Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Onderstepoort Journal of Veterinary Research
On-line version ISSN 2219-0635
Print version ISSN 0030-2465
Onderstepoort j. vet. res. vol.80 n.1 Pretoria Jan. 2013
ORIGINAL RESEARCH
Construction and immunogenicity of a ΔapxIC/ompP2 mutant of Actinobacillus pleuropneumonias and Haemophilus parasuis
Qiong Liu; Yuheng Gong; Yuqin Cao; Xintian Wen; Xiaobo Huang; Qigui Yan; Yong Huang; Sanjie Cao
College of Veterinary Medicine and Institute of Preventive Veterinary Medicine, Sichuan Agricultural University, China
ABSTRACT
The apxIC genes of the Actinobacillus pleuropneumoniae serovar 5 (SC-1), encoding the ApxI-activating proteins, was deleted by a method involving sucrose counter-selection. In this study, a mutant strain of A. pleuropneumoniae (SC-1) was constructed and named AapxIC/ ompP2. The mutant strain contained foreign DNA in the deletion site of ompP2 gene of Haemophilus parasuis. It showed no haemolytic activity and lower virulence of cytotoxicity in mice compared with the parent strain, and its safety and immunogenicity were also evaluated in mice. The LD50 data shown that the mutant strain was attenuated 30-fold, compared with the parent strain (LD50 of the mutant strain and parent strain in mice were determined to be 1.0 x 107 CFU and 3.5 x 105 CFU respectively). The mutant strain that was attenuated could secrete inactivated ApxIA RTX toxins with complete antigenicity and could be used as a candidate live vaccine strain against infections of A. pleuropneumoniae and H. parasuis.
Introduction
Actinobacillus pleuropneumoniae (AP) is the causative agent of porcine pleuropneumoniae, a highly contagious disease contributing to fatalities in pig production worldwide (Sebunya & Saunders 1983). The pathogen is transmitted by air or direct contact with infected pigs (Chiers et al. 2002). Haemophilus parasuis is a commensal bacterium in the upper respiratory tract of conventional pigs, but under appropriate conditions, can invade and cause severe systemic disease (Glasser's disease) (Hoefling 1991). Both bacteria are members of the family of Pasteurellaceae; their growth requires a V factor (NAD), but no X factor (hemin). In recent years, the porcine industry has suffered a significant increase in the incidence, morbidity and mortality associated with Glasser^s disease (Oliveira & Pijoan 2004) an infection often combined with AP, which has caused serious economic loss world-wide.
The Apx is characterised as one of the major virulence determinants of A. pleuropneumoniae (Frey & Nicolet 1990). Up till now, four different Apx toxins (ApxI to ApxIV) have been described and varying combinations of Apx toxins are produced in different A. pleuropneumoniae serovars. For instance, serovars 1, 5, 9 and 11 generate ApxI and ApxII (Tascon et al. 1993). Amongst the Apx toxins, ApxI, which is secreted by the most virulent serovars such as serovars 1, 5, 9, 10 and 11, has shown strong haemolytic activity and cytotoxic activity (Kamp et al. 1991). Meanwhile, ApxI, ApxII and ApxIII are strongly immunogenic and involved in the induction of protective immunity. The toxin ApxI is encoded on polycistronic operons apxICABD (Frey et al. 1994). In these operons, the A gene encodes the protoxin, whilst the C gene encodes a cyltransferase, which is involved in the post-translational activation of the protoxin in the cytoplasm (Issartel, Koronakis & Hughes 1991). The B and D genes respectively encode an ATPase and a protein adaptator to facilitate Apx toxin secretion (Thanabalu et al. 1998).
The inactivated whole-bacteria vaccine has been demonstrated to elicit protection against infection from the same serovar of A. pleuropneumoniae (Hensel et al. 2000) and subunit vaccines have been tested to provide some immunity with reduction in clinical symptoms and pulmonary lesions (Goethe et al. 2000). However, in terms of inactivated whole-bacteria vaccine, it was not able to generate cross protection to infection from heterologous serovars or to provide long-lasting immunity. Secondly, in terms of subunit vaccines, although the subunit vaccines contained the mixed outer membrane proteins and although proteins secreted from several serovars or recombinant toxins have been shown to protect animals from clinical infection (Maas et al. 2006), the relatively higher production cost of mixing different antigens would undoubtedly limit their application. In contrast, pigs surviving infection with one serovar are at least partially protected against infection by other serovars. Consequently, developing a live vaccine by attenuation of A. pleuropneumoniae has become a dominant research field.
The outer membrane protein P2 (ompP2) of H. parasuis is encoded by the ompP2 gene and is an immunodominant porin with considerable antigenic heterogeneity amongst different serovars of H. parasuis (Mullins et al. 2009). The ompP2 has been targeted as a potential vaccine candidate. As a porin, ompP2 plays a role in colonisation and has been shown binding to specific components of human mucin (Reddy et al. 1996). It was demonstrated that ompP2 could be expressed in H. parasuis and that it shows good antigenicity.
Inactivated bacterial vaccines and subunit vaccines are currently the main ways of preventing porcine pleuropneumoniae. However, the multitude of serovars (15 serovars) and the limited cross-protection conferred by bacterins or Apx toxin-based subunit vaccines (Jolie, Mulks & Thacker 1995) makes treatment of A. pleuropneumoniae infection difficult. A live vaccine is considered to offer the best prospects of obtaining cross-protection against this bacterium. In the current study, an attenuated mutant A. pleuropneumoniae strain with the ompP2 gene of H. parasuis serovar 5 (Nagasaki) (Takahashi et al. 2001) was developed as a potential vaccine candidate without introducing an antibiotic resistance marker by using a sucrose counter-selection strategy (Oswald et al. 1999). Actinobacillus pleuropneumoniae serovar 5 (SC-1), is one of the predominant serovars in China, and produces both apxI and apxII toxins. The apxIC region was targeted for disruption with a gene of H. parasuis to produce a mutant strain that secretes an inactive toxin with full antigenic properties. Here, we describe the construction of the homologous recombination vector, the characterisation of the resultant mutant strain and the testing of its virulence and protective efficacy against A. pleuropneumoniae and H. parasuis infection in mice.
Materials and methods
Bacteria isolates and growth conditions
Haemophilus parasuis serovar 5 (Nagasaki) and A. pleuropneumoniae serovar 5 (SC-1) were selected for this study. The bacterial strains were both grown on tryptic soy agar (TSA, DifcoTM, BD) and in tryptic soy broth (TSB, DifcoTM, BD) supplemented with 1% (wt/vol) NAD. All bacterial strains were grown at 37 °C.
Escherichia coli DH5α was routinely maintained in Luria-Bertani medium (LB) or maintained in LB supplemented with 100 mg/mL of ampicillin or 50 mg/mL of kanamycin when containing the relevant vector.
The vector pBOSKAIC, constructed by Dr Guobin Wang, was used for constructing the vector of the homologous recombination.
Construction of vector for homologous recombination
The first step was to transform the restriction site on the right homologous arm of homologous recombination vector pBOSKAIC with SpeI and MluI. The primers used in this study are listed in Table 1. A PCR of the right homologous arm was conducted by initial denaturing at 94 °C for 5 min, followed by 30 cycles of denaturing at 94 °C for 45 sec, annealing at 55 °C for 30 sec and extension at 72 °C for 1.5 min, with a final extension at 72 °C for 10 min. The reconstructive vector was called pBOSKΔIC-1. The ompP2 gene of H. parasuis was then amplified by PCR with the same PCR conditions as described above, The amplicons of ompP2 were checked in agarose gel electrophoresis, purified and cloned in pMD-simple-19T vector (TaKaRa Biotechnology [Dalian] Co., Ltd), followed by subcloning between SpeI and MluI sites of the reconstructive vector pBOSKΔIC-1. The vector was use for homologous recombination and called pBOSKΔapxIC/ ompP2 (Figure 1).

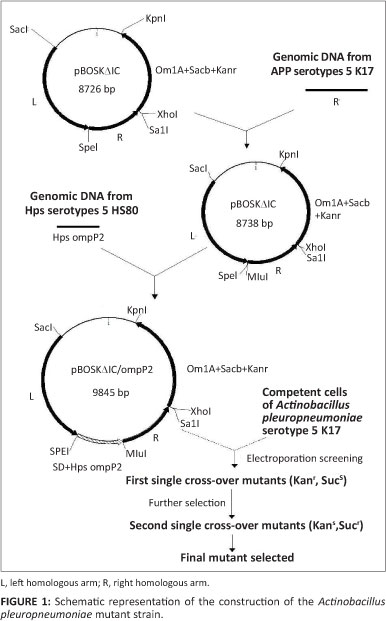
Electroporation of Actinobacillus pleuropneumoniae and selection of recombinants
The transfer vector pBOSKΔapxIC/ompP2 was extracted by TM Vector Mini Kit II (OMEGA). The competent cells of A. pleuropneumoniae serovar 5 (SC-1) were prepared according to the general optimisation protocol for the transformation of bacteria provided by the Electroporation System (Gene Pulser Xcell™, BIO-RAD). Pulse conditions were as follows: cuvette 0.2 cm-diameter; voltage 2.5 kV; capacitance 25 uF; resistance 800 Ω; time constant 5 ms. After electroporation, the cells were transferred into 1 mL TSB supplemented with NAD and incubated immediately at 37 °C, at 180 r/min for 3 h. The recovered cells were plated onto the TSA plates, supplemented with 1% (wt/vol) NAD and 50 ug/mL of kanamycin, incubated overnight at 37 °C.
Kanamycin resistant colonies were selected and inoculated onto TSA-sucrose plates, which were incubated at 37 °C to confirm that the sucrose sensitivity was conferred by transfer of the vector (Figure 1).
Identification
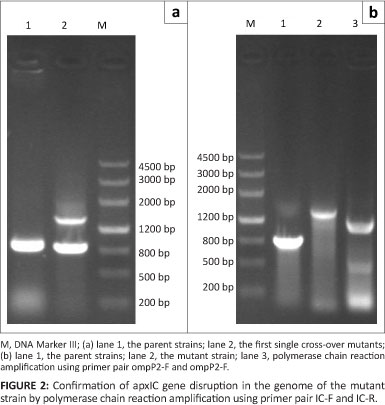
The genomic DNA of the parent and mutant strains was extracted individually. Primer pairs IC-F and IC-R were used to amplify the relevant fragments from the genomic DNA of the parent and mutant strains. The DNA fragments of these strains were separated on a 1% agarose gel.
Growth and genetic stability of the mutant strain
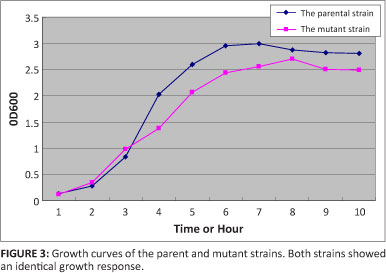
The mutant strain was inoculated into TSB supplemented with 1% (wt/vol) NAD, The bacterial concentration was determined at OD600 every hour, and the value recorded. The growth curves were drawn to compare features of growth cycles.
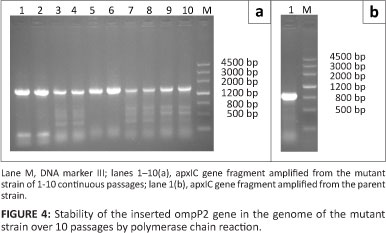
The genetic stability of the inserted ompP2 gene in the genome of the mutant strain was tested. This was done by propagating the mutant strain for 10 passages serially in TSB supplemented with 1% (wt/vol) NAD, and then amplifying the relevant fragments by PCR from the colony of each passage.
Haemolytic activity and NAD dependency test
The haemolytic activity and NAD dependency of the parent and mutant strains were examined. Briefly, they were inoculated onto sheep blood agar plates overnight at 37 °C. Escherichia coli DH5α was used as negative control. The haemolytic activity of the strains was recorded. At the same time, the growth was observed of the mutant strain inoculated in the TSB, supplemented with 1% (wt/vol) NAD and incubated overnight at 37 °C.
Safety and vaccination efficacy of the mutant strain in mice
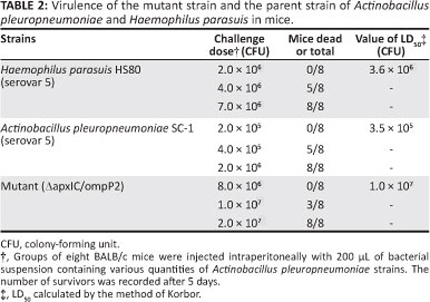
Cultures of A. pleuropneumoniae serovar 5 were grown overnight at 37 °C in TSB supplemented with NAD, followed by dilution to 1:1000. The diluted culture was incubated again until the OD600 reached 0.8. At this point, the viable count of A. pleuropneumoniae ΔapxIC/ompP2 was found to three concentrations (Table 2). For the virulence test, three groups of eight 6-week-old female Balb/C mice (Sichuan Province Huaxi Experimental Animal Centre, Chengdu, China) were injected intraperitoneally (i.p.) with 200 µL TSB medium containing various bacterial counts of A. pleuropneumoniae or H. parasuis. Survival of the mice was recorded after 72 h (Table 2). The lethal dose of 50% (LD50) value of the mutant strain AapxIC/ompP2 was calculated by Karber's method.
Serological measurement
Serum was isolated from whole blood collected from mice at four points in time: before vaccination (day 0), before the secondary vaccination (day 14), before the third vaccination (day 28) and before challenge (day 42). Inactivated A. pleuropneumoniae and H. parasuis were used as a somatic antigen by ELISA. Briefly, the 96-well Enzyme-linked immunosorbent assay (ELISA) microplates were coated overnight at 4 °C with 0.08 mg/mL of inactivated A. pleuropneumoniae and H. parasuis diluted in 100 mL of 50 mM sodium carbonate buffer, pH 9.6. Plates were washed three times with washing buffer (0.05% Tween 20 in PBS, PBST) and blocked for 1 h at 37 °C with blocking buffer (1% BSA in PBST). After three washes, serum samples were diluted to 1:100 in blocking buffer, added to each well (100 uL per well) in duplicate and incubated for 1 h at 37 °C. After three washes, 100 µL of HRP-conjugated goat antis mice IgG (SBA, America), diluted to 1:5000 in blocking buffer, was added to each well and incubated at 37 °C for 1 h. After three washes, 50 uL TMB was added to each well at room temperature in dark reaction for 15 min The reaction was then stopped by adding 100 uL of 2 M H2SO4 per well. The absorbance was read at 450 AA with an ELISA micoplate reader (MODEL 680, BIO-RAD).
Results
Identification of Actinobacillus pleuropneumoniae apxIC mutant lacking antibiotic resistance markers
After electroporation of A. pleuropneumoniae competent cells with the vector pBOSKΔapxIC/ompP2, some transformants were screened from growth on TSA plates containing kanamycin and sucrose, respectively. Three transformants with the phenotype of Kanr and Sucs were chosen for propagation in TSB medium without kanamycin or sucrose to promote the second order recombination event and excised the kanamycin resistance and sacB genes. Finally, the cells were further grown overnight in TSB-NAD in the absence of kanamycin, then grown onto TSA-Sucrose plates. The Sucr colonies were replica plated onto TSA-Sucrose and TSA-Sucrose-Kanr plates to choose the colonies with the Kans and Sucr phenotype.
The ApxIC gene of A. pleuropneumoniae was identified by PCR (Figure 2), the genomic DNA of the parental strain had an amplicon of about 800 bp and the mutant stain an amplicon about 1400 bp. The amplification of the ompP2 gene of H. parasuis yielded a band of 1100 bp. This suggests that the ompP2 gene of H. parasuis was inserted into the ApxIC gene of A. pleuropneumoniae at the deletion site.
Growth and genetic stability of the mutant strain
No obvious difference was observed in vitro growth curves of the parental strain and mutant strains, indicating that the ompP2 gene of H. parasuis that took the place of the apxIC gene had no significant influence on the growth of A. pleuropneumoniae (Figure 3).
A PCR fragment with a size of 1400 bp was amplified from all ten serial passages in TSB supplemented with 1% (wt/vol) NAD, indicating that the ompP2 gene of H. parasuis was stably inserted into the genome of the mutant strain, and the apxIC gene of A. pleuropneumoniae had been deleted (Figure 4).
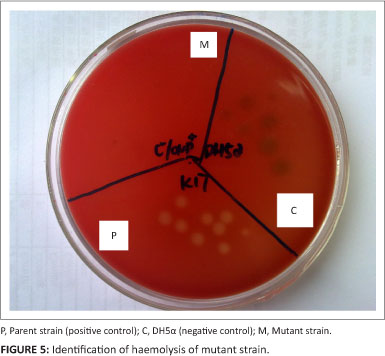
Haemolytic activity test
The functional activity of the ApxI toxin produced by ΔapxIC/ompP2 was examined in the haemolytic assay. Fully active ApxI toxin had haemolytic activity, as demonstrated by clear zones surrounding the colonies of A. pleuropneumoniae on sheep blood agar (Figure 5, Section P). The apxIC gene of A. pleuropneumoniae was deleted, and the ompP2 gene of H. parasuis showed no haemolytic activity, thus the mutant strain was lacking haemolytic activity. This was evident from the absence of a clear zone surrounding the colonies of the A. pleuropneumoniae mutant on sheep-blood agar (Figure 5, Section M).
Safety of the mutant strain in mice
To test the virulence of the mutant strain, various concentrations of A. pleuropneumoniae ΔapxIC/ompP2 and serovar 5 of H. parasuis and A. pleuropneumoniae parent strain were prepared and administrated to mice via the intraperitoneal route. No deaths were observed in mice that received 2.0 x 106 CFU of H. parasuis and 8.0 x 106 CFU of AapxIC/ompP2, whilst 100% mortality was recorded in mice that received the parent strain at doses of 2.0 x 106 CFU. Therefore, compared to the parent strain and serovar 5 of H. parasuis, virulence of the mutant was attenuated at least 30-fold (Table 2).
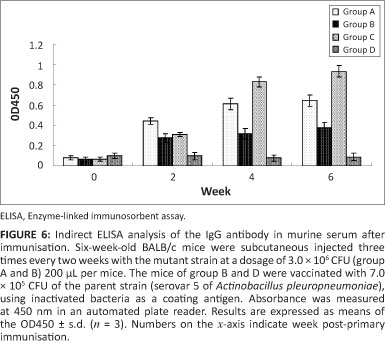
Serological measurement
Serum was isolated from whole blood collected from mice at four points in time: before vaccination (day 0), before the secondary vaccination (day 14), before the third vaccination (day 28) and before challenge (day 42). Antibodies against the somatic antigen of inactivated A. pleuropneumoniae and H. parasuis were examined using ELISA (see Figure 6). The ELISA titres were defined as the highest serum dilution resulting in OD450 twice and the negative control serum at a dilution of 1:100.
Discussion
It is known that ApxI toxin is a very important member of the RTX family and common structural features are present in the RTX toxins. When used as a subunit vaccine, it could offer a good level of protection in mice against A. pleuropneumoniae infection (Seah, Frey & Kwang 2002). The ApxIC contains 13 repeated nonapeptides and plays an important role during the interaction between ApxI toxins (Issartel et al. 1991). In earlier studies, expression of the apxC gene was generally inactivated by insertion of foreign marker genes or by deletion of the gene (Liu et al. 2007).
These mutant strains have the potential to reverse virulence by releasing the inserted genes and transmitting the marked antibiotic resistance gene to the animal's normal bacterial flora. Live vaccines are considered to offer the best prospect for obtaining cross-protection against A. pleuropneumoniae serovar 5 and H. parasuis serovar 5. In 1999, Oswald constructed an A. pleuropneumoniae UreaC mutant strain using a new method, the sucrose counter-selectable system, but its application in A. pleuropneumoniae has been limited so far (Oswald et al. 1999). The method demonstrated that it is possible to modify the A. pleuropneumoniae genome without inserting an antibiotic resistance marker.
Based on these previous studies, we constructed a live attenuated ApxI inactivation mutant strain by deleting the ApxIC toxin, and inserting the ompP2 gene of H. parasuis. The A. pleuropneumoniae serovar 5 (SC-1) was selected as the parent strain for the mutant construction, because it is one of the most prevalent serovars in China, and is capable of secreting ApxI and ApxII.
The results indicate that the mutant strain ΔapxIC/ ompP2 is capable of secreting ApxI, ApxII and ompP2 of H. parasuis after deletion of apxIC as the ΔapxIC/ompP2 still contains apxIB and apxID genes, which encode proteins that are essential for the secretion of the toxin (Welch 1991). Compared with the parent strain, virulence of the mutant strain (ΔapxIC/ompP2) was attenuated 30-fold, providing more evidence that the ApxI of A. pleuropneumoniae serovar 5 is necessary for full virulence (Goethe et al. 2000).
In terms of haemolytic activity, the mutant strain had lower visible haemolytic activity on agar plates. It was proved that apxIC gene was absence from the genome of A. pleuropneumoniae (SC-1). The serological measurement of mice serum show that the mutant strain provided lower protection against H. parasuis than A. pleuropneumoniae. This is possibly because A. pleuropneumoniae is the parent strain, whilst the expression of ompP2 gene in the mutant strain is not enough to protect against invasion of H. parasuis. Another aspect is that the ompP2 gene is not the most important antigen offi. parasuis, but the mutant strain could still protect miceagainst H. parasuis and A. pleuropneumoniae.
In summary, the mutant strain AapxIC/ompP2 had greatly reduced virulence. It needs to be pointed out that the mouse model is only a preliminary assay and full studies in pigs are needed to test this mutant in future.
Conclusion
In this study, we successfully constructed a mutant strain of A. pleuropneumoniae (SC-1) named AapxIC/ompP2, whilch can provide the protection against H. parasuis and A. pleuropneumoniae.
Acknowledgements
This work was supported by the Science Expertise of Public Interest Career - Agriculture (No.201303034).
Competing interests
The authors declare that they have no financial or personal relationship(s) which may have inappropriately influenced them in writing this article.
Authors' contributions
S.C. (Sichuan Agricultural University) was the project leader, Q.L. (Sichuan Agricultural University) and Y.G. (Sichuan Agricultural University) were responsible for experimental and project design and Y.C. (Sichuan Agricultural University), X.W. (Sichuan Agricultural University) , X.H. (Sichuan Agricultural University), Y.H. (Sichuan Agricultural University) and Q.Y. (Sichuan Agricultural University) performed some of the experiments.
References
Chiers, K., Donne, E., Van Overbeke, I., Ducatelle, R. & Haesebrouck, F., 2002, 'Actinobacillus pleuropneumoniae infections in closed swine herds: infection patterns and serological profiles', Veterinary Microbiology 85, 343-352. http:// dx.doi.org/10.1016/S0378-1135(01)00518-1 [ Links ]
Frey, J., Haldimann, A., Nicolet, J., Boffini, A. & Prentki, P., 1994, 'Sequence analysis and transcription of the apxI operon (hemolysin I) from Actinobacillus pleuropneumoniae', Gene 142, 97-102. http://dx.doi.org/10.1016/0378-1119(94)90361-1 [ Links ]
Frey, J. & Nicolet, J., 1990, 'Haemolysin patterns of Actinobacillus pleuropneumoniae', Journal of Clinical Microbiology 28, 232-236. [ Links ]
Goethe, R., Gonzales, O.F., Lindner, T. & Gerlach, G.F., 2000, 'A novel strategy for protective Actinobacillus pleuropneumoniae subunit vaccines: Detergent extraction of cultures induced by iron restriction', Vaccine 19, 966-975. http://dx.doi.org/10.1016/S0264-410X(00)00212-7 [ Links ]
Hensel, A., Huter, V., Katinger, A., Raza, P., Strnistschie, C. & Roesler, U., 2000, 'Intramuscular immunization with genetically inactivated (ghosts) Actinobacillus pleuropneumoniae serotype 9 protects pigs against homologous aerosol challenge and prevents carrier state', Vaccine 18, 2945-2955. http://dx.doi.org/10.1016/S0264-410X(00)00107-9 [ Links ]
Hoefling, D.C., 1991, 'Acute myositis associated with Haemophilus parasuis in primary SPF sows', Journal of Veterinary Diagnostic Investigation 3, 354-345. http://dx.doi.org/10.1177/104063879100300418 [ Links ]
Issartel, J.P., Koronakis, V. & Hughes, C., 1991, 'Activation of Escherichia coli prohaemolysin to the mature toxin by acyl carrier protein-dependent fatty acylation', Nature 351, 759-761. http://dx.doi.org/10.1038/351759a0 [ Links ]
Jolie, R.A., Mulks, M.H. & Thacker, B.J., 1995, 'Cross-protection experiments in pigs vaccinated with Actinobacillus pleuropneumoniae subtypes 1A and 1B', Veterinary Microbiology 45, 383-391. http://dx.doi.org/10.1016/0378-1135(94)00145-M [ Links ]
Kamp, E.M., Popma, J.K., Anakotta, J. & Smits, M.A., 1991, 'Identification of haemolytic and cytotoxic proteins of Actinobacillus pleuropneumoniae by use of monoclonal antibodies', Infection and Immunity 59, 3079-3085. [ Links ]
Liu, J.L., Bei, W.C., Lin, L.W., Xu, Y.D. & Chen, H.C., 2007, 'Construction and characteristics of a recombinant strain apx II C- /apx I A+ of Actinobacillus pleuropneumoniae serotype 7', WeiSheng Wu Xue Bao 47, 973-977. [ Links ]
Maas, A., Meens, J., Baltes, N., Hennig-Pauka, I. & Gerlach, G.F., 2006, 'Development of a DIVA subunit vaccine against Actinobacillus pleuropneumoniae infection', Vaccine 24, 7226-7237. http://dx.doi.org/10.1016/j~.vaccine.2006.06.047 [ Links ]
Mullins, M.A., Register, K.B., Bayles, D.O., Loving, C.L., Nicholson, T.L. & Brockmeier, S.L., 2009, 'Characterization and comparative analysis of the genes encoding Haemophilus parasuis outer membrane proteins P2 and P5', Journal of Bacteriology 191, 5988-6002. http://dx.doi.org/10.1128/JB.00469-09 [ Links ]
Oliveira, S. & Pijoan, C., 2004, 'Haemophilus parasuis: New trends on diagnosis, epidemiology and control', Veterinary Microbiology 99, 1-12. http://dx.doi.org/10.1016/j.vetmic.2003.12.001 [ Links ]
Oswald, W., Tonpitak, W., Ohrt, G. & Gerlach, G., 1999, 'A single-step transconjugation system for the introduction of unmarked deletions into Actinobacillus pleuropneumoniae serotype 7 using a sucrose sensitivity marker', FEMS Microbiology Letters 179, 153-160. http://dx.doi.org/10.1111/j.1574-6968.1999.tb08721.x [ Links ]
Reddy, M.S., Bernstein, J.M., Murphy, T.F. & Faden, H.S., 1996, 'Binding between outer membrane proteins of nontypeable Haemophilus influenzae and human nasopharyngeal mucin', Infection and Immunity 64, 1477-1479. [ Links ]
Seah, J.N., Frey, J. & Kwang, J., 2002, 'The N-terminal domain of RTX toxin ApxI of Actinobacillus pleuropneumoniae elicits protective immunity in mice', Infection and Immunity 70, 6464-6467. http://dx.doi.org/10.1128/IAI.70.11.6464-6467.2002 [ Links ]
Sebunya, T.N. & Saunders, J.R., 1983, 'Haemophilus pleuropneumoniae infection in swine: A review', Journal of The American Veterinary Medical Association 182, 1331-1337. [ Links ]
Tascon, R.I., Rodriguez-Ferri, E.F., Gutierrez-Martin, C.B., Rodriguez-Barbosa, I., Berche, P. & Vazquez-Boland, J.A., 1993, 'Transposon mutagenesis in Actinobacillus pleuropneumoniae with a Tn10 derivative', Journal of Bacteriology 175, 5717-5722. [ Links ]
Takahashi, K., Naga, S., Yagihashi, T., Ikehata, T., Nakano, Y. & Senna, K., 2001, 'A cross-protection experiment in pigs vaccinated with Haemophilus parasuis serovars 2 and 5 bacterins, and evaluation of a bivalent vaccine under laboratory and field conditions', Journal of Veterinary Medical Science 63, 487-491. http://dx.doi.org/10.1292/jvms.63.487 [ Links ]
Thanabalu, T., Koronakis, E., Hughes, C. & Koronakis, V., 1998, 'Substrate-induced assembly of a contiguous channel for protein export from E. coli: Reversible bridging of an inner-membrane translocase to an outer membrane exit pore', Embo Journal 17, 6487-6496. http://dx.doi.org/10.1093/emboj/17.22.6487 [ Links ]
Welch, R.A., 1991, 'Pore-forming cytolysins of gram-negative bacteria', Molecular Microbiology 5, 521-528. http://dx.doi.org/10.1111/j.1365-2958.1991.tb00723.x [ Links ]
![]() Correspondence to:
Correspondence to:
Sanjie Cao
Postal address: 46 Xinkang Road
Yucheng District, Yaan city, Sichuan Province, China
Email: csanjie@gmail.com
Received: 09 Sept. 2012
Accepted: 03 Dec. 2012
Published: 06 Mar. 2013














