Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.103 n.9-10 Pretoria Sep./Oct. 2007
RESEARCH LETTERS
Re-interpreting the evidence for bipedality in Homo floresiensis
Maria B. Blaszczyk; Christopher L. Vaughan
Department of Human Biology, Faculty of Health Sciences, University of Cape Town, Observatory 7925, South Africa
ABSTRACT
The unveiling in October 2004 of the remains of a pygmy-sized hominin recovered from a cave on the island of Flores, Indonesia, sparked an intense series of debates within the palaeoanthropology community. The discoverers diagnosed it to be a new species of Homo, which they called Homo floresiensis, and they interpreted the postcranial morphology as being 'consistent with human-like obligate bipedalism'. We have examined the morphology with the aim of determining whether biomechanical evidence supports the claim that this hominin—known as LB1—was indeed habitually bipedal. LB1's innominate bone differs from that of modern humans through the marked lateral flaring of the ilium, while her femur has a small head and a relatively long neck. Although these features are also found in australopithecines and are commonly regarded as 'primitive' traits, we concluded that none would have prevented her from exhibiting an efficient, bipedal gait. Having established that LB1 walked on two legs, we employed the principle of dynamic similarity to speculate how she might have walked. Assuming the gait of LB1 was dynamically similar to that of modern Homo sapiens, we used known dimensionless parameters, together with her leg length (0.55 m), to estimate her fundamental gait parameters: step length = 0.45 m, step frequency = 2.48 steps/second and speed = 1.11 m/s. Our review has provided insights regarding the way in which LB1 and her fellow diminutive hominins walked about the island of Flores over 18 000 years ago.
Introduction
One of the key traits that separate hominins from all other primates is the habitual use of a bipedal gait. Humans are the only extant species in the hominin group, and our bipedality is the most distinctive adaptation from our closest living relatives, the anthropoid apes.1, 2 This single characteristic is seen to be such a defining feature that skeletal adaptations to bipedalism are frequently used to identify our extinct hominin ancestors and relatives, and attempts at classification of these extinct species are often made on the basis of these adaptations. Wood and Collard,3 for example, have proposed that the fossil hominins be divided into two groups on the basis of locomotor inferences that have been made from their postcranial morphologies. The first group displayed a form of terrestrial bipedalism combined with an ability to climb proficiently, thus employing a mixed strategy, and include Praeanthropus, Australopithecus, and Homo habilis. The second group, consisting of Homo erectus, Homo ergaster, Homo heidelbergensis and Homo neanderthalensis, was committed to modern human-like terrestrial bipedalism, with a very limited arboreal facility. Wood and Collard3 furthermore argued that a fossil species should be included in the Homo genus only if it met the criteria of 'a postcranial skeleton whose functional morphology is consistent with modern human-like obligate bipedalism', and, on this basis, concluded that Homo habilis should be excluded from the genus. More recent analyses of the approximately 6-million-year-old Orrorin tugenensis fossils have, however, found this species' femur to be morphologically more like that of modern humans than australopithecines,4 which led Pickford et al.5 to conclude that Orrorin was a habitual biped. If this deduction is correct, the story of the evolution of bipedalism and its place in classificatory schemes is likely to change.
The unveiling in October 2004 of the remains of a pygmy-sized hominin recovered from a cave on the island of Flores, Indonesia, has sparked an intense series of debates, and the issue of how to classify the specimen is one of the major controversies.6–9 In describing the skeleton, catalogued as LB1, Brown et al.10 diagnosed it to be a new species of Homo, and thus gave it the name Homo floresiensis. Although many experts support the taxonomic decision of Brown and his colleagues, others have challenged their interpretation and offered alternative theories.8 These include the view that the primitive characteristics of LB1's anatomy suggest an ancestor such as Homo habilis, or that it might perhaps be an offshoot of Australopithecus.6
So what led Brown and colleagues to place LB1 in the genus Homo? Although they acknowledged that the LB1 skeleton showed a mosaic of primitive and derived traits, and were impressed by the characteristics LB1 shared with early hominins such as the australopithecines, they concluded that 'the facial and dental proportions, postcranial anatomy consistent with human-like obligate bipedalism, and a masticatory apparatus most similar in relative size and function to modern humans all support assignment to the genus Homo.'10 The recreation of the LB1 skeleton in the illustrative pose in National Geographic11 is certainly suggestive of a bipedal gait (see Fig. 1). The interpretation of the postcranial morphology as being 'consistent with humanlike obligate bipedalism' is thus one of the major pillars upon which the claim by Brown et al.10 rests.
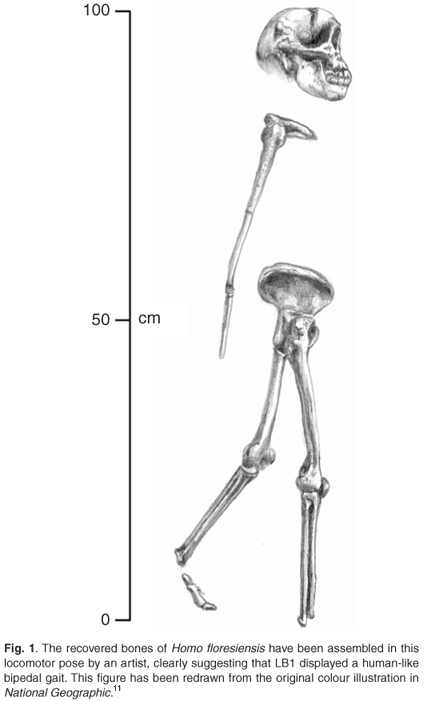
Features of the postcranial skeleton of Homo floresiensis
The recovery of the LB1 skeleton took place during an archaeological excavation at Liang Bua, Flores, in September 2003.12 The recovered elements of the postcranial skeleton included a fairly complete right leg and left innominate bone of the pelvis and less complete left leg, hands and feet. The vertebral column, sacrum, scapulae, clavicles and ribs were represented only by fragments, while the arms were reported more recently.13
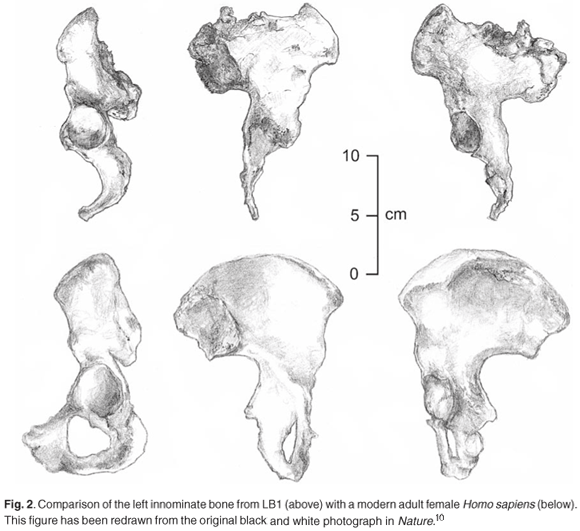
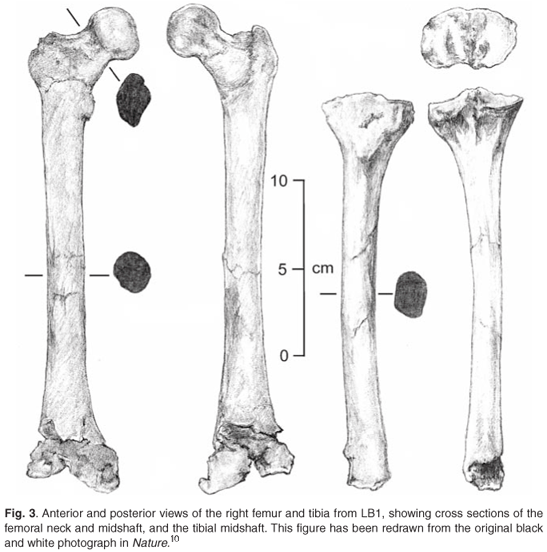
Describing the postcranial elements, Brown et al.10 noted that, 'in common with all bipedal hominins, the iliac blade is short and wide.' That is where their discussion of LB1's bipedality began and ended. They went on to describe the marked lateral flare of the ilium, whose blade would have projected more laterally relative to the plane of the acetabulum than in modern humans (Fig. 2), before going on to discuss the anatomy of the femur (Fig. 3). They asserted that the femur's overall anatomy was most consistent with the broad range of variation found in Homo sapiens, with, for example, strong development of the intertrochanteric crest as is characteristically found in our species.10 However, they also described the biomechanical neck length of the femur (55.5 mm) as being long relative to the femoral head diameter (31.5 mm), a feature of the australopithecines and early Homo (Table 1). They noted that several indices of femoral size and shape—the relationship between femoral head size and midshaft circumference (66 mm), and femoral length (280 mm) and sub-trochanteric shaft size (525 mm2)—fell within the chimpanzee and australopithecine range of variation.10,14 Their description of the tibia showed that the relationship between the midshaft cross section (347 mm2) and the length of the tibia (235 mm) was in the chimpanzee range of variation, and distinct from Homo.

In addition to structural characteristics related to requirements of bipedality, some of the most obvious differences between human and ape skeletons are 'proportional differences, involving not only the relative lengths of the long bones and trunk but also bone girth and joint surface size directly reflecting the different requirements of weight transfer through human and ape skeletons'.15 It is somewhat surprising that Brown et al.10 did not consider these proportions in their discussion on LB1's mode of locomotion. As several of the indices characterizing LB1's lower limb fall within the australopithecine and chimpanzee range of variation (Table 1), might it not follow that LB1's locomotor mode shared features with those predicted for australopithecines, or even those displayed by chimpanzees? What is the relative importance of these and other morphological features in predicting a species' mode of locomotion? A comparison of the biomechanical characteristics of bipedality and quadrupedalism and their respective morphological indicators in modern humans and our closest living relatives are helpful in answering these questions.
Morphological indicators of arboreal and bipedal locomotion
Quadrupedalism is the most common mode of locomotion among primates, although they certainly display a wide array of locomotor behaviours. Apes are, however, largely arboreal and thus the basic mechanical conditions of climbing in trees can be related to their skeletal anatomy.1 The most obvious requirement for a bipedal gait is an upright posture, which in turn requires that the centre of mass (CoM) of the body remain directly over the rectangular area formed by the supporting feet. This explains, for example, why chimpanzees, when walking bipedally, do so with a bent-hip/bent-knee (BHBK) gait.16 A BHBK gait is fatiguing if significant distances are walked,17 whereas the 'stiff-legged' gait of humans allows for an effective exchange of gravitational potential and kinetic energy, which minimizes energy consumption.18,19 Morphological features that allow extension of the lower limb joints during locomotion, such as the lumbar lordosis, are consequently seen to be indicators of a bipedal gait.
The ape pelvis displays several features common to other quadrupeds, such as ischial corpora that are elongated, and ilial blades that lie mostly in the coronal plane.20 The ape's long ischium is adapted to power the hip in extension by giving the hamstrings a long moment arm, while the lengthened ilium provides gluteus medius and minimus with great fibre length, which is mechanically advantageous for quadrupedal locomotion and during climbing.1 In contrast, the human pelvis is short, squat and basin-shaped. The human ilium is unique among primates by virtue of the fact that it is wider than it is high,15 thus eliminating any restrictive contact between the ilia and the lower lumbar vertebrae20 and facilitating bipedal posture. Another exclusively human feature is the greater sciatic notch on the sacral margin of the ilium, created by retroflexion of the ilium, and resulting in a far greater angle between the ilium and the ischium than that seen in apes. This allows the trunk to be held upright without compromising the biomechanical relationship between the ischium and the femur.15
Differences in the morphology of the femur in humans and apes include the degree of obliquity of the femoral shaft, its mediolateral curvature and cortical thickness, and the relative size of the femoral condyles. The latter three parameters reflect the difference in weight transfer during quadrupedal versus bipedal locomotion.15 In humans the knee joint is closer to the mid-line of the body, and placement of the feet facilitates a smoother and more efficient gait.15,16,21
Indicators of bipedality in the fossil record
One of the big debates in palaeoanthropology has centred on the locomotor mode of australopithecines, with the Australopithecus afarensis specimen AL-288-1, commonly known as 'Lucy', being particularly extensively studied because her locomotor anatomy is reasonably intact.22 Numerous studies have pointed out that the morphology of the pelvic and lower limb bones of Australopithecus present a mosaic pattern of features.23–25 They retain some primitive, ape-like traits, such as short lower limbs and relatively small lower limb and vertebral joints, but also have derived, more human-like features associated with bipedalism, including short, laterally facing iliac blades and valgus knees. In addition, Australopithecus displays unique features not found in either apes or humans, such as an exceptionally wide and laterally flared pelvis.26 The debates regarding the locomotor mode of Australopithecus are many, the literature is extensive, and a comprehensive review of these debates thus falls outside the scope of this paper. Certain areas of the debate are, however, relevant to the present discussion. As Brown et al.10 have pointed out, the postcranial skeleton of LB1, like that of AL-288-1, 'combines a mosaic of primitive, unique and derived features,' and the two specimens share certain morphological features, such as a broad pelvis with lateral flaring of the ilium and a long neck of femur in relation to femoral head diameter (Figs 2 and 3). These latter two features are often regarded as 'primitive' and have frequently been cited as evidence of incomplete bipedal adaptation in australopithecines,23,24 and an assessment of their implications for locomotion is thus required.
A small femoral head in relation to the femoral neck length has commonly been assumed to result in excessively high hip-joint stresses in bipedal stance, which would not be compatible with habitual bipedalism. Lovejoy et al.27 challenged this assumption by means of a biomechanical analysis of the static force and relative joint stress that would have acted on the femoral head of one australopithecine, Sts-14, as compared to that of the average of 20 Amerindians. The femoral head of Sts-14 is missing, but it was possible to make inferences from the femoral neck. They calculated the ratio of femoral head stress in Homo sapiens to that in Sts-14 to be 2.03 when normalized for total body weight. Thus, a small femoral head does not necessarily lead to high stresses; it is only one of the factors governing forces within the total pelvi-femoral complex.27 The same can be said for other features: inter-acetabular distance alone does not predict joint reaction forces; the extent of lateral flaring of the ilia and the femoral neck also need to be considered. An increase in either of these two factors will result in an increased lateral biomechanical lever arm, tending to reduce joint stresses. The biomechanical evidence therefore supports the assertion that morphological features such as lateral flaring of the ilia and long femoral necks are not evidence of inefficient bipedalism in Australopithecus; rather, if considered as a whole, they can be indicative of a human-like bipedal gait.
Another feature of Australopithecus that is sometimes cited as being reflective of an inefficient gait is short legs. Some anthropologists have equated short legs with increased energy expenditure because a greater stride length, made possible by longer limbs, will lead to reduced cadence for a given velocity.28 This means that the leg swings less often, so power requirements decrease, and less energy is used. As Kramer and Eck29 have pointed out, however, stride length is only one aspect governing energy expenditure. Longer legs may decrease cadence for a given velocity, but they also have a higher mass moment of inertia and as a result the power required in each step is increased, so it does not automatically follow that lengthening of the lower limb decreases power requirements. Steudel30 found no significant relationship between cost of locomotion versus limb length in 21 species of mammals, including walking and running in humans, while more recent data suggest that longer legs increase daily energy expenditure (DEE) in hominins, with the DEE for H. erectus estimated to be 84% greater than that of Australopithecus.31
The case for bipedality in Homo floresiensis
A cursory glace at the short, squat innominate bone of LB1, with its curved, ventrally extending iliac blades (Fig. 2), is enough to convince most people with a basic knowledge of comparative anatomy that this hominin walked on two legs. The ilium displays all of the main characteristics that distinguish the pelves of the Hominidae from those of their quadrupedal primate relatives: shortness, increase in breadth, and retroflexion to create the greater sciatic notch.15 These unique morphological characters have all been shown to be specific adaptations to bipedalism.
What is less obvious from LB1's locomotor anatomy is the type of bipedality and the degree to which it was displayed. LB1's innominate bone differs from that of modern humans through the marked lateral flaring of its ilium (Fig. 2), and her femur has a small head and a relatively long neck (Fig. 3). She also has relatively short legs, and the proportions of many of the characters of her lower limb bones fall within the chimpanzee and australopithecine range of variation (Table 1). Do these differences accordingly mean that the floresiensis gait was significantly different from that of sapiens? Not necessarily.
Proportional differences in long bones, which are traditionally studied through indices, are used to characterize a species and their relationships to other species (Table 1). For example, the humerofemoral index (ratio of lengths of humerus to femur) among the higher primates demonstrates that humans have unusually long lower limbs, and apes unusually short ones, in relation to their upper limbs, whereas arboreal Old World monkeys have a more or less isometric relationship.15 In this respect, arboreal quadrupedal monkeys resemble bipedal humans more closely than they do arboreal quadrupedal apes. Therefore, proportional differences in long bones, while commonly used in functional interpretations of interspecies relationships, cannot in and of themselves be good predictors of locomotor mode. The same is true of the length of lower limb bones in relation to the rest of the body; LB1's legs are short, but this would not have precluded her from having displayed an efficient bipedal gait. As we have argued and as Kramer and Eck29 have pointed out, because shorter legs require less power than do longer ones, to posit that short legs represent an incomplete adaptation to bipedal gait is not tenable.
Lastly, we turn to the so-called 'primitive' features of LB1's postcranial morphology, her broad pelvis and her femur with long neck and small head (Figs 2 and 3). If these features are interpreted from a purely comparative point of view, one might be led to expect that LB1's gait was distinct from that of modern humans—that is, differences in locomotor anatomy are consistent with differences in gait. This line of analysis might be carried even further, leading one to postulate that, as humans are the exemplar of efficient bipedalism among primates and their anatomy will reflect this, any deviation will tend to signal a less efficient gait. When scrutinized rigorously, however, these kinds of assumptions do not always hold water.25 A good example is the debunking of the myth that a smaller femoral head automatically means higher hip-joint stresses. On the contrary, a careful biomechanical analysis revealed that smaller heads can exist in combination with lower hip-joint stresses than those found in modern humans.27
The above example demonstrates the dangers of interpreting morphological features in isolation. Wide hips have been seen as a poor adaptation to bipedalism because they would increase hip joint reaction forces.32 That wider hips as a feature by themselves would do this is true, as they increase the lever arm at which body weight operates. But wide hips are not an isolated feature of LB1's anatomy—they are found co-existing with a marked lateral iliac flare and long neck of femur, two features which tend to act in the opposite direction to bring down reaction forces. When we consider the features of LB1's pelvis in relation to each other rather than in isolation, therefore, we can see that none of them would have prevented her from exhibiting an efficient, habitual, bipedal gait. Perhaps Kramer and Eck29 have accorded the big differentiating features their most likely significance, with arguments for another hominin's habitual bipedality: 'australopithecines like AL-288-1 were proficient bipeds not despite their short legs and wide pelves but, rather, because of them'. The same could be said of LB1.
Estimating plausible gait parameters for Homo floresiensis
Having established that LB1 walked on two legs, it is reasonable to ask how she walked. To assist in this endeavour, Vaughan and Blaszczyk33 have employed the principle of dynamic similarity, an extension of geometric similarity (where one object is made identical to another by multiplying all lengths by a constant factor). In dynamic similarity, all lengths, times and forces are multiplied by separate constants. In bipedal locomotion, two different systems exhibit dynamic similarity when they walk with equal Froude numbers (Fr),

where v is the speed of walking in m/s, g is the gravitational acceleration (9.81 m/s2) and LL is the leg length in metres.34 The Froude number has been adapted from naval architecture and allows the comparison of locomotion in different-sized bipeds.35 It follows that the independent gait parameters step length (SL) and step frequency (SF) may be normalized to yield dimensionless parameters

where the product of dimensionless step length (λ) and dimensionless step frequency (ϕ) combine to yield dimensionless speed β (= λ × ϕ)36 and the substitution of Equations (2) and (3) yields the square root of the Froude number:

Equations (2), (3) and (4) enable us to gain key insights regarding plausible gait parameters for LB1. Based on a sample of 669 normal subjects, ranging in age from children aged 5 all the way up to elderly adults in their 90s, we have shown that the dimensionless parameters for dynamically similar gait have values of λ = 0.814 ± 0.041, ϕ = 0.588 ± 0.023, and β = 0.478 ± 0.025.35 These values may be contrasted with data for children younger than 5 during neuromaturation,36 or children with cerebral palsy evaluated before and after surgery.37
Vaughan and Blaszczyk33 argue that the anthropometric indices of H. floresiensis and H. sapiens support the assertion that the locomotor morphologies of these two species are geometrically similar, and they have assumed that the gait of LB1 was dynamically similar to that of modern Homo sapiens. This makes it possible to add the length of LB1's femur (280 mm) and tibia (235 mm) as provided by Brown et al.,10 plus an estimate of her malleolar height (35 mm), to yield a leg length LL = 0.55 m. Equations (2), (3) and (4) then yield values of SL = 0.45 ± 0.02 m, SF = 2.48 ± 0.10 steps/second, and v = 1.11 ± 0.06 m/s (Fig. 4).
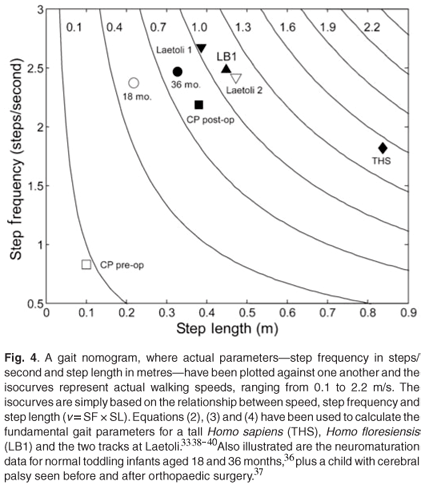
For purposes of comparison, the same equations may be applied to the fossilized footprints discovered in Laetoli, East Africa by Leakey and Hay,38 which were formed in wet volcanic ash almost 4 million years ago by two hominins.18 Charteris et al.39 reported the step lengths (SL) of the two tracks as 0.387 m and 0.472 m. By assuming dynamic similarity for the gait of these two australopithecines,33 Equation (2) yields leg length (LL) and then Equation (3) provides an estimate of step frequency (SF). As seen in Fig. 4, the values for LB1 lie between those for the two Laetoli hominins. Also included in Fig. 4 are values for a tall Homo sapiens (height of 2.01 m and LL = 1.028 m), two cohorts of normal children at age 18 and 36 months,18 and a child with cerebral palsy seen before and after surgery.37 The tall Homo sapiens with LL = 1.028 m has a longer step length (SL = 0.89 m), a lower step frequency (SF = 1.85 steps/s), and a much greater walking speed (v = 1.63 m/s) than LB1 and the Laetoli hominins.33
The values for the walking speeds of the Laetoli hominins (1.03 and 1.14 m/s) were greater than the values predicted by Charteris et al.39 and Alexander,40 although a more recent analysis based on evolutionary robotics has predicted speeds for these australopithecines in excess of 1.0 m/s.41 In the case of LB1, her anthropometric indices are very similar to the Australopithecus afarensis specimen AL-288-1 ('Lucy'), but somewhat different from a modern female Homo sapiens (Table 1). While these differences cannot be ignored, especially when applying dynamic similarity theory, Crompton et al.17 have demonstrated that Lucy's gait would have been comparable to that of modern humans.
Although the theory of dynamic similarity has been successfully applied across several different species,34,35 some authors have concluded that Froude number corrections are not adequate predictors of the mass-specific cost of transport.42 Despite such findings, we believe it is quite plausible that LB1, with a height estimated to be approximately 1.06 m,10 walked efficiently at a speed of just more than 1.0 m/s.33
Concluding remarks
The island of Flores is approximately 350 km long from west to east, 80 km at its widest point from north to south, and occupies an area of 14 300 km2 (ref. 11), which is approximately the same size as the state of Connecticut in the U.S. If LB1 had walked at her natural speed, it would have taken her about 88 hours to traverse the length of the island, assuming continuous walking along a straight and flat path. By comparison, the tall Homo sapiens in Fig. 4 would have taken approximately 64 hours to cover the same course, that is, about a day less than LB1.
Interestingly, the discoverers of LB1 have postulated that the Homo floresiensis species may have arisen from an ancestral population of Homo erectus and that their long-term isolation on Flores resulted in endemic dwarfing.10,11 Speed of walking is proportional to the square root of leg length [see Equation (4)], but energy expenditure for walking at a particular speed is proportional to body mass.43 Since body mass is proportional to volume (for a particular density), energy utilization is roughly proportional to the cube of leg length. This suggests that a reduction in size resulting from dwarfism would have had a far more dramatic impact on energy consumption than on walking speed. Thus, a shorter LL may have slightly curtailed LB1's ranging behaviour (a 27% reduction in walking speed compared to tall Homo sapiens), but this would have been more than offset by the significantly lower calorie requirements (an 85% reduction in energy expenditure compared to tall Homo sapiens).
Dynamic similarity is not an exact science,34,35 but it does nevertheless provide us with some insights regarding the way in which LB1 and her fellow pygmy-sized hominins might have walked about the island of Flores over 18 000 years ago.
We gratefully acknowledge the financial support of the Medical Research Council and National Research Foundation of South Africa, and the Wellcome Trust of Great Britain. Annelie O'Hagen drew the skeletal illustrations in Figs 1–3.
1. Preuschoft H. (2004). Mechanisms for the acquisition of habitual bipedality: are there biomechanical reasons for the acquisition of upright bipedal posture? J. Anat. 204, 363–384. [ Links ]
2. Robinson J.T. (1972). Early Hominid Posture and Locomotion. University of Chicago Press, Chicago. [ Links ]
3. Wood B.A. and Collard M. (1999). The human genus. Science 284, 65–71. [ Links ]
4. Sawada Y., Pickford M., Senut B., Itaya T., Hyodo M., Miura T., Kashine C., Chujo T. and Fujii H. (2002). The age of Orrorin tugenensis, an early hominid from the Tugen Hills, Kenya. C. R. Palevol. 1, 293–303. [ Links ]
5. Pickford M., Senut B., Gommery D. and Treil J. (2002). Bipedalism in Orrorin tugenensis revealed by its femora. C. R. Palevol. 1, 191–203. [ Links ]
6. Wong K. (2005). The littlest human. Sci. Am. 292(2), 40–49. [ Links ]
7. Dalton R. (2004). Little lady of Flores forces rethink of human evolution. Nature 431, 1029. [ Links ]
8. Balter M. (2005). Small but smart? Flores hominid shows signs of advanced brain. Science 307, 1386–1389. [ Links ]
9. Lahr M.M. and Foley R. (2004). Human evolution writ small. Nature 431, 1043–1044. [ Links ]
10. Brown P., Sutikna T., Morwood M.J., Soejono R.P., Jatmiko, Saptomo E.W. and Due R.A. (2004). A new small-bodied hominin from the Late Pleistocene of Flores, Indonesia. Nature 431, 1055–1061. [ Links ]
11. Morwood M., Sutikna T. and Roberts R. (2005). The people time forgot. National Geographic April, 2–15. [ Links ]
12. Morwood M.J., Soejono R.P., Roberts R.G., Sutinka T., Turney C.S.M., Saptomo E.W. and Due R.A. (2004). Archaeology and age of a new hominin from Flores in eastern Indonesia. Nature 431, 1087–1091. [ Links ]
13. Morwood M.J., Brown P., Jatmiko, Sutinka T., Saptomo E.W., Westaway K.E., Due R.A., Roberts R.G., Maeda T., Wasisto S. and Djubiantono T. (2005). Further evidence for small-bodied hominins from the Late Pleistocene of Flores, Indonesia. Nature 437, 1012–1017. [ Links ]
14. Haeusler M. and McHenry H.M. (2004). Body proportions of Homo habilis reviewed. J. Hum. Evol. 46, 433–465. [ Links ]
15. Aiello L.C. and Dean M.C. (1990). An Introduction to Human Evolutionary Anatomy. Academic, London. [ Links ]
16. Jenkins F.A.J. (1972). Chimpanzee bipedalism: cineradiographic analysis and implications for the evolution of gait. Science 178, 877–879. [ Links ]
17. Crompton R.H., Li Y., Wang W., Gunther, M.M. and Savage R. (1998). The mechanical effectiveness of erect and bent-hip, bent-knee bipedal walking in Australopithecus afarensis. J. Hum. Evol. 35, 55–74. [ Links ]
18. Vaughan C.L. (2003). Theories of bipedal walking: an odyssey. J. Biomech. 36, 513–523. [ Links ]
19. Cavagna G.A., Heglund N.C. and Taylor C.R. (1977). Mechanical work in terrestrial locomotion: two basic mechanisms for minimizing energy expenditure. Am. J. Physiol. 233, 243–261. [ Links ]
20. Lovejoy C.O. (2005). The natural history of human gait and posture I: Back and pelvis. Gait Posture 21, 95–112. [ Links ]
21. Schmitt D. (2003). Insights into the evolution of human bipedalism from experimental studies of humans and other primates. J. Exp. Biol. 206, 1437–1448. [ Links ]
22. Johanson D.C., Lovejoy C.O., Kimbel W.H., White T.D., Ward S.C., Bush M.B., Latimer B.L. and Coppens Y. (1982). Morphology of the Pliocene partial hominid skeleton (AL 288-1) from the Hadar Formation, Ethiopia. Am. J. Phys. Anthropol. 57, 403–451. [ Links ]
23. Berge C. (1994). How did the autralopithecines walk? A biomechanical study of the hip and thigh in Australopithecus afarensis. J. Hum. Evol. 26, 259–273. [ Links ]
24. Stern J. and Susman R. (1983). The locomotor anatomy of Australopithecus afarensis. Am. J. Phys. Anthropol. 60, 279–317. [ Links ]
25. Ward C.V. (2002). Interpreting posture and locomotion of Australopithecus afarensis: Where do we stand? Yrbk Phys. Anthropol. 45, 185–215. [ Links ]
26. Rak Y. (1991). Lucy's pelvis anatomy, its role in bipedal gait. J. Hum. Evol. 20, 273–290. [ Links ]
27. Lovejoy C.O., Heiple K.G. and Burnstein A.H. (1973). The gait of Australopithecus. Am. J. Phys. Anthropol. 38, 757–780. [ Links ]
28. Jungers W.L. (1982). Lucy's limbs: skeletal allometry and locomotion in Australopithecus afarensis. Nature 297, 676–678. [ Links ]
29. Kramer P.A. and Eck G.G. (2000). Locomotor energetics and leg length in hominid bipedality. J. Hum. Evol. 38, 651–666. [ Links ]
30. Steudel K. (1996). Limb morphology, bipedal gait, and the energetics of hominid locomotion. Am. J. Phys. Anthropol. 99, 345–356. [ Links ]
31. Steudel-Numbers K.L. (2006). Energetics in Homo erectus and other early hominins: the consequences of increased lower-limb length. J. Hum. Evol. 51, 445–453. [ Links ]
32. Hunt K.D. (1994). The evolution of human bipedality: ecology and functional morphology. J. Hum. Evol. 26, 183–202. [ Links ]
33. Vaughan C.L. and Blaszczyk M.B. (in press). Dynamic similarity predicts gait parameters for Homo floresiensis and the Laetoli hominins. Am. J. Hum. Biol. [ Links ]
34. Alexander R.M. (1976). Estimates of speeds of dinosaurs. Nature 261, 129–130. [ Links ]
35. Vaughan C.L. and O'Malley M.J. (2005). Froude and the contribution of naval architecture to our understanding of bipedal locomotion. Gait Posture 21, 350–362. [ Links ]
36. Vaughan C.L., Langerak N.G. and O'Malley M.J. (2003). Neuromaturation of human locomotion revealed by non-dimensional scaling. Exp. Brain Res. 153, 123–127. [ Links ]
37. Vaughan C.L. and O'Malley M.J. (2005). A gait nomogram used with fuzzy clustering to monitor functional status of children and young adults with cerebral palsy. Dev. Med. Child Neurol. 47, 377–383. [ Links ]
38. Leakey M.D. and Hay R.L. (1979). Pliocene footprints in the Laetoli beds at Laetoli, northern Tanzania. Nature 278, 317–323. [ Links ]
39. Charteris J., Wall J.C. and Nottrodt J.W. (1982). Pliocene hominid gait: new interpretations based on available footprint data from Laetoli. Am. J. Phys. Anthropol. 58, 133–144. [ Links ]
40. Alexander R.M. (1984). Stride length and speed for adults, children, and fossil hominids. Am. J. Phys. Anthopol. 63, 23–27. [ Links ]
41. Sellers W.I., Cain G.M., Wang W. and Crompton R.H. (2005). Stride lengths, speeds and energy costs in walking of Australopithecus afarensis: using evolutionary robotics to predict locomotion of early human ancestors. J. R. Soc. Interface 2, 431–441. [ Links ]
42. Steudel-Numbers K.L. and Weaver T.D. (2006). Froude number corrections in anthropological studies. Am. J. Phys. Anthopol. 131, 27–32. [ Links ]
43. Zarrugh M.Y., Todd F.N. and Ralston H.J. (1974). Optimisation of energy expenditure during level walking. Eur. J. Appl. Physiol. 33, 293–306. [ Links ]
Received 2 August. Accepted 4 October 2007.
Author for correspondence. E-mail: kit.vaughan@uct.ac.za














