Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
Onderstepoort Journal of Veterinary Research
versión On-line ISSN 2219-0635
versión impresa ISSN 0030-2465
Onderstepoort j. vet. res. vol.81 no.1 Pretoria ene. 2014
ORIGINAL RESEARCH
Distribution patterns and predilection muscles of Trichinella zimbabwensis larvae in experimentally infected Nile crocodiles (Crocodylus niloticus Laurenti)
Louis J. La GrangeI, II; Samson MukaratirwaII
IDepartment of Agriculture, Rural Development and Land Administration, South Africa
IISchool of Life Sciences, University of KwaZulu-Natal, South Africa
ABSTRACT
No controlled studies have been conducted to determine the predilection muscles of Trichinella zimbabwensis larvae in Nile crocodiles (Crocodylus niloticus) or the influence of infection intensity on the distribution of the larvae in crocodiles. The distribution of larvae in muscles of naturally infected Nile crocodiles and experimentally infected caimans (Caiman crocodilus) and varans (Varanus exanthematicus) have been reported in literature. To determine the distribution patterns of T. zimbabwensis larvae and predilection muscles, 15 crocodiles were randomly divided into three cohorts of five animals each, representing high infection (642 larvae/kg of bodyweight average), medium infection (414 larvae/kg of bodyweight average) and low infection (134 larvae/kg of bodyweight average) cohorts. In the high infection cohort, high percentages of larvae were observed in the triceps muscles (26%) and hind limb muscles (13%).
In the medium infection cohort, high percentages of larvae were found in the triceps muscles (50%), sternomastoid (18%) and hind limb muscles (13%). In the low infection cohort, larvae were mainly found in the intercostal muscles (36%), longissimus complex (27%), forelimb muscles (20%) and hind limb muscles (10%). Predilection muscles in the high and medium infection cohorts were similar to those reported in naturally infected crocodiles despite changes in infection intensity. The high infection cohort had significantly higher numbers of larvae in the sternomastoid, triceps, intercostal, longissimus complex, external tibial flexor, longissimus caudalis and caudal femoral muscles (p < 0.05) compared with the medium infection cohort.
In comparison with the low infection cohort, the high infection cohort harboured significantly higher numbers of larvae in all muscles (p < 0.05) except for the tongue. The high infection cohort harboured significantly higher numbers of larvae (p < 0.05) in the sternomastoid, triceps, intercostal, longissimus complex, external tibial flexor, longissimus caudalis and caudal femoral muscles compared with naturally infected crocodiles. Results from this study show that, in Nile crocodiles, larvae of T. zimbabwensis appear first to invade predilection muscles closest to their release site in the small intestine before occupying those muscles situated further away. The recommendation for the use of masseter, pterygoid and intercostal muscles as sampling sites for the detection of T. zimbabwensis in crocodiles is in contrast to the results from this study, where the fore- and hind limb muscles had the highest number of larvae. This study also supports the use of biopsy sampling from the dorso-lateral regions of the tail for surveillance purposes in both wild and commercial crocodile populations.
Introduction
Knowing the predilection muscles of Trichinella spp. larvae in different hosts is important in order to improve the detection of the parasite in animal hosts, especially those with low levels of infection (Kapel, Webster & Gamble 2005). Artificial digestion is the only diagnostic method currently approved for the detection of Trichinella spp. in sylvatic animals (European Commission 2005). Gottstein, Pozio and Nöckler (2009) emphasised the importance of sensitivity of the test as animal hosts do not develop observable clinical symptoms and factors such as sample size and sampling site influence the test sensitivity.
Several studies have been conducted to determine predilection sites of different Trichinella spp. in various hosts (Kapel et al. 1994, 1995, 2005; Kociecka, Van Knapen & Ruitenberg 1980; Reina, Muñoz-Ojeda & Serrano 1996; Serrano & Pérez-Martín 1999). These studies have led to recommendations on the type of muscle to sample, the quantity and the appropriate method(s) for detection in several animal hosts (Gottstein et al. 2009). Studies have shown that the predilection sites of Trichinella spp. are influenced by several factors, including differences between species (Kapel et al. 2005; Wright et al. 1989), initial levels of infection (Serrano & Pérez-Martín 1999) and host characteristics (Hurnikova et al. 2004; Kapel et al. 1995; Kapel et al. 2005; Reina et al. 1996; Soule et al. 1989). The distribution of larvae in several muscles of naturally infected crocodiles, experimentally infected caimans (Caiman crocodilus) and varans (Varanus exanthematicus) has been reported previously (La Grange, Govender & Mukaratirwa 2013; Pozio et al. 2004). No controlled studies have been conducted to determine predilection muscles in Nile crocodiles and the influence of infection intensity on the distribution of Trichinella zimbabwensis larvae in crocodiles.
Current testing protocols for the detection of Trichinella larvae in muscles for export of crocodile meat in South Africa require that samples be collected from the anterior limbs of the slaughtered animals, in contrast to the European Commission Regulation 2075/2005 (European Commission 2005), which recommends sampling from the masseter, pterygoid or intercostal muscles in crocodiles.
The objective of this study was to determine the distribution patterns and predilection sites of T. zimbabwensis larvae in experimentally infected Nile crocodiles and the influence of intensity of infection.
Materials and methods
Study animals
Fifteen 7-year-old Nile crocodiles (13 males and two females) within the size range of 1.35 m - 1.80 m in length were used in the study. The crocodiles consisted of a group of animals not intended for commercial purposes and were sourced from a commercially farmed population. The study animals were housed on a smallholding belonging to the Mpumalanga Tourism and Parks Agency (MTPA) on the outskirts of Mbombela (Nelspruit), Mpumalanga province, South Africa. A fenced enclosure (10 m x 5 m) was constructed using 65 mm diamond mesh fencing with a single access gate. Gallamine triethiodide (Kyron) (40 mg/mL) was used to immobilise the animals at a dose of 0.4 mL/animal injected intramuscularly on the lateral aspect of the tail base before being transported to the experimental housing.
The experiment was carried out from January 2012 to March 2012, when climatic conditions ensured good feeding and optimal physical condition of the animals. Each animal was identified by clipping the horizontal scutes on the tail. The animals were randomly divided into three equal cohorts of five animals each to form high infection, medium infection and low infection cohorts. For the high infection cohort, both the left and right scutes were clipped in sequence according to the number assigned to the animal; for the low and medium infection cohorts only the scutes on the left and right were clipped respectively. Animal husbandry and feeding practices for the study animals were followed as described by the South African National Standard for crocodiles in captivity SANS 631:2009 (SABS Standards Division 2009). Pre-experimental data collected from the animals included weight, sex and length of the animals.
The enclosure allowed for a temperature gradient in the water to provide for optimal thermoregulation. The pond was drained weekly and replenished with fresh water during capture and sampling.
Source and preparation of infective material
Trichinella zimbabwensis-infected meat was sourced from a crocodile experimentally infected with T. zimbabwensis. Muscle tissue was collected from various sites, minced and thoroughly mixed by hand (in protective clothing) using a ladle. An amount of 100 g of the homogenised sample was subjected to artificial digestion (Nöckler & Kapel 2007) and larvae counted under a dissecting microscope (40 x magnification) using a gridded petri dish (European Commission 2005). Infection level was determined to be 30 larvae per gram (lpg) of the homogenised sample. Infective material for each individual animal was calculated and separately packaged and refrigerated at 4 °C until the day of infection. All of the crocodiles were infected with the same batch of infective material.
Infection of study animals
The high infection crocodile cohort were infected with 10 000 larvae (average 642 larvae/kg of bodyweight or 333 g of infective material), the medium infection cohort with 5000 larvae (average 414 larvae/kg of bodyweight or 167 g of infective material) and the low infection cohort with 2000 larvae (average 134 larvae/kg of bodyweight or 67 g of infective material) following infective dose in reptiles as reported by Pozio et al. (2004).
The animals were monitored continuously for side-effects or regurgitation of infective material for at least 30 min, following infection.
Biopsy sampling
Muscle biopsies were collected from the dorso-lateral aspects of the tail base on day 28 post-infection (p.i.) in all cohorts. Prior to biopsy, a muscle relaxant, gallamine triethiodide (40 mg/mL) (Kyron) was administered intramuscularly at a dose of 0.4 mL to each animal. A local anaesthetic, lignocaine (Kyron) was administered around the biopsy site. A minimum of 10 g of muscle tissue was removed with a scalpel and forceps, taking care not to collect tissue from the deeper musculature. The samples were refrigerated at 4 °C until testing. The biopsy wound was closed by pressing the flap of skin back into its original position over the wound and held in place with sutures. A dose of 0.1 mIU/kg penicillin was administered intramuscularly (Huchzermeyer 2003) to prevent wound infections.
Euthanasia of infected animals and postmortem sampling
Animals from each cohort were euthanised on day 60 p.i. following procedures outlined by Beaver et al. (2001). Carcasses were skinned and approximately 50 g of muscle samples were collected from the base of the tongue, external pterygoid, sternomastoid, triceps brachii, longissimus complex, intercostal pillars, longissimus caudalis and caudal femoral muscles or muscle group of each animal using diagrams of the musculature as presented by Richardson, Webb and Manolis (2002). In addition to the above, 10 g samples were collected from the superficial, lateral aspects of the longissimus complex and ilioischial muscles of the tail to mimic biopsy samples in live animals. All samples were placed in leak-proof containers and refrigerated at 4 °C until tested for the presence of T. zimbabwensis larvae.
Artificial digestion
Pooled samples representing individual animals were prepared by collecting 10 g of muscle tissue from all of the individual muscles (tongue, pterygoid, sternomastoid, triceps, intercostal, longissimus complex, external tibial flexor, longissimus caudalis and caudal femoral) and combining them in a single sample of 90 g. For individual muscle digestion, 25 g of tissue was used from each muscle and 10 g of muscle tissue were used to mimic biopsies. Samples were digested artificially according to Nöckler and Kapel (2007).
Data analysis
Data obtained from naturally infected crocodiles (La Grange et al. 2013) were included in the analyses and compared with data from this study. Data were normalised [log10(x+1)] and one of the naturally infected animals that had an unusually high level of infection compared with other naturally infected animals reported by La Grange et al. (2013) was removed from the analysis.
Muscles were grouped together to represent the cranial, abdominal and caudal muscle regions of crocodiles. The cranial group of muscles included the tongue, pterygoid and sternomastoid. The abdominal muscle group consisted of the triceps, intercostals, longissimus complex and tibial flexor muscles. The caudal muscle group was represented by the longissimus caudalis and caudal femoral muscles, including the biopsy samples. The mean infection intensity of the muscles in each of these three regions was calculated for each infection cohort and expressed as a percentage of the combined mean of all three regions.
The mean larval burdens were compared using analysis of variance (IBM SPSS Statistics 19) to determine the differences within the muscles of each cohort. A t-test was used to compare larval burdens between the superficial and deep musculature of the tail and between the dorsal and ventral biopsy samples. Significance level was set at p < 0.05.
Results
The mean lpg in pooled samples for the high, medium and low infection cohorts was 5.18, 0.37 and 0.02 respectively (see Table 1). The lpg in pooled samples was comparable with that from naturally infected crocodiles, although lower than that of the high infection cohort but higher than that of the medium and low infection cohorts of this study. A high mean lpg was also observed in all of the individual muscles of the high infection cohort when compared with natural infections. The mean lpg was also greater in the sternomastoid, triceps, longissimus complex and external tibial flexor muscles of the medium infection cohort when compared with natural infections.
In the low infection cohort, muscle larvae established in very low numbers (< 0.04 lpg) in all five of the individuals and in one animal only a single larva was detected in the pooled sample. The highest percentage of larvae in the four remaining animals was found in the intercostal (36%), longissimus complex (27%), forelimb (20%) and hind limb (10%) regions with the highest percentage of larvae occupying the intercostal and longissimus complex muscles. In the medium infection cohort all of the muscle regions tested were infected but the higher percentage of larvae was found in the triceps muscles (50%), sternomastoid (18%) and hind limbs (13%). A more even distribution of larvae was noted in the high infection cohort, with triceps muscles (26%) and hind limbs (13%) harbouring the most larvae. In naturally infected crocodiles the triceps muscles harboured 17% of all larvae with most of the remaining larvae spread more evenly amongst the pterygoid (12%), superficial longissimus caudalis (12%), external tibial flexor (10%), intercostal (10%), ilioischial (10%) and deep longissimus caudalis (9%). The high infection cohort had significantly higher numbers of larvae in the sternomastoid (p = 0.007), triceps (p = 0.006), intercostal (p < 0.001), longissimus complex (p = 0.002), external tibial flexor (p = 0.001), longissimus caudalis (p < 0.001) and caudal femoral muscles (p < 0.001) when compared with the medium infection cohort. The high infection cohort harboured significantly higher numbers of larvae in the pterygoid (p = 0.023), sternomastoid (p < 0.001), triceps (p < 0.001), intercostal (p < 0.001), longissimus complex (p = 0.001), external tibial flexor (p < 0.001), longissimus caudalis (p < 0.001) and caudal femoral muscles (p < 0.001) when compared with the low infection cohort. The high infection cohort also harboured significantly higher numbers of larvae in the sternomastoid (p = 0.001), triceps (p < 0.001), intercostal (p < 0.001), longissimus complex (p = 0.001), external tibial flexor (p < 0.001), longissimus caudalis (p = 0.001) and caudal femoral muscles (p < 0.001) compared with the naturally infected crocodiles.
The abdominal muscle groups harboured the highest number of larvae in all of the cohorts and revealed less variation in the distribution of larvae between the caudal and cranial muscle groups (see Table 2). Significantly higher numbers of larvae were found in the cranial muscle group of crocodiles in the high infection cohort compared with medium infection (p = 0.001), low infection (p < 0.001) and naturally infected (p < 0.001) crocodiles. The high infection cohort also showed significantly higher numbers of larvae in the abdominal muscle groups compared with medium infection (p < 0.001), low infection (p < 0.001) and naturally infected (p < 0.001) crocodiles and in the caudal muscle groups compared with medium infection (p < 0.001), low infection (p < 0.001) and naturally infected (p < 0.001) crocodiles. No significant differences in larval burdens were observed in any of the muscle groups between any of the other cohorts. In the high and medium infection cohorts, the abdominal regions harboured 56% and 59% of larvae respectively, followed by the cranial (21% and 28% respectively) and caudal (23% and 13% respectively) muscle groups. In the low infection cohort, the abdominal muscle group harboured 82% of all larvae, with the remaining larvae almost equally spread between the caudal (10%) and cranial (8%) muscle groups. The naturally infected crocodiles showed very little difference in distribution of larvae between the three muscle groups, as 38% of larvae were found in the abdominal, 33% in the caudal and 29% in the cranial muscle groups.
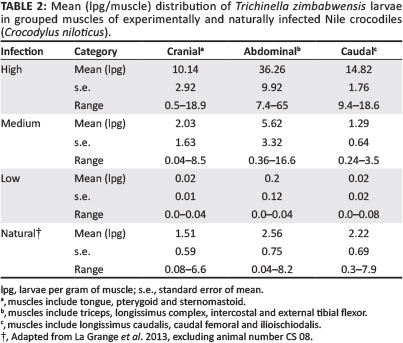
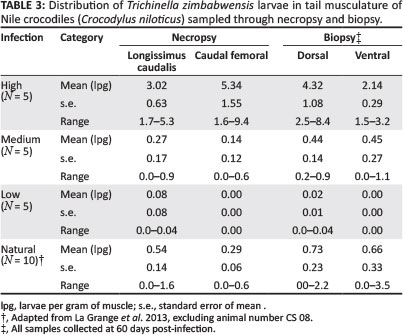
Artificial digestion and examination of the biopsy samples collected on day 28 p.i. showed no larvae in any of the three cohorts. However, biopsy samples collected on day 60 p.i. were positive for T. zimbabwensis larvae (see Table 3). Differences observed in larval distribution between the superficial and deep musculature of the tail were not significant, although the dorsal biopsy samples in all the cohorts harboured slightly more larvae (1.25 lpg) on average than the deeper musculature (0.88 lpg). Conversely, the biopsy samples collected from the ventral muscles harboured a smaller average number of larvae (0.78 lpg) than the deeper musculature (1.21 lpg). Mean larval burdens in tail musculature for individual cohorts are shown in Table 3.
Ethical considerations
Ethical approval for the study was obtained through the Animal Ethics Research Committee of the University of KwaZulu-Natal (reference number 035/12/Animal). Where applicable, ARRIVE guidelines for reporting in vivo animal experiments (Kilkenny et al. 2010) have been adhered to.
Discussion
In this study, crocodiles that received a lower number of larvae relative to their bodyweight showed higher infection intensity after 60 days p.i. in the high infection cohort. Similar results were reported by Hurnikova et al. (2004) in a study involving Red foxes (Vulpes vulpes) experimentally infected with T. zimbabwensis but the observed difference, in contrast to results from this study, was not significant. The high numbers of larvae observed in the triceps, intercostal and external tibial flexor muscles of the crocodiles do not support the findings observed in experimentally infected caimans (C. crocodilus) and varans (V. exanthematicus) where the tongue harboured the highest number of larvae (Pozio et al. 2004). However, the importance of the muscles of the fore- and hind limbs in sylvatic carnivores as reported by Kapel et al. (1994) and Kapel et al. (1995) is in agreement with findings from this study. It appears that predilection sites of T. zimbabwensis in Nile crocodiles are not influenced by the locomotive potential of muscles as seen in foxes (Kapel et al. 1994; Kapel et al. 1995) since in crocodiles the limbs are rarely associated with the high frequency and intensive locomotive behaviour seen in land-based animals. Crocodiles generally travel only short distances on land at slow speed and mainly use their large tail muscles to swim and propel themselves when acquiring prey (Richardson et al. 2002). Thus, in crocodiles the notion that the most active muscles are the most parasitised seems not to apply (Reina et al. 1996). However, the locomotive behaviour of crocodiles when submerged at the bottom of rivers and lakes has not been studied intensively and their leg musculature may play a more significant role under such circumstances than is currently known. Additionally, the leg musculature may be more oxygenated compared with other muscle groups, which could also explain the parasite's tendency for predilection for these muscles (Serrano et al. 1999).
The results from this study showed the triceps muscle to harbour the most larvae in natural (17%), medium (50%) and high infection (26%) groups. In the low infection cohort, the intercostal muscles harboured the most larvae (40%). Deviations in predilection patterns between different levels of infection were also noted in similar studies in mammals (Gamble, Gajadhar & Solomon 1996; Serrano & Pérez-Martín 1999). The results show that larvae primarily establish in those muscles in close proximity to the abdominal muscle region and disperse in relatively equal numbers to the cranial and caudal muscle regions, further away from their initial release site in the small intestine, as infection levels are increased. The dispersion to and subsequent increase of larval numbers in the cranial and caudal muscle regions appear to be correlated with a simultaneous and proportional decrease of larvae in the abdominal muscle region, eventually leading to an approximately equal distribution in all three muscle regions. This phenomenon cannot be explained and requires further investigation. Individually, however, the muscles favoured by the larvae as predilection sites retain their proportionally higher numbers of larvae. This is consistent with the hypothesis that larvae of Trichinella will primarily seek out predilection muscles in cases of low infection and will invade alternative muscle groups that are available as the infection intensity is increased (Wright et al. 1989). However, this does not explain the relatively more uniform regional distribution of larvae observed in naturally infected crocodiles compared with crocodiles of the high infection cohort. This phenomenon may be the result of secondary and subsequent recurring infections of crocodiles in the wild. Importantly, the groups of crocodiles with naturally acquired infections comprised individuals with large variations in size that were for the most part much larger and therefore much older than those used in the experimental cohorts. The naturally infected crocodiles additionally were derived from various habitats in the Kruger National Park. It is known that host factors such as hormone levels and the age of the animal as well as environmental factors such as temperature and season may also impact on the immune response of crocodiles (Brown et al. 2001; Ludovisi et al. 2013). The potential for variation in individual immunological status of naturally infected crocodiles could therefore also influence the distribution patterns of larvae in these animals.
In experimental studies involving T. spiralis and T. pseudospiralis in monkeys, larvae were detected in biopsy samples from day twelve and day 24 respectively (Kociecka et al. 1980). In goats infected with T. spiralis, muscle larvae were detected from day 20 p.i. (Reina et al. 1996) and from day 21 in heavily infected horses (Soule et al. 1989). In this study biopsy, samples from all three experimental cohorts collected on day 28 p.i. tested negative for T. zimbabwensis larvae but larvae were detected in biopsy samples collected on day 60 p.i. These differences between crocodiles and mammals cannot be accurately explained at this stage; however, studies in caimans and varans experimentally infected with T. zimbabwensis and T. papuae showed that muscle tissue damage occurred more rapidly in mammals than in reptiles and suggested that the host physiology could play a role in this (Pozio et al. 2004). Results from this study appear to support this hypothesis, especially considering that the metabolic rate of a crocodile is far lower than that of a human of similar mass (Huchzermeyer 2003). The study by Pozio et al. (2004) showed that larvae of T. zimbabwensis had a longer growth period and subsequently developed to a larger size in reptilian hosts than in mammals. This longer growth period was hypothesised to cause a delay in larvae establishing in muscle tissue (Pozio et al. 2004).
In mammals, the establishment of larvae can also be influenced by infection intensity; larvae tend to establish earlier in high infections than in low infections (Soule et al. 1989). Kociecka et al. (1980) also demonstrated that the rate of larval establishment varied between different species of Trichinella.
Conclusion
Trichinella zimbabwensis larvae successfully established in the muscles of all the experimentally infected animals in this study. Results from the high infection cohort are in agreement with findings in experimentally infected Red foxes (V. vulpes) (Hurnikova et al. 2004), where larger infection doses correspond with a lower infection intensity. It is not clear whether the decrease in parasite numbers is purely a host strategy aimed at survival or whether the parasite itself is in some way responsible for the decrease of its own numbers to ensure preservation of the host. Despite habitat and physiological differences between crocodiles and land-based mammals, results from this study support the importance of leg musculature as a predilection site for Trichinella spp. in sylvatic carnivores (Kapel et al. 1995). Although crocodiles appear to use their limbs less frequently and intensively compared with land-based mammals, more research is required to elucidate the use of their limbs when submerged at the bottom of rivers and lakes.
Furthermore, results from this study show that, in Nile crocodiles, larvae of T. zimbabwensis appear first to invade predilection muscles closest to their release site in the small intestine before occupying those muscles situated further away. This is in agreement with the hypothesis of Wright et al. (1989) that the larvae of Trichinella spp. would establish first in predilection muscles before occupying other available muscles. Studies involving additional experimental animals should be conducted to further investigate this hypothesis and to establish the influence of challenge infections on the distribution patterns of these larvae.
In Nile crocodiles, based on this study, the tongue does not appear to be a predilection site for T. zimbabwensis larvae as is the case in varans and caimans (Pozio et al. 2004). The difference in predilection muscles observed between caimans (Pozio et al. 2004) and Nile crocodiles in this study further supports the importance of host characteristics as a determinant for predilection (Hurnikova et al. 2004; Kapel et al. 1995; Kapel et al. 2005; Reina et al. 1996; Soule et al. 1989). Additional factors that may impact the immunological response of crocodiles, such as temperature, season, age of the animal and hormone levels (Brown et al. 2001; Ludovisi et al. 2013), may also have led to variations in larval distribution patterns as observed between caimans (Pozio et al. 2004) and crocodiles in this study.
The recommendation for the use of masseter, pterygoid and intercostal muscles as sampling sites for the detection of T. zimbabwensis in crocodiles (European Commission 2005) is contrary to the results from this study, where the fore- and hind limb muscles had the highest number of larvae. In this study, biopsy samples from all three experimental cohorts collected on day 28 p.i. tested negative for T. zimbabwensis larvae, which were detected subsequently in biopsy samples collected on day 60 p.i.
These results support the hypothesis that larvae of T. zimbabwensis might undergo a longer period of growth in reptilian hosts, resulting in delayed establishment in the muscles (Pozio et al. 2004). Results from this study support the use of biopsy sampling from the dorso-lateral regions of the tail for surveillance purposes in both wild and commercial crocodile populations (La Grange et al. 2013).
Acknowledgements
Our sincere gratitude to the following organisations and people, without whom this work would not have been possible: Mrs T. La Grange for her assistance with the many late hours of data capturing and laboratory work; Dr J.P. Raath of Wildlife Pharmaceuticals and Wildlifevets.com for the generous financial, technical and logistical support provided; Mpumalanga Tourism and Parks Agency for providing the housing facilities for experimental animals used in this study; Dr D. Brugman for his assistance during the post mortem examinations; the Crocodile Specialist Group for providing financial assistance in the form of a grant through their Student Research Assistance Scheme; Seronera and Wilderness Roads crocodile farms for providing experimental animals and food; and Dr D. Pfukenyi, Prof. M. Hosie and Ms L. Laubscher for their assistance with the final data analysis.
Competing interests
The authors declare that they have no financial or personal relationship(s) which may have inappropriately influenced them in writing this article.
Authors' contributions
L.L.G (Department of Agriculture and the University of KwaZulu-Natal) was the project leader, responsible for experimental and project design and performing all of the experiments and data analysis. S.M. (University of KwaZulu-Natal) supervised the project, assisted with experimental and project design, made conceptual contributions and assisted with data interpretation.
References
Beaver, B.V., Reed, W., Leary, S., McKiernan, B., Fairfield, B., Schultz, R. et al., 2001, '2000 Report of the American Veterinary Medical Association Panel on Euthanasia', Journal of the American Veterinary Medical Association 218, 682-687. http://dx.doi.org/10.1128/JCM.39.1.285-292.2001 [ Links ]
Brown, D., Schumacher, I., Nogueira, M., Richey, L.J., Zacher, L.A., Schoeb, T.R. et al., 2001, 'Detection of antibodies to a pathogenic Mycoplasma in American alligators (Alligator mississippiensis), broad-nosed caimans (Caiman latirostris), and Siamese crocodiles (Crocodylus siamensis)', Journal of Clinical Microbiology 39, 285-292. [ Links ]
European Commission, 2005, 'Commission Regulation (EC) No. 2075/2005 of 5 December 2005 laying down specific rules on official controls for Trichinella in meat', Official Journal of the European Union L338, 60-82. [ Links ]
Gamble, H.R., Gajadhar, A.A. & Solomon, M.B., 1996, 'Methods for the detection of trichinellosis in horses', Journal of Food Protection 59, 420-425. [ Links ]
Gottstein, B., Pozio, E. & Nöckler, K., 2009, 'Epidemiology, diagnosis, treatment, and control of trichinellosis', Clinical Microbiology Reviews 22, 127-145. http://dx.doi.org/10.1128/CMR.00026-08 [ Links ]
Huchzermeyer, F.W., 2003, Crocodiles: Biology, husbandry and diseases, CABI Publishing, Wallingford. http://dx.doi.org/10.1079/9780851996561.0000 [ Links ]
Hurnikova, Z., Dubinsky, S., Mukaratirwa, S., Foggin, C. & Kapel, C.M.O., 2004, 'Infectivity and temperature tolerance of non-encapsulating Trichinella zimbabwensis in experimentally infected red foxes (Vulpes vulpes), Helminthologia 41, 189-192. [ Links ]
Kapel, C., Henrikson, S.A., Berg, T.B. & Nansen, P., 1995, 'Trichinella infections in Arctic foxes from Greenland: Studies and reflections on predilection sites of muscle larvae', Journal of Helminthology 69, 325-330. http://dx.doi.org/10.1017/S0022149X00014905 [ Links ]
Kapel, C., Henrikson, S.A., Dietz, H.H., Henrikson, P. & Nansen, P., 1994, 'A study on the predilection sites of Trichinella spiralis larvae in experimentally infected foxes (Alopex lagopus, Vulpes vulpes)' Acta Veterinaria Scandinavica 35, 125-132. [ Links ]
Kapel, C., Webster, P. & Gamble, H., 2005, 'Muscle distribution of sylvatic and domestic Trichinella larvae in production animals and wildlife', Veterinary Parasitology 132, 101-105. http://dx.doi.org/10.1016/j.vetpar.2005.05.036 [ Links ]
Kilkenny, C., Brown, W.J., Cuthill, I.C., Emerson, M. & Altman, D.G., 2010, 'Improving bioscience research reporting: The ARRIVE guidelines for reporting animal research', PLoS Biology 8, e1000412. http://dx.doi.org/10.1371/journal.pbio.1000412 [ Links ]
Kociecka, W., Van Knapen, F. & Ruitenberg, E.J., 1980, 'Trichinella pseudospiralis and T. spiralis infections in monkeys, I: Parasitological aspects', in C.W. Kim, E.J. Ruitenberg & J.S. Teppema (eds.), Proceedings of the Fifth International Conference on Trichinellosis, Noordwijk aan Zee, The Netherlands, September 1-5, 1980, pp. 199-202. [ Links ]
La Grange, L.J., Govender, D. & Mukaratirwa, S., 2013, 'The occurrence of Trichinella zimbabwensis in naturally infected wild crocodiles (Crocodylus niloticus) from the Kruger National Park, South Africa', Journal of Helminthology 87, 91-96. http://dx.doi.org/10.1017/S0022149X12000089 [ Links ]
Ludovisi, A., La Grange, L.J., Gómez-Morales, M.A. & Pozio, E., 2013, 'Development of an ELISA to detect the humoral immune response to Trichinella zimbabwensis in Nile crocodiles', Veterinary Parasitology 194(2-4), 189-192. http://dx.doi.org/10.1016/j.vetpar.2013.01.053 [ Links ]
Nöckler, K. & Kapel, C.M.O., 2007, 'Detection and surveillance for Trichinella: Meat inspection hygiene, and legislation', in J. Dupouy-Camet & K.D. Murrell (eds.), FAO/WHO/OIE guidelines for the surveillance, management, prevention and control of trichinellosis, pp. 69-97, World Organisation for Animal Health Press, Paris. [ Links ]
Pozio, E., Marucci, G., Casulli, A., Sacchi, L., Mukaratirwa, Foggin, C.M. et al., 2004, 'Trichinella papuae and Trichinella zimbabwensis induce infection in experimentally infected varans, caimans, pythons and turtles', Parasitology 128, 333-342. http://dx.doi.org/10.1017/S0031182003004542 [ Links ]
Reina, D., Munoz-Ojeda, M. & Serrano, F., 1996, 'Experimental trichinellosis in goats', Veterinary Parasitology 62, 125-132. http://dx.doi.org/10.1016/0304-4017(95)00835-7 [ Links ]
Richardson, K.C., Webb, G.J.W. & Manolis, S.C., 2002, Crocodiles inside out: A guide to the crocodilians and their functional morphology, Surrey Beatty, Chipping Norton, NSW. [ Links ]
Serrano, F.J. & Pérez-Martín, J., 1999, 'Influence of infection intensity on predilection sites in swine trichinellosis', Journal of Helminthology 73, 251-254. [ Links ]
Soule, C., Dupouy-Camet, J., Georges, P., Ancelle, T., Gillet, J.P., Vaissaire, J. et al., 1989, 'Experimental trichinellosis in horses: Biological and parasitological evaluation', Veterinary Parasitology 31, 19-36. http://dx.doi.org/10.1016/0304-4017(89)90005-8 [ Links ]
SABS Standards Division, 2009, Crocodiles in captivity (SANS 631:2009), South African Bureau of Standards, Pretoria. [ Links ]
Wright, K.A., Matta, I., Hong, H.P. & Flood, N., 1989, 'Trichinella larvae and the vasculature of the murine diaphragm', in C.E. Tanner, A.R. Martinez Fernandez & F. Bolas-Fernandez (eds.), Trichinellosis, pp. 70-75, CSIC Press, Madrid. [ Links ]
 Correspondence:
Correspondence:
Louis La Grange
Private Bag X11309
Nelspruit 1200, South Africa
Email: croc.research@gmail.com
Received: 29 July 2013
Accepted: 16 Jan. 2014
Published: 21 Feb. 2014














