–24. [ Links ]
2. Dalebout M.L., Mead J.G., Baker C.S., Baker A.N. and van Helden A.L. (2002). A new species of beaked whale Mesoplodon perrini sp. n. (Cetacea: Ziphiidae) discovered through phylogenetic analyses of mitochondrial DNA sequences. Mar. Mamm. Sci. 18(3), 577–608. [ Links ]
3. van Helden A.L., Baker A.N., Dalebout M.L., Reyes J.C., Van Waerebeek K. and Baker C.S. (2002). Resurrection of Mesoplodon traversii (Gray, 1874), senior synonym of M. bahamondi Reyes, Van Waerebeek, Cardenas and Yañez, 1995 (Cetacea: Ziphiidae). Mar. Mamm. Sci. 18(3), 609–621. [ Links ]
4. Dalebout M.L., Baker C.S., Cockcroft V.G., Mead J.G. and Yamada T.K. (2004). A comprehensive and validated molecular taxonomy of beaked whales, family Ziphiidae. J. Heredity 95, 459–473. [ Links ]
5. Bianucci G. (1997). The Odontoceti (Mammalia: Cetacea) from Italian Pliocene. The Ziphiidae. Palaeont. ital. 84, 163–192. [ Links ]
6. Lambert O. (2005). Systematics and phylogeny of the fossil beaked whales Ziphirostrum du Bus, 1868 and Choneziphius Duvernoy, 1851 (Cetacea, Odontoceti), from the Neogene of Antwerp (north of Belgium). Geodiversitas 27(3), 443–497. [ Links ]
7. Bianucci G., Lambert O. and Post K. (2007). A high diversity in fossil beaked whales (Odontoceti, Ziphiidae) recovered by trawling from the sea floor off South Africa. Geodiversitas 29(4), 5–62. [ Links ]
8. Compton J.S., Wigley R.A. and McMillan I.K. (2004). Late Cenozoic phosphogenesis on the western shelf of South Africa in the vicinity of the Cape Canyon. Mar. Geol. 206, 19–40. [ Links ]
9. Whitmore F.C., Jr., Morejohn G.V. and Mullins H.T. (1986). Fossil beaked whales – Mesoplodon longirostris dredged from the ocean bottom. Natl Geog. Res. 2(1), 47–56. [ Links ]
10. Wigley R.A. and Compton J.S. (2006). Late Cenozoic evolution of the outer continental shelf at the head of the Cape Canyon, South Africa. Mar. Geol. 226, 1–23. [ Links ]
11. Krammer R., Baumann K-H. and Henrich R. (2006). Middle to late Miocene fluctuations in the incipient Benguela Upwelling System revealed by the calcareous nannofossil assemblages (ODP Site 1085A). Palaeogeogr. Palaeoclimatol. Palaeoecol. 230, 319–334. [ Links ]
12. Udeze C.U. and Oboh-Ikuenobe F.E. (2005). Neogene palaeoceanographic and palaeoclimatic events inferred from palynological data: Cape Basin off South Africa, ODP Leg 175. Palaeogeogr. Palaeoclimatol. Palaeoecol. 219, 199–223. [ Links ]
13. Waring G.T., Hamazaki T., Sheehan D., Wood G. and Baker S. (2001). Characterization of beaked whale (Ziphiidae) and sperm whale (Physeter macrocephalus) summer habitat in shelf-edge and deeper waters off the northeast U.S. Mar. Mamm. Sci. 17(4), 703–717. [ Links ]
14. McLeod C.D. and Zuur A.F. (2005). Habitat utilization by Blainville's beaked whales off Great Abaco, northern Bahamas, in relation to seabed topography. Mar. Biol. 147, 1–11. [ Links ]
15. Ross G.J.B. (1984). The smaller cetaceans of the south east coast of southern Africa. Ann. Cape Prov. Mus. (Nat. Hist.) 15(2), 173–410. [ Links ]
16. Mead J.G. (1989). Beaked whales of the genus Mesoplodon. In Handbook of Marine Mammals, vol. 4, River dolphins and the larger toothed whales, eds S.H. Ridgway and R. Harrison, pp. 349–430. Academic Press, London. [ Links ]
17. Dalebout M.L. Ross G.J.B., Baker C.S., Anderson R.C., Best P.B., Cockcroft V.G., Hinsz H.L., Peddemors V. and Pitman R.L. (2003). Appearance, distribution and genetic distinctiveness of Longman's beaked whale, Indopacetus pacificus. Mar. Mamm. Sci. 198, 421–461. [ Links ]
18. MacLeod C.D., Santos M.B. and Pierce G.J. (2003). A review of data on diets of beaked whales: evidence of niche separation and geographic segregation between three genera. J. Mar. Biol. Assoc. 83, 651–655. [ Links ]
19. MacLeod C.D., Santos M.B, Pierce G.J. and Lopez A. (2006). Relative prey size consumption in toothed whales: implications for prey selection and level of specialisation. Mar. Ecol. Prog. Ser. 326, 295–307. [ Links ]
20. Schreer J.F. and Kovacs K.M. (1997). Allometry of diving capacity in air-breathing vertebrates. Can. J. Zool. 75, 339–358. [ Links ]
21. Hooker S.K. and Baird R.W. (1999). Deep-diving behaviour of the northern bottlenose whale, Hyperoodon ampullatus (Cetacea: Ziphiidae). Proc. R. Soc. Lond. B 266, 671–676. [ Links ]
22. Minamikawa S., Iwasaki T. and Kishiro T. (2007). Diving behaviour of a Baird's beaked whale, Berardius bairdii, in the slope water region of the western North Pacific: first dive records using a data logger. Fish. Oceanogr. 16(6), 573–577. [ Links ]
23. Tyack P.L., Johnson M., Soto N.A., Sturlese A. and Madsen P.T. (2006). Extreme diving of beaked whales. J. Exp. Biol. 209, 4238–4253. [ Links ]
24. Heyning J.E. and Mead J.G. (1996). Suction feeding in beaked whales: morphological and experimental evidence. Contrib. Sci. 464, 1–12. [ Links ]
25. Werth A.J. (2006). Odontocete suction feeding: experimental analysis of water flow and head shape. J. Morphol. 267(12), 1415–1428. [ Links ]
26. Buffrénil V. de, Zylberberg L., Traub W. and Casinos A. (2000). Structural and mechanical characteristics of the hyperdense bone of the rostrum of Mesoplodon densirostris (Cetacea, Ziphiidae): summary of recent observations. Hist. Biol. 14, 57–65. [ Links ]
27. Zioupos P., Currey, J.D., Casinos A. and Buffrénil V. de (1997). Mechanical properties of the rostrum of the whale Mesoplodon densirostris, a remarkably dense bony tissue. J. Zool. 241(4), 725–737. [ Links ]
28. Mead J.G., Walker W.A. and Houck. W.J. (1982). Biological observations on Mesoplodon carlhubbsi (Cetacea: Ziphiidae). Smith. Contr. Zool. 344, 1–25. [ Links ]
29. Heyning J.E. (1984). Functional morphology involved in intraspecific fighting of the beaked whale, Mesoplodon calhubbsi. Can. J. Zool. 62, 1645–1654. [ Links ]
30. McLeod C.D. (2002). Possible functions of the ultradense bone in the rostrum of Blainville's beaked whale (Mesoplodon densirostris). Can. J. Zool. 80, 178–84. [ Links ]
31. MacLeod C.D. (2006). How big is a beaked whale? A review of body length and sexual size dimorphism in the Family Ziphiidae. J. Cet. Res. Manag. 7, 301–308 [ Links ]
Received 28 January. Accepted 20 March 2008.
This article is accompanied by supplementary material online at www.sajs.co.za
* Author for correspondence. E-mail: bianucci@dst.unipi.it
Bianucci G., Post K. and Lambert O. (2008). Beaked whale mysteries revealed by seafloor fossils trawled off South Africa. S. Afr. J. Sci. 104, 140–142.
LIST OF CHARACTERS USED IN THE CLADISTIC ANALYSIS
1: Mesorostral ossification of the vomer filling the mesorostral groove
(0) No.
(1) Ossification median, not showing a median suture between the lateral walls of the vomer in the rostrum base area.
(2) Ossification lateral or lateral and median, in any case showing a median suture between the lateral walls of the vomer in the rostrum base area.
2: Acute ventral margin of the alveolar groove posteriorly reaching the antorbital notch and visible in dorsal view as a strengthening of the rostrum base
(0) No.
(1) Yes.
3: Asymmetry of the premaxillary sac fossae
(0) Absent or weak, ratio between measurements 18 and 17 > 0.70.
(1) Moderate, ratio from 0.70 to 0.40.
(2) High, ratio < 0.40.
4: Premaxillary sac fossa laterally overhanging the maxilla
(0) No.
(1) Yes.
5: Ascending process of the premaxilla in lateral view
(0) Rectilinear.
(1) Slightly concave.
(2) Concave with posterodorsal portion vertical.
(3) Concave with posterodorsal portion partly overhanging the bony nares.
6: Constriction on the ascending process of the right premaxilla (between premaxillary sac fossa and premaxillary crest)
(0) Roughly absent, ratio between measurements 20 and 22 > 0.80.
(1) Moderate constriction, ratio from 0.80 to 0.61.
(2) Strong constriction, ratio <0.61.
7: Elliptical fossa on the anterior surface of the ascending process of the premaxilla
(0) No.
(1) Yes.
8: Vertex elevation
(0) Absent to weak, ratio between measurements 28 and 16 < 0.50.
(1) Moderate, ratio from 0.50 to 1.0.
(2) Strong, ratio > 1.0.
9: Premaxillary crest direction (taken on the anterior edge in dorsal view)
(0) No crest or crest transversely directed.
(1) Crest anterolaterally directed.
(2) Crest posterolaterally directed.
10: Width of the premaxillary crests
(0) Small, ratio between measurements 21 and 16 < 1.0.
(1) Moderate, ratio from 1.0 to 1.25.
(2) Large, ratio >1.25.
11: Distance between premaxillary crests
(0) Large, ratio between measurements 24 and 16 > 0.25.
(1) Reduced, ratio < 0.25.
12: Nasal elongation
(0) Anterior tip of nasal posterior to or in line with the premaxillary crest.
(1) Anterior tip of nasal anterior to the premaxillary crest.
13: Anteromedian excavation of the dorsal surface of the nasal
(0) No.
(1) Slight anteromedian concavity.
(2) Well-defined anteromedian depression.
(3) Deep excavation.
14: Inclusion of the nasal in the premaxillary crest
(0) No.
(1) For a short distance along the posteromedian angle of the premaxillary crest.
(2) Until about half-way along the median margin of the crest.
(3) Reaching the anteromedian margin of the crest.
15: Contact between nasal and premaxillary crest
(0) Wide, on the whole length of the premaxilla on the vertex.
(1) Reduced, on the posterior half of the nasal.
16: Interparietal as an isolated rounded protuberance on the posterior of the vertex
(0) No.
(1) Yes.
17: Anteromedian projection of the supraoccipital between the posterior margins of the maxillae
(0) No.
(1) Yes, lower than the vertex.
(2) Yes, roughly reaching dorsally the level of the vertex.
18: Number of alveoli on the mandible bearing teeth, erupted in adult males
(0) More than two pairs, associated with functional maxillary teeth.
(1) Two enlarged pairs, apical to sub-apical.
(2) One enlarged pair, apical.
(3) One enlarged pair, not apical.