Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489
Print version ISSN 0038-2353
S. Afr. j. sci. vol.103 n.7-8 Pretoria Jul./Aug. 2007
RESEARCH IN ACTION
The diversity of culturable airborne fungi in an active South African gold mine
C.H. PohlI, *; W. KrielII; P. VenterIII; E. Van HeerdenI; J. AlbertynI
IDepartment of Microbial, Biochemical and Food Biotechnology, University of the Free State, P.O. Box 339, Bloemfontein 9300, South Africa
IIDepartment of Plant Sciences, University of the Free State. cUnit for Applied Food Safety & Technology, Department of Agriculture and Environmental Development, Central University of Technology, Bloemfontein, South Africa
ABSTRACT
Several studies have been conducted on the presence and diversity of airborne fungal spores in residential and occupational areas. This is the first investigation on this topic in the mining environment. The impact of fungi—especially as opportunistic pathogens—on their health and productivity needs to be assessed, considering the compromised health of a large number of HIV-infected miners in South Africa. Accordingly, the aim of this study was to determine the abundance of fungal spores as well as the diversity of culturable airborne fungi in an active gold mine. The results indicate that three of the sampled sites in the mine contain abundant numbers of culturable airborne fungi. The first site was next to an auxiliary extraction fan, which could act as an accumulator for airborne fungal spores from the rest of the mine. The two other sites had high human activity, disturbances from which may contribute to an elevated fungal load in the air. The presence of timber supports at one site may serve as a growth substrate for several fungal genera. The diversity of filamentous fungal genera increased from outside the mine to deeper into the mine, whereas the diversity of yeast genera followed an opposite trend, with no yeasts detected deep in the mine. Although most of the isolates identified are not pathogens, they could still have a negative impact (either as opportunistic pathogens or allergens) on workers, especially immunocompromised persons if exposed to them for prolonged periods.
Introduction
No assessment has yet been made of the impact of fungi on the health and performance of mine workers, although extensive research has been conducted on occupational hazards associated with the unique and often harsh conditions of mining activities. The ability of fungi to act as opportunistic pathogens in immunocompromised persons is of specific importance.1 It is known that fungal diseases are increasing among HIV-infected people, and almost any fungus is now considered to be able to cause disease in an immunocompromised person. Furthermore, fungi and their spores are known to be allergenic, causing occupational asthma, irritation of the respiratory system and dermatitis in otherwise healthy people.2–4 In addition, the respiration of fungal spores and the subsequent low-level exposure to mycotoxins and β-D-glucan could have adverse effects on workers' immune systems.5
This study aims to provide a better understanding of possible diversity of pathogens and/or allergens in the air circulating through mines because the mining environment provides a unique habitat for fungi.
Methods
See appendix.
Results and discussion
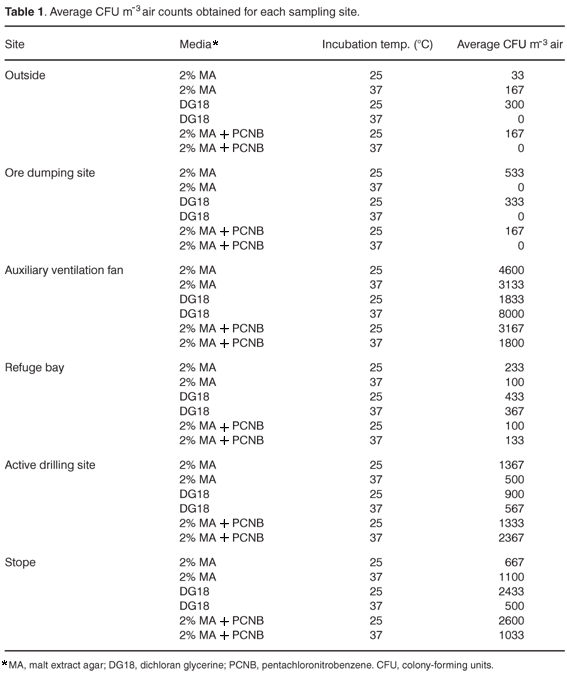
Table 1 (see supplementary material online at www.sajs.co.za) lists the average CFU/m3 air count obtained for each sampling site. The main observation that can be made from these data is that the site with the greatest CFU/m3 air value was next to the auxiliary ventilation fan. Since the literature records that there is a positive correlation between wind speed and spores/m3 in the air,12 it is expected that this site will also have the highest recorded wind speed (5.97 m s–1). During an extensive literature search, no health-related limits to permissible amounts of fungal spores in the air could be found, although several governments are formulating guidelines for building owners and occupational-health officers. Our counts do not appear to be excessively high compared with other reports of viable and non-viable spores/m3.12,13 However, Bush and Portnoy3 state that indoor CFU/m3 counts of 1000 or more are deemed as hazardous indoor fungal contamination. If this is taken into account, this site, as well as the active drilling site and the stope would be of concern, as counts of more than 1000/m3 were obtained for these three locations. We suggest that it is unlikely that fungi are actively growing next to the auxiliary ventilation fan, but that this site rather acts as an accumulator of fungal spores that are present in the rest of the mine. The origin of fungal contamination at the active drilling site needs further investigation. The timber supports in the stope can be a possible source of fungal growth and contamination at this site. Because these samples were taken in winter, it would be interesting to compare these counts with results for summer, when higher spore counts would be expected.12
The diversity of filamentous fungi and yeasts obtained from these samples is shown in Table 2 (see supplementary material online). The majority of the isolated filamentous fungi are known to occur either with cellulose-containing material, such as wood, or other living or dead plant material or as soil fungi. These substrates are abundant in and around the mine and may serve as sources of fungi. The isolated yeasts were found to be mainly those associated with soil and humans, elements of the mining environment.
Although no specific trend is evident regarding the total number of genera (filamentous fungi and yeasts) from the different sites, it is interesting to note that the number of recorded filamentous fungal genera, increased from outside the mine (6 genera) and at the beginning of the level of the ore dumping site (5 genera) to deeper in the mine (7 genera at the ventilation fan and active drilling site; 9 genera at the stope). In the light of this observation, the higher diversity in the refuge bay (9 genera) is against expectation, as it has a direct air supply from the surface. This may indicate the presence of growing fungi in biofilms that line the ventilation ducts of the refuge bay. The findings regarding the number of yeast genera show a contrasting trend. There are more yeast genera obtained from the samples taken outside, and no yeast isolates obtained from the samples taken in the stope.
Although very few human pathogens were identified in this study, we identified several opportunistic pathogens. This could constitute a health hazard, especially for those mine workers who are HIV positive or who have conditions such as tuberculosis.
Conclusions
We conclude that certain mining sites, especially those with high wind speed and human activity, contain health-risk related numbers and diversity of culturable fungal spores in the air. Although most of the fungal isolates identified were not human pathogens, inhalation of these fungal spores could have a negative effect on the health and productivity of mine workers—especially those with HIV or other pre-existing conditions, such as tuberculosis.
This is the first study of this nature in the mining environment, and several interesting questions still await investigation. Some of these include the seasonal variation in fungal spore diversity and numbers, together with a study to determine the correlation between the occurrence of fungi and reports of fungal related disease.
This study was funded by the Safety in Mines Research Advisory Committee (Grant no. SIM 03-06-02). We thank O. Moloko, S. Fuku and K. Pudumo for their assistance.
1. Warnock D.W. and Richardson M.D. (1982). Preface. In Fungal Infection in the Compromised Patient, eds D.W. Warnock and M.D. Richardson, pp. xi. John Wiley, Chichester. [ Links ]
2. Verhoeff A.P. and Burge A.H. (1997). Health risk assessment fungi in home environments. Ann. Allergy Asthma Immunol. 78, 544–556. [ Links ]
3. Bush K.R. and Portnoy M.J. (2001). The role and abatement of fungal allergens in allergic diseases. J. Allergy Clin. Immunol. 107, 430–440. [ Links ]
4. Davis J.P. (2001). Moulds, toxic moulds, and indoor air quality. California Res. Bureau. 8(1), 1–18. [ Links ]
5. McNeel S. and Kreutzer R. (1996). Fungi and indoor air quality. Health Environ. Digest 10(2), 9–12. [ Links ]
6. Hoekstra E.S., Samson R.A. and Summerbell R.C. (2004). Methods for the identification and isolation of fungi in the indoor environments. In Introduction to Food-and Airborne Fungi, 7th edn. Centraalbureau voor Schimmelcultures, Utrecht. [ Links ]
7. Klich M.A. (2002). Identification of Common Aspergillus Species. Centraalbureau voor Schimmelcultures, Utrecht. [ Links ]
8. Samson R.A., Hoekstra E.S. and Frisvad J.C. (2004). Introduction to Food-and Airborne Fungi, 7th edn. Centraalbureau voor Schimmelcultures, Utrecht. [ Links ]
9. Abarca M.L., Accensi F., Cano J. and Cabañes F.J. (2004). Taxonomy and significance of black aspergilli. Ant. Leeuwenhoek 86, 33–49. [ Links ]
10. Nelson P.E., Toussoun T.A. and Marasas W.F.O. (1983). Fusarium species: an illustrated manual for identification, p. 193. Pennsylvania State University Press, University Park. [ Links ]
11. Kurtzman C.P. and Robnett C.J. (1998). Identification and phylogeny of ascomycetous yeasts from analysis of nuclear large subunit (26S) ribosomal DNA partial sequences. Ant. Leeuwenhoek 73, 331–371. [ Links ]
12. Sakiyan N. and Inceoglu O. (2003). Atmospheric concentrations of Cladosporium Link and Alternaria Nees spores in Ankara and the effects of meteorological factors. Turk. J. Bot. 27, 77–81. [ Links ]
13. Krysinska-Traczyk E. and Dutkiewicz J. (2000). Aspergillus candidus: a respiratory hazard associated with grain dust. Ann. Agric. Environ. Med. 7(2), 101–109. [ Links ]
This article is accompanied by supplementary material online at www.sajs.co.za
* Author for correspondence. E-mail: pohlch.sci@mail.uovs.ac.za
Sampling sites
The samples were taken during winter in an operational gold mine in the Free State province of South Africa (Clark 1880 coordinates: Y20955.883; X3128473,179; Z1394.456). The shaft chosen is one of the main ventilation shafts for the mine and is divided into two sections, one that allows fresh air to enter the mine and another by which air is returned to the surface by means of extraction fans. We took samples at six different sites in order to obtain information on the distribution of culturable fungal spores throughout the shaft.
Because fungal spores circulating in the air in the mine could be entering from outside the mine, the first site of interest was at the intake of the down-cast shaft, which serves as one of the main sources of air for this part of the mine. The second site was at the beginning of the level (at a depth of 668.5 m) where the main activity was ore-dumping. The third site was next to an auxiliary ventilation fan that returns air to the surface. The fourth site was inside a refuge bay. Of note is that these bays have a separate air supply directly from the surface. The fifth location was an active drilling site, with high human activity. The sixth site was a stope, with high human activity as well as the presence of timber props.
Sampling techniques
An SAS Super 90 single-stage impactor was used, with three different media, in order to detect the greatest diversity of fungal species.6 These were dichloran glycerine selective agar (DG18), containing 5.0 g l–1 peptone, 10.0 g l–1 dextrose, 1.0 g l–1 monopotassium phosphate, 0.5gl–1 magnesium sulphate, 0.002 g l–1 dichloran, 0.1g l–1 chloramphenicol, 18.0 g l–1 glycerol and 15.0 g l–1 agar; 2% malt extract agar (2% MA), containing 20.0 g l–1 malt extract and 15.0 g l–1 agar as well as 2% malt extract agar supplemented with pentachloronitrobenzene (PCNB) to restrict the growth of zygomycetes. The sampling time was 30 seconds at 30 l min–1. In addition, wind speed was recorded for each site.
Cultivation
Different incubation temperatures (25º and 37ºC) were used as the objective was to determine the diversity of culturable fungi, including possible pathogens. Randomly selected representative colonies were further purified on 2% MA at the two temperatures. Standard safety procedures were adopted when handling potential pathogens.
Enumeration and identification
The number of colony-forming units (CFUs) counted on the surface of the plates was recorded to determine whether significant numbers of culturable spores are present in the air at the different sites. Data were corrected for the statistical possibility of multiple spores passing through the same hole in the sampler. The corrected or probable count was used to calculate the average colony-forming units (CFU) m–3 air sampled. Filamentous isolates were identified to genus, and where possible, species level, using standard taxonomic keys based on colony characteristics and morphology.7–10 For identification of yeast isolates the D1/D2 region of the 26S rDNA was PCR amplified using primer pair NL1 (5'-GCATATCAATAAGCGGAG GAAAAG) and NL4 (5'-GGTCCGTGTTTC AAGACGG).11 Sequencing reactions were performed with the ABI PrismTM Big Dye terminatorTM V3.1 cycle sequencing ready reaction kit, and data collected on an ABI Prism 377 DNA sequencer (Applied Biosystems). Sequences were reverse complemented and compared by using Sequence Navigator V 1.0.1 and assembled using Auto-assembler V1.4.0.
Supplementary material to:
Pohl C.H., Kriel W., Venter P., Van Heerden E. and Albertyn J. (2007). The diversity of culturable airborne fungi in an active South African gold mine. S. Afr. J. Sci. 103, 277–278.