Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.103 n.9-10 Pretoria Sep./Oct. 2007
HISTORY OF SCIENCE
Louis Pasteur, fermentation, and a rival
K.L. Manchester
School of Molecular and Cell Biology, University of the Witwatersrand, Private Bag 3, WITS 2050, South Africa. E-mail: keith.manchester@wits.ac.za
Accusations of plagiarism, probably unjustified, concerning two
eminent scientists over the first demonstration of fermentation by
living organisms, still persist after a century and a half
ONE HUNDRED AND FIFTY YEARS AGO, IN August 1857, Louis Pasteur gave a lecture to the Société des Sciences de Lille entitled 'Lactate fermentation', published soon after as a Mémoire.1 The title may sound unexciting, but this historic paper marked Pasteur's first statement of a germ theory and a specific role for microorganisms in metabolic processes. Pasteur was then 34 and professor of chemistry and dean of the new Faculté des Sciences in Lille (Fig. 1), in the heart of the sugar beet growing region of northern France. Pasteur's appointment was in part intended to give help to industrialists in Lille, but the opening sentence of his paper makes clear that he was led to consider fermentations following his researches on the amyl alcohols and their remarkable crystallographic properties.2 His studies on fermentations led to a protracted dispute with a contemporary, Antoine Béchamp, over the priority of his findings which, remarkably, is maintained to this day and with which the latter part of this article deals.

Pasteur's work on fermentation
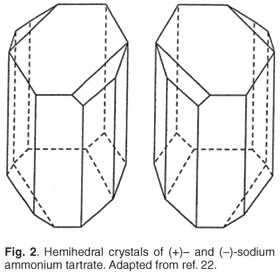
It will be recalled that, in 1846, as a student at the École Normale Supérieure in Paris, Pasteur had had the remarkable perspicacity and good fortune to notice that the sodium ammonium salt of paratartaric acid, a form of tartaric acid that was indistinguishable chemically from the commonly occurring tartaric acid except that solutions were not optically active, crystallized to give mixtures of two mirror image enantiomorphic forms (Fig. 2). These crystal forms, if carefully separated by hand and then dissolved in water, gave optically active solutions of opposite rotations.3 Given the optical activity of many organic compounds of natural origin, Pasteur had deduced that asymmetry (Pasteur used the term 'dissymmetry') was a feature of the chemistry of life and that this would correlate with hemihedral crystals. Optically active amyl alcohol recovered in distillates from alcoholic fermentations, particularly of grains, did not fit with this rule, and this discovery had led him to a special interest in the amyl alcohols. Nevertheless, and a little confusingly, the paper presented in Lille dealt instead with lactic fermentation,1 Pasteur declaring that: 'I intend to establish that, just as there is an alcoholic ferment, the yeast of beer, which is found everywhere that sugar is decomposed into alcohol and carbonic acid, so also there is a particular ferment, a lactic yeast, always present when sugar becomes lactic acid.' And this is precisely what he found. Critics have pondered over the years whether Pasteur formed his views on the basis of the evidence or decided what the result must be and proceeded to prove it!
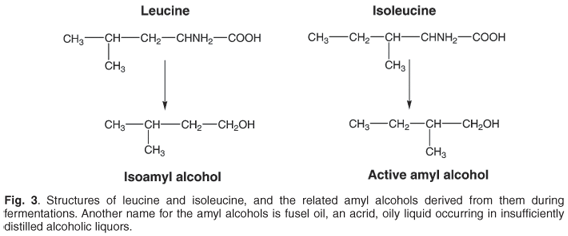
A prevailing view promoted by Justus von Liebig, up to this time, was that fermentation represented a form of decomposition, perhaps initiated by yeasts but proceeding essentially as a result of instabilities arising in molecules in fermentation liquors. On this view, amyl alcohol possessed optical activity because it retained some of the properties of the fermenting sugars. Pasteur considered that amyl alcohol was too dissimilar to sugars for this to be so (in which he was correct), and that its optical activity derived from the living organism producing it (in which he was wrong). Subsequent work showed that the two amyl alcohols of fermentation constitute breakdown products of the amino acids leucine and isoleucine, present in nitrogenous materials in the fermentation media. Although both leucine and isoleucine exist in dextro (+) and laevo (–) rotatory forms, their equivalent degradation to optically inactive isoamyl acohol and (optically) active amyl alcohol results in loss of the asymmetry of a carbon atom in isoamyl alcohol, which is retained in active amyl alcohol (Fig. 3). To Pasteur's considerable distress, the two alcohols, as their barium sulphamylates, had exactly the same crystal form.2
Fermentation correlative with life
Yeast, when incubated with sugar alone, gradually disintegrates. Pasteur recognized this event as one of the most important points in Liebig's theory of fermentation.4 If fermentation, Liebig argued, is a consequence of the development and multiplication of cells, as others claimed, incubations containing sugar alone should not produce alcohol, since such a medium lacks the other essential conditions for cell growth and division. Nevertheless, alcohol is produced under these conditions.
Pasteur showed that it was as a result of the growth of yeast cells, which can feed off the remnants of dead cells, that fermentation occurs.4 Thus, he reached the celebrated conclusion that 'the breakdown of sugar into alcohol and carbonic acid is an action correlating with a vital phenomenon', that is, fermentation is a property of living cells. Pasteur was also able to refute Liebig's claim by showing that yeast grows and ferments sugar in medium devoid of albuminoid (proteinaceous) material, although containing ammonia and salts.5
But having made one clear and important advance, Pasteur now risked pushing 0himself into a potentially dubious position, this time in relation to vitalism, that is that certain metabolic properties of living matter cannot be observed outside the cell and that only those fermentations carried out by cells were to be regarded as 'proper' fermentations. There was clear evidence of soluble ferments (that is, enzymes) that operated outside cells, two of the best-known examples being the actions of diastase on starch and invertase on sucrose. Thus it was not the ferment that was living, but the cells that produced it, a point established unequivocally only with Buchner's production, 40 years later in 1897, two years after Pasteur's death, of a juice from yeast capable of ethanolic fermentation.6 Pasteur was aware of such a possibility but was never able experimentally to show it, and we know that in Buchner's case the discovery was largely a matter of luck.
Antoine Béchamp
While Pasteur was busy with his fermentations, another chemist was also actively studying the possible influence of living organisms to bring about fermentation. Pierre Jacques Antoine Béchamp was born the son of a miller in 1816 (Pasteur, the son of a tanner, in 1822). Initially studying pharmacy in Strasbourg and teaching in various of the faculties in the university, in 1854 he succeeded Pasteur (who had moved to Lille) as professor of chemistry. In 1856 he was appointed professor of medical chemistry and pharmacy in the faculty of medicine of the University of Montpellier (Fig. 4), where he worked for 20 years. Béchamp believed that a question of pure chemistry often turns into one of 'subtle physiology'. (Presumably the same could be said of Pasteur, where his academic study of stereoisomerism led to fermentation.) Béchamp was anxious to establish whether the partial inversionª (hydrolysis) to glucose and fructose of cane sugar, dissolved in water and left to stand in stoppered bottles at room temperature for up to nine months, was the result of an action of water alone or had some other cause.

His first published work on this topic,7 in 1855, was to show that, if to the sucrose solution he added 25% calcium or zinc chloride, no inversion was observed. In the solution without salts some mould appeared within a month, but in subsequent months it did not increase in extent, though inversion was then taking place. At this date it was known that acids could bring about inversion of sucrose. Béchamp concluded from this experiment that the acidity of salts was not comparable to the acidity of an acid, yet water acted on the sucrose by virtue of its acid nature, although not showing an acid reaction with coloured indicators. He was surprised to see no effect of zinc chloride, since it showed an acid reaction. In this paper, Béchamp makes no suggestion that the appearance of the mould was a significant factor in the occurrence of inversion. Whether the thought had passed through his mind we do not know, which is unfortunate because it is this uncertainty which constitutes the basis of the controversy, which began to erupt around this time, over priority between Béchamp and Pasteur as to who first showed the production of ferments by living organisms.
In a subsequent paper,8 however, published at the beginning of 1858, Béchamp pointed out that experiments he had conducted since 1855 forced him to modify his earlier conclusions. He now believed that cold water alone does not invert sucrose, but that the reaction, when it occurs, is the result of a 'true fermentation'. He concluded that moulds do not develop in the absence of air and, in their absence, inversion does not occur; but if simple solutions of sucrose are in contact with air, then moulds develop and inversion occurs in proportion to the development of the mould. With these findings, Béchamp also regarded himself as the first to show the action of ferments in sugar solutions in the absence of proteinaceous material, that is, the ferment had to arise within the living cell.
Controversy between Béchamp and Pasteur in the academies
Things began quietly. At a meeting of the Sociétés des Savantes in 1862, Pasteur, in the presence of Béchamp, claimed precedence for showing the appearance of living organisms in a medium devoid of albuminoid matter. The meeting report9 reads:
M. Béchamp quoted some experiments, in which the transformation of cane sugar into grape sugar, brought about under the influence of air, is always accompanied by moulds. These experiments agree with the results obtained by M. Pasteur, who hastened to acknowledge that the fact put forward by M. Béchamp is one of the most rigid exactness.
The Académie des Sciences was an important venue for airing and developing views and a place to put forward novel ideas. In 1864, Béchamp felt moved to present a Mémoire10 in which he suggested that only soluble ferments (like invertase, to which he gave the name zymase) were constant in their actions. The organized ferments (Pasteur's 'properly called' fermentations) generated variable amounts of products, according to circumstances, because they reflected the nutritional activities of cells which consume organic materials, breaking them down and converting them into simpler forms. It often requires several successive fermentations (in modern terms, several enzymes) to produce the total effect. For Béchamp, alcoholic fermentation and the fermentations by organized ferments are not 'properly called' fermentations—they are simply manifestations of nutrition, a remarkably prescient idea. In 1872 we again find Béchamp telling the Academy11 that he believed he was the first to point out that organized ferments can develop in media in the absence of proteinaceous material and that fermentation is essentially an act of nutrition which includes excretion.
Conflict erupted yet again at the international medical congress held in London in 1881. Describing a session concerning the role of bacteria in disease,12 Béchamp, now from Lille, wrote:
M. Pasteur began to lecture and suddenly, in my presence, before I had said a word, he condemned me in a general anathema towards all aspects of heterogenesis.b I was waiting to speak, because I was due to lecture after him. But soon I was obliged to go down from my place to the front to sit opposite M. Pasteur because he had dared to say "that even if there were any points in my results, I had only incorporated his ideas and made them mine". In short M. Pasteur had just claimed a priority of views and made an accusation of unprecedented plagiarism. In an indignant voice I demanded of M. Pasteur to prove his assertion, since I would myself show him that the contrary was true. M. Pasteur, refusing a public discussion, left the session.
Béchamp goes on to say that The Times newspaper carried full details of the incident. Actually, The Times report of 8 August13 was more restrained and the summary of Pasteur's lecture (delivered in French, as was Béchamp's) was directed towards criticism of the work of Charlton Bastian over spontaneous generation. However, Béchamp is reported to have 'affirmed that the microzymas in chalk [see below] did exist and that if Pasteur has not obtained such results it was because his experiments were badly conducted. Béchamp held that the cause of disease and death lay in the animal itself.' The report of the same session in the British Medical Journal14 describes Béchamp as 'vindicating his claim to priority in the discovery of the organisms [microzymes] which caused the fermentation of milk. He also defended the accuracy of his experimental methods from the aspersions cast upon them by Pasteur.'
Études sur la Bière versus Les Microzymas13
In his book Les Microzymas, published in 1883, Béchamp describes how Pasteur, in 1876 in his Études sur la Bière,15 'in cold blood' tried to demolish him once and for all. Pasteur wrote:
The first note of Béchamp on the inversion of sucrose is in 1855.7 There is no mention there of the influence of moulds, the second where he states this influence is of 4 January 1858,8 after my work on lactic fermentation, published 30 November 18571 where I establish for the first time that the lactic ferment is an organized living being, that albuminoid materials do nothing in the cause of fermentation, after also my first work on alcoholic fermentation published on 21 December 1857.4 What is certain, one is at pains to point out, is that Béchamp, who since 1855 has not suggested the action of moulds on sugar, although he had noted their presence, has now modified his former conclusions.
The implications of this is that the change of Béchamp's ideas, which took place between his first paper in 18557 and the note which appeared in January 1858,8 occurred after he had heard of Pasteur's work as presented to the Academy in November1 and December4 1857. To us, familiar with long intervals between submission of a manuscript and its eventual publication, this charge would seem improbable, but on occasion publication could be very rapid.
Béchamp's answers in Les Microzymas12 to Pasteur's accusations are, first, outrage that Pasteur could make such suggestions, and secondly that all his (Béchamp's) new ideas were contained in his memoir of 1857. Unfortunately, the latter does not exist, but an apologist16 describes how this memoir is his paper in Annales de Chimie,17 which for unknown reasons appeared only in September 1858. Dates of submission of manuscripts are not indicated in the final publication.
It is interesting to note, in parenthesis, how both Béchamp and Pasteur started their careers more as physical scientists than biologists, but were gradually led into biology through the study of aspects of fermentation, then turned their attention to the diseases of man and of higher animals. Béchamp, like Pasteur, also worked on the diseases of wine and of silkworms, making significant findings which did not attract the publicity of Pasteur's studies, and again Béchamp felt he had reason to accuse Pasteur of plagiarizing his work.18 Béchamp's anger at what he considered the impropriety of Pasteur's behaviour and the error of his views of disease, which he described as 'the greatest scientific silliness of the age', consumed him for the rest of his long life.19
The microzymes
The microzymes are a form of life that Béchamp, over a period of 30 years, believed that he had discovered, beginning, as he points out in his book of this name,12 with his experiments carried out in the 1850s on the influence on moulds on the hydrolysis of sucrose. In the book he tries to bring together all the relevant data that brought him to the belief that the microzymes are at the basis of all life and death.
When examining solutions in which the hydrolysis of sucrose was taking place, Béchamp observed extremely small microscopic forms, similar to those seen in fermentations. In his paper of 1857 [1858],17 he designates them as little bodies and thus he came to regard the molecular granulations of the histologists as being organized and living ferments.
Béchamp came up with a startling finding in 1866.20 It was a well-established procedure to add chalk, mainly powdered limestone, to lactic and other acid fermentations to maintain neutrality. But Béchamp asks whether this is the only role of the chalk, which, as well as consisting of the fossil remains of crustaceans, he finds still contains a whole generation of extremely small organisms, smaller than the yeasts. Not only do they exist, but they are alive, despite their extreme geological age. They grow with a rare energy like ferments [yeasts]. They are the most active ferments Béchamp has encountered and they nourish themselves on very diverse organic substances.
To the organisms from lime (killed when heated to 300°C) Béchamp gives the name Microzyma cretae, but microzymes (meaning 'minute ferments') are found everywhere, including in soil. Béchamp's paper20 is logically presented, with sequential arguments, and the remarkable nature of his results is clear to him. Could it be that Béchamp was genuinely observing specific bacteria? Or was it a case of artefacts in the microscopes of the time?
Promotion of Béchamp against Pasteur
Béchamp's views would by now have been long since forgotten had they not been espoused by anti-vivisectionists and protagonists of alternative medicine. Pasteur 's use of experimental animals aroused the wrath of the former, and the desire of the latter to believe that disease originating from microorganisms is a fiction, set them firmly against Pasteur in favour of Béchamp's ideas.
Following the death of Béchamp in 1908, an event ignored in France, a Dr Montague Leverson from Baltimore persuaded a writer and sympathizer, Ethel Douglas Hume, to put together some notes he had collected as a book that was published first in 1923 under the title Béchamp or Pasteur? A Lost Chapter in the History of Biology and pours scorn on Pasteur 's efforts and accuses him of plagiarism and fabrication.16 The book was republished in 1932 and has gone through numerous subsequent editions and modifications of title (more recently being attributed to Douglas Hume!).c It is of course increasingly true that much human disease and suffering cannot be attributed to infection but stems from changes within us, but in this the microzymes do not have a place.
With a medical colleague, Alfred Estor, Béchamp observed granulations in cells (they mainly studied liver of different species).21 In the physiological state these granules, which they considered to be microzymes, are spherical, but outside the cell they develop into bead-like elongated structures, eventually recognizable as bacteria. From observations of bacteria in blood, they conclude that bacteria, far from being the cause of illness, are, to the contrary, the result.
Thus, in death the microzymes become bacteria, eventually reducing the cells of higher organisms to dust, and then reverting to microzymes. Airborne germs arise from microzymes in dead plant and animal life. Béchamp writes: 'The microzyme is at the beginning and at the end of every cell organization. It is the fundamental anatomical element by which the cellules, the tissues, the organism, the whole of an organism are constituted living.'12
Coda
It seems likely that, in the 1850s and 1860s, Béchamp and Pasteur were independently making similar discoveries—a not unknown phenomenon in science. Accusations of plagiarism are therefore probably not justified. Pasteur without question was aggressive and intolerant of opposition and treated Béchamp shabbily, but Béchamp led himself into a theory of such generality that was both its strength and its weakness—it could be used to explain too much, but did not lend itself to experimental testing. In a France increasingly idolizing Pasteur and his memory, Béchamp was bound to become increasingly ignored. Only Miss Hume16 and followers, for their own reasons, have kept his name alive.
1. Pasteur L. (1857). Mémoire sur la fermentation appelée lactique. Compt. Rend. 45, 913–916. [ Links ]
2. Pasteur L. (1856). Isomorphisme entre des corps isomères, les uns actifs, les autres inactifs sur la lumière polarisée. Compt. Rend. 43, 1259–1264. [ Links ]
3. Pasteur L. (1848). Mémoire sur la relation qui peut exister entre la forme cristalline et la composition chimique, et sur la cause de la polarisation rotatoire. Compt. Rend. 26, 535–538. [ Links ]
4. Pasteur L. (1857). Mémoire sur la fermentation alcoolique. Compt. Rend. 45, 1032–1036. [ Links ]
5. Pasteur L. (1858). Nouveaux faits concernant l'histoire de la fermentation alcoolique. Compt. Rend. 47, 1011–1013. [ Links ]
6. Buchner E. (1897). Alcoholische Gährung ohne Hefezellen. Ber. Dt. Chem. Ges. 30, 117–124. [ Links ]
7. Béchamp A. (1855). Note sur l'influence que l'eau pure et certaines dissolutions salines exercent sur le sucre de canne. Compt. Rend. 40, 436–438. [ Links ]
8. Béchamp A. (1858). De l'influence que l'eau pur ou chargée de diverse sels exerce à froid sur the sucre de canne. Compt. Rend. 46, 44–47. [ Links ]
9. Meeting report (1862). Revues des Sociétés des Savantes I, 81. [ Links ]
10. Béchamp A. (1864). Sur la fermentation alcoolique. Compt. Rend. 58, 601–605. [ Links ]
11. Béchamp A. (1872). Seconde observation sur quelques communications récentes de M. Pasteur, notamment sur la théorie de la fermentation alcoolique. Compt. Rend. 75, 1519–1523. [ Links ]
12. Béchamp A. (1883). Les Microzymas: L'hétérogénie, l'histogénie, la physiologie et la pathologie. Librairie J.-B. Baillière, Paris. [ Links ]
13. Report of International Medical Congress (1881). The Times (London), 8 August, p. 6. [ Links ]
14. Report of International Medical Congress (1881). Br. Med. J. 2, 547–548. [ Links ]
15. Pasteur L. (1876). Études sur la Bière. Gautier-Villars, Paris. [ Links ]
16. Hume E.D. (1923). Béchamp or Pasteur? A lost chapter in the history of biology. Covici-McGee, Chicago. [ Links ]
17. Béchamp A. (1858). De l'influence que l'eau pure, ou chargée de divers sels, exerce, à froide, sur le sucre de canne. Ann. Chim. 3rd ser., 54, 28–42. [ Links ]
18. Béchamp A. (1867). Lettre adressée à M. le Président, au sujet de la communication faite par M. Pasteur le 29 avril dernier. Compt. Rend. 64, 1042–1043. [ Links ]
19. Béchamp A. (1903). Louis Pasteur: ses plagiats chimicophysiologiques et medicaux. Chez l'auteur, Paris. [ Links ]
20. Béchamp A. (1866). Du rôle de la craie dans les fermentations butyrique et lactique, et des organismes actuellement vivants qu'elle contient. Compt. Rend. 63, 451–455. [ Links ]
21. Béchamp A. and Estor A. (1868). De l'origine et du développement des bactéries. Compt. Rend. 66, 859–863. [ Links ]
22. Findlay A. (1965). A Hundred Years of Chemistry, 3rd edn, p. 47. Wiley, New York. [ Links ]
a All naturally occurring sugars rotate the plane of polarized light to the right [dextrorotatory; a plus (+) rotation] or to the left [laevorotatory; a minus (–) rotation]. Polarimetry, a procedure dating from the early years of the 19th century, was once one of the easiest ways of following a reaction such as the hydrolysis of sucrose. Sucrose has a specific rotation of +67°, glucose of +53° and fructose –92°. An equimolar mixture of glucose and fructose is therefore laevorotatory, whereas that of sucrose is dextrorotatory. Invertase thus catalyses a reaction that results in the inversion of the plane of polarization from being to the right to being to the left. An old name for fructose is invert sugar or laevulose, and for glucose, dextrose (or grape sugar), as opposed to cane sugar for sucrose.
b Heterogenesis could mean either the birth or organization of a living being otherwise than from a parent of the same kind—a process called by Charlton Bastian archebiosis—or the generation of animal or vegetable life of low organization from inorganic (i.e. non-living) matter—called by Thomas Huxley abiogenesis.
c The most recent edition I have been able to track down is published by Health Research, in 2003 (ISBN-10 0787311286).There are also items on the Web.














