Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Journal of Science
versión On-line ISSN 1996-7489
versión impresa ISSN 0038-2353
S. Afr. j. sci. vol.103 no.9-10 Pretoria sep./oct. 2007
RESEARCH IN ACTION
Examination of Blepharis aspera as a possible Cu–Ni indicator plant
Bonang B.M. NkoaneI, II, *; Grethe WibetoeII; Walter LundII; Berhanu M. AbegazI; Nelson TortoI
IDepartment of Chemistry, University of Botswana, Private Bag UB 00704, Gaborone, Botswana
IIDepartment of Chemistry, University of Oslo, P.O. Box 1033, N-0315 Oslo, Norway
ABSTRACT
BLEPHARIS ASPERA WAS COLLECTED FROM A copper–nickel mineralized area in Botswana and examined as a possible Cu–Ni indicator plant for biogeochemical prospecting. Different plant parts and the host soils were analysed using ultrasonic slurry sampling electrothermal atomic absorption spectrometry. All plant parts accumulated Cu and Ni in above-normal amounts, although not in hyperaccumulator concentrations. The leaf to soil concentration ratio varied little with metal concentration in the soil. We propose Blepharis aspera as a Cu indicator plant. The accumulation behaviour of Blepharis aspera is compared with another previously studied metallophyte, Helichrysum candolleanum, that is growing in the same area.
Introduction
We present the results of a study of Blepharis aspera Obermeyer (family Acanthaceae ), whose occurrence has not been reported in Botswana before, as a possible Cu–Ni indicator plant. Recently,1 a similar Cu–Ni study was carried out on two metal-tolerant plants (metallophytes) —Blepharis diversispina (Nees) C.B. Clarke (Acanthaceae) and Helichrysum candolleanum H. Buek (Asteraceae)—in which the latter was reported to be a possible Cu–Ni indicator. Metallophytes cope differently with high levels of metals in the soil, and therefore it is desirable to establish the metal tolerance of these plants.
There are various types of metal-tolerant behaviour (displaying exclusion, accumulation and indicator) of plants that grow in mineralized soils.1–3
Experimental
Blepharis aspera is a shrub with purple-bluish flowers (Fig. 1). It was identified at the Royal Botanic Gardens, Kew, U.K. (voucher number 03SEL BD1). It grows in Zimbabwe and northern parts of South Africa.4,5 The plants and the host soils analysed were collected from Selkirk, an active copper–nickel mine, in northeastern Botswana, on three sampling trips carried out on 13/14 November 2002, 12/13 March 2003 and 20/21 August 2004. The samples were air-dried and ground to a fine powder (less than 63 µm).

Copper and nickel were determined using a Perkin-Elmer Zeeman atomic absorption spectrometer (AAnalyst 800, PE; Überlingen, Germany), set at 222.6 nm for Cu and 232.0 nm for Ni, and ultrasonic slurry sampling ETAAS with dilute nitric acid and Triton X-100 diluent. Certified reference materials were used for method validation. Details of the analytical procedure were described previously.1
Results and discussion
Of the four sampling locations visited in Botswana (Selkirk, Thakadu, Malaka and Nakalakwana), B. aspera was found only at Selkirk, which had high soil Cu and Ni concentrations in the range 0.38–1.9% Cu and 0.17–0.37% Ni. Thus, the plant can be classified as a metallophyte. To investigate if it could be used as a Cu–Ni indicator plant, it was important to know if B. aspera accumulated Cu and Ni in high amounts, to establish the type of metal tolerance (exclusion or accumulation), and also to determine the leaf to soil metal concentration ratios.
Figures 2A and B show the metal concentrations in the roots, stem, leaves and flowers, for Cu and Ni, respectively, arranged according to increasing soil metal content. The plant accumulated both metals mostly in the stem or roots, and least in the flowers. The metal concentrations recorded can be seen in Fig. 2. These values are higher than the corresponding concentrations typically found in plants.6–8
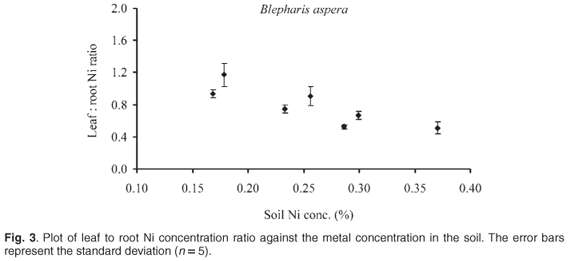
The leaf to root ratios of copper concentration (L/R ratios) were in all cases less than 0.6 and seemed to be independent of metal concentration in the soil, whereas the Ni L/R ratio decreased with increasing metal content in the soil range tested as shown in Fig. 3. Blepharis aspera therefore behaved as a copper excluder in all soils investigated (L/R < 1), and as a nickel excluder in relatively high-Ni soils (concentrations greater than about 2.5 mg g–1).2
The metal transfer coefficient (TC),6,9 which is defined as the ratio of the metal concentration in the plant to the total metal concentration in the soil, was used to estimate metal uptake and transfer to the shoot. In our study, the metal concentration in the leaf was used as an estimate of the plant concentration. The TCs for B. aspera were in the range 0.01–0.02 for Cu and 0.1–0.2 for Ni. The Ni TCs were higher than the corresponding copper values by an order of magnitude, indicating that for the same metal concentration in the soil, Ni had higher uptake and translocation. The Cu TCs varied little with soil metal concentrations in the range 0.38–1.9%, suggesting a metal uptake and translocation that was proportional to the soil metal concentration. As such, B. aspera can be proposed as a Cu indicator plant for biogeochemical prospecting.
Also, the Ni TCs did not vary much. However, as the Ni concentration range of the soil was smaller (0.17–0.37%) than for Cu, more data over a wider concentration range are needed to propose B. aspera as a Ni indicator plant.
Our work so far has revealed that the two metallophytes, B. aspera and H. candolleanum, which grow in the same mineralized area, accumulate Cu and Ni differently in their various plant parts, implying different modes of metal tolerance. Helichrysum candolleanum has a tendency to take up metals and transfer them to the higher parts (leaves and flowers), which indicates accumulator behaviour,1 whereas B. aspera tends to restrict most of the metal to the stem and roots, a feature of excluder behaviour. Although B. aspera may thus be classified as an excluder plant, it still transfers some metal to the leaves in proportion to the soil metal content, especially for Cu. This suggests that it may be used for biogeochemical prospecting, in which a constant plant: soil ratio is of particular interest3 because it reflects the degree of mineralization in the soil. Helichrysum candolleanum also had relatively constant Cu and Ni TCs over a wide range of soil metal concentrations, implying metal uptake and translocation proportional to the soil concentration, and hence could act as a Cu–Ni indicator plant.1 Of the two species, H. candolleanum may be a better Cu and Ni indicator for biogeochemical prospecting because of its ability to shift metals to its above-ground parts and thus can be more readily sampled in the field.7
We are grateful for financial assistance from the Norwegian Universities Committee for Development Research and Education, Norway, and for the contribution from the Department of Biology (Botany), University of Botswana. We are grateful to Kaj Vollesen, of The Royal Botanic Gardens, Kew, for the identification of B. aspera.
1. Nkoane B.B.M, Sawula G.M., Wibetoe G. and Lund W. (2005). Identification of Cu and Ni indicator plants from mineralised locations in Botswana. J. Geochem. Explor. 86, 130–142. [ Links ]
2. Baker A.J.M. (1981). Accumulators and excluders – strategies in the response of plants to heavy metals. J. Plant Nutr. 3, 643–654. [ Links ]
3. Baker A.J.M. and Brooks R.R. (1989). Terrestrial higher plants which hyperaccumulate metallic elements – a review of their distribution, ecology and phytochemistry. Biorecovery 1, 81–126. [ Links ]
4. Van Wyk B. and Malan S. (1988). Field Guide to the Wild Flowers of the Witwatersrand and Pretoria Region. Struik, Cape Town. [ Links ]
5. The Flora of Zimbabwe: Online: http://www. zimbabweflora.co.zw [ Links ]
6. Adriano D.C. (2001). Trace Elements in Terrestrial Environments: Biochemistry, Bioavailability and Risks of Metals, 2nd edn. Springer-Verlag, New York. [ Links ]
7. Kabata-Pendias A. and Pendias H. (1992). Trace Metals in Soils and Plants, 2nd edn. CRC Press, Boca Raton, Florida. [ Links ]
8. Loneragan J.F. and Robson A.D. (eds). (1981). Copper in Soils and Plants. Academic Press, New York. [ Links ]
9. Henning B.J., Snyman H.G. and Aveling T.A.S. (2001). Plant–soil interactions of sludge-borne heavy metals and the effect on maize (Zea mays L.) seedling growth. Water SA 27, 71–78. [ Links ]
* Author for correspondence. E-mail: bnkoane@bmr.co.bw and nkoanebbm@hotmail.com














