Servicios Personalizados
Articulo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares en Google
Similares en Google
Compartir
South African Dental Journal
versión On-line ISSN 0375-1562
versión impresa ISSN 0011-8516
S. Afr. dent. j. vol.73 no.4 Johannesburg may. 2018
REVIEW
Insights into the "Stemness" of Cultured Adult Human Dental Pulp Fibroblast-like cells
Basson NJI; Moodley DSII; Olivier AIII; Hiss DIV
IPhD, Oral and Dental Laboratory, Dental Faculty, University of the Western Cape, Cape Town
IIDS Moodley PhD, Oral and Dental Laboratory, Dental Faculty, University of the Western Cape, Cape Town
IIIA Olivier MSc, Oral and Dental Laboratory, Dental Faculty, University of the Western Cape, Cape Town
IVD Hiss PhD, Department of Medical Biosciences, University of the Western Cape Cape Town
INTRODUCTION
Fibroblasts are the most numerous cells occurring in the dental pulp. Although distributed throughout the tissue, they are particularly abundant in the coronal portion of the pulp in the cell-rich zone. The function of pulpal fibroblasts is to produce and maintain the proteins of the extracellular matrix of the pulp.1
Other fibroblast-like cells also occurring throughout the cell-rich zone and the pulp core, and often related to blood vessels, are undifferentiated mesenchymal cells. These cells represent the pool of cells from which the connective tissue cells of the pulp, such as odontoblasts and fibroblasts, are formed.1 In recent times, a more modern term for undifferentiated mesenchymal cells has emerged: "adult mesenchymal stem cells" (MSC's). They appear to be tissue-specific cells capable of giving rise to cells that are committed to differentiation once given the proper signal.
Adult MSC's have a appearance similar to small young fibroblasts, are indistinguishable from fibroblasts morphologically and share the same expression of surface antigens as fribroblasts.2 They are however far more multipotent in terms of differentiation into cell types. Defined as undifferentiated cells that can continuously produce unaltered daughter cells, they also have the ability under specific growth conditions to differentiate into multiple lineages of mesenchymal tissues such as osteogenic, adipogenic and myogenic cells. Given these properties, there is an enormous potential for the application of MSC's for cell therapy in regenerative medicine and tissue engineering in the craniofacial region.3 The capacity to stimulate stem cells in culture is indispensable in regenerative medicine.4 Therefore the isolation of adult MSC's from dental tissue has drawn a lot of interest amongst scientists over the last number of years, The cells have been isolated from a number of different types of human dental tissue, including human exfoliated deciduous teeth,6 periodontal ligament,6 dental follicle,7 apical papilla,8 and the adult dental pulp.9,10
Although significant information has been gained about MSC's over the last number of years, important questions remain unanswered regarding the identity of these cells, such as their relation to fibroblasts.2'1113 Both fibroblasts and MSC's are plastic adherent cells that may be isolated from adult dental pulp using tissue culture techniques. In our laboratory we found it easy to isolate and culture "fibroblasts" from human dental pulp by using the explant method whereby cells grow out of a piece of tissue placed in a suitable growth medium.14 However, since adult MSC's have an appearance similar to small young fibroblasts and indeed are indistinguishable from fibroblasts morphologically, the question arose as to their true identity. Therefore we investigated the "sternness" of these cultured fibroblast-like cells and the ease of establishing specific tissue lines from them.
MATERIALS AND METHODS
Isolation of fibroblast-like cells
This research project was registered with the Ethical Committee of the University of the Western Cape. Ethical clearance for the collection and use of extracted teeth was obtained from each individual donor. Impacted third molars and premolars removed for orthodontic purposes were used in the isolation of fibroblasts, Twenty-one freshly extracted teeth, from 21 individuals, were used. The teeth were collected in a carrier medium comprising Dulbecco's modified Eagle medium (DMEM), 10% foetal bovine serum, 1% penicillin, 1% streptomycin, 0.2% fungizone and 0.2% gentamycin. Within 24 hours after removal, the teeth were placed in gauze, cracked open with a vice and the pulps removed (Figure 1). The pulps were rinsed several times in phosphate buffered saline (PBS) and the middle-third of the pulp removed, cut into small pieces and placed in petri dishes. Each piece of tissue was covered with a drop of foetal bovine serum (FBS) to facilitate attachment to the petri dish and allowed to dry for one hour after which DMEM (without fungizone and gentamycin) was added.
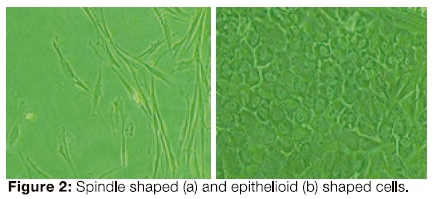
The specimens were incubated at 37°C in an atmosphere of 5% C02 at a humidity of 95%. After 14 days, the outgrowths of cells (Figure 2) were dissociated using trypsin-versene for five minutes and then the cells were stored in liquid nitrogen. In due course, and as was required, frozen cells from each cell line were gently thawed and grown, using standard procedures.
Morphological characteristics
The general morphology of the fibroblast cells was observed microscopically using an Olympus CK2 inverted microscope at 400X magnification. Cells were grown to confluence and at the first passage the cell lines were allowed to grow for 24 hours in 5ml petri dishes containing glass coverslips to which the cells would attach. After washing in PBS, the attached cells were fixed with cold acetone for 20 minutes, washed again in PBS and then stained with 1% crystal violet for 10 minutes. The stained cells were again washed, dehydrated in alcohol and xylene and mounted on glass slides. The remaining sub-lines were allowed to continue growing to confluence, and at the second and third passages, were treated as above.
Growth parameters
Cells grown in DMEM were dissociated with trypsin-versene and plated in 24-well plates at a seeding density of 1.2 χ 104 cells/ml. The cells were grown with a medium change every third day until confluence was obtained after eight days. Every 24 hours the cells from three randomly selected wells were dissociated to obtain cell suspensions and the cells were counted in duplicate with a Neubauer haemocytometer. Logarithms of the average counts against times were used to plot growth curves.
Flow cytometry
To test for MSC properties, fibroblast cells from each cell line were subjected to flow cytometry analysis for expression of positive (CD90, CD105 and CD73) and negative (CD34, CD11b, CD19, CD45 and HLA-DR) MSC markers. The cells, harvested by trypsin-versene for 5 min, were washed in cold PBS supplemented with 0.5% Bovine Serum Albumin and aliquots of 2 χ 105 cells were labelled for 30 min in the dark at 4°C with a Stemflow Human Mesenchymal Stem Cell Analysis Kit. The analysis was performed on a Canto II flow cytometer using an isotope control cocktail (BD Biosciences).
Differentiation Assays
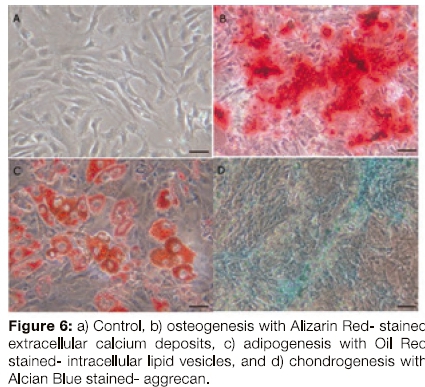
In vitro differentiation of the isolated cells into osteoblasts, adipocytes and chondroblasts were performed using StemPro® Differentiation kits (GIBCO, LTC Tech SA, Johannesburg) for each respective differentiation.
All differentiation protocols were performed at the third passage. For each differentiation, cells were seeded into a 5ml petrl dish and grown to confluence of about 60%. The conventional complete culture medium was then replaced with the induction medium from the StemPo® kit. Medium changes were performed twice a week for two weeks. The cell monolayer was rinsed once with PBS and fixed with 4% formalin for 30 minutes at room temperature. The formalin was removed and the cells rinsed twice with deionized water.
Osteogenesis was shown by staining the monolayer of cells with 2% Alizarin Red S solution for three minutes. The stain was removed and the cells washed three times with deionized water. The red stained calcium-rich extracellular matrix was then observed visually under a light microscope.
Chondrogenesis was shown by staining the monolayer of cells with 1% Alcian Blue solution (prepared in 0,1 Ν HCl) for 30 minutes. The cells were washed three times with 0,1 Ν HCl and once with deionized water. The blue stained proteoglycans synthesised by the chondrocytes were then observed visually under a light microscope.
Adipogenesis was shown by staining the monolayer of cells with Oil Red O solution for 30 minutes. The cells were washed three times with deionized water. The presence of intracellular lipid droplets was observed microscopically.
Colony-forming potential
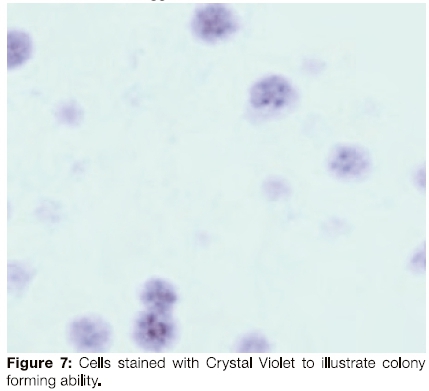
To test for colony-forming ability, cells were grown to 70-80% confluency in DMEM, dissociated with trypsin-versene and plated at about 100 cells per 100-mm tissue culture dish. The cells were Incubated for 10-14 days at 37 °C in 5% humidified C02, washed with PBS and stained with 0.5% Crystal Violet in methanol for 5-10 min at room temperature. The plates were washed twice with PBS and inspected for visible colonies.
RESULTS
Isolation of fibroblast-like cells
The ages of the donors varied from 14 to 35 years with an average of 27.5 years. Only thirteen of the pulp specimens obtained from the 21 individuals produced outgrowth. The rate of outgrowth of cells differed between the various specimens. Some produced a substantial outgrowth of cells that allowed dissociation at 14 days, while others produced no outgrowth. Six pulp cell lines were established. All were able to actively proliferate after freezing and storing under liquid nitrogen.
General morphology
Two different types were observed, a slender spindle shaped cell (Figure 2a), and an epithelioid cell (Figure 2b). When the cells were sparsely distributed in the early stages of the passage, all the pulp cell lines displayed the typical spindle shaped microscopic features of cultured fibroblasts. However, as soon as the cultures were allowed to grow to confluence they tended to become rounded with an epithelioid shape (Figure 2b).
Growth parameters
The recorded growth curves indicated that the cultured cells progressed through a characteristic growth pattern of a lag phase, exponential phase and a plateau phase (Figure 3). The lag phase, during which the cells adjusted to their new environment lasted approximately 22 hours, the phase of exponential growth 40 hours and the change in the slope of the growth curves which showed that confluence was approached was seen at approximately five days. Full confluence was reached at eight days and no significantly higher counts were obtained thereafter.

Flow cytometry
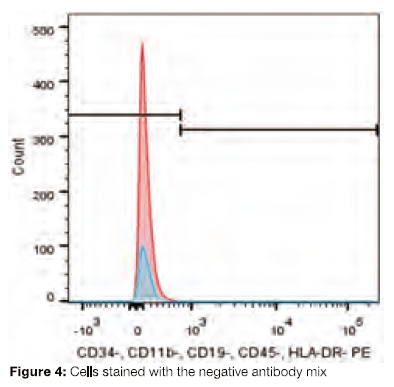
The graph in figure 4 represents the overlay of cells stained with the isotype control (red) and cells stained with the negative antibody mix (blue). The fibroblast cell lines were all negative for CD34, CD11b, CD19 CD45 and HLA-DR.
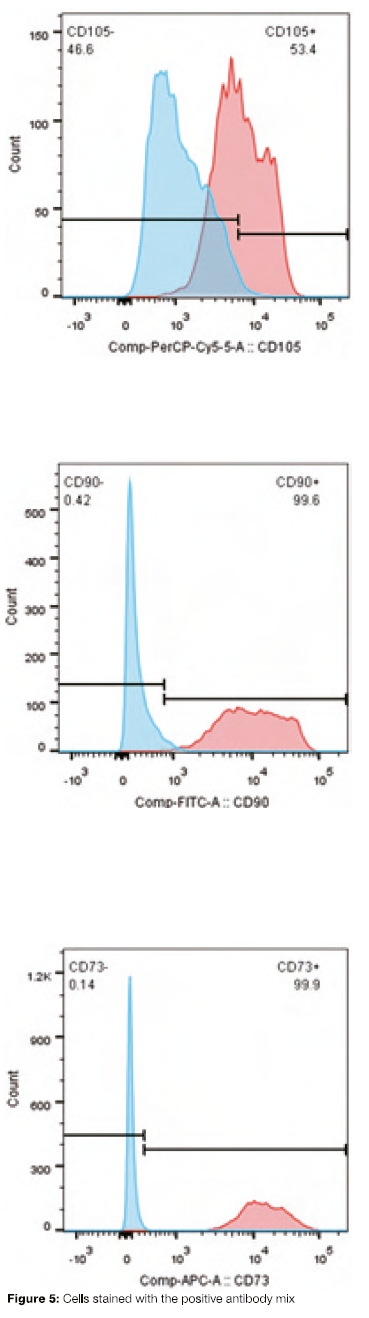
The graphs in figurure 5 represent the overlay of cells stained with the isotype control (red) and cells stained with the positive antibody mix (blue) containing CD90, CD73 and CD105. There was a clear shift in CD90 and CD73, but CD105 showed only a slight shift and only 53.4% of the cells were positive for CD105. On the other hand, 99.6% of the cells were positive for CD90 and 99.9% of the cells were positive for CD73.
Differentiation Assays
Osteogenic, chondrogenic and adipogenic differentiation was observed two weeks after onset of the differentiation protocol. Osteogenic differentiation was shown by the presence of red stained mineralized extracellular matrix deposition (Figure 6b), adipogenic differentiation by the presence of red stained intracellular lipid droplets (Figure 6c) and chondrogenic differentiation by the blue staining of proteoglycans (Figure 6d).
Colony-forming potential
All the cell lines expressed colony forming ability (Figure 7).
DISCUSSION
According to the Mesenchymal and Tissue Stem Cell Committee of the International Society for Cellular Therapy15, MSC's should be plastic-adherent when maintained in standard culture conditions, should express surface markers CD105, CD73, and CD90, but should lack expression of hematopoietic markers CD45, CD34, CD14, or CD11b, CD79 alpha. Furthermore, MSC's should be multipotent with an ability to differentiate at least into osteoblasts, adipocytes, and chondroblasts.15
The cells we isolated from human dental pulps had typical fibroblast morphology. On testing the above mentioned properties we found that these cells complied with all the criteria for MSC's and can therefore be regarded as such.
The low success rate of establishing cell lines (28%) could be ascribed to poor pulp cell growth and the limited survival rate of the cells. This highlights the difficulty in establishing reliable human pulp MSC lines. No difference was observed between the different ages of individuals. However, there was a better success rate of establishing a cell line with third molar pulps than with premolars because of the larger size of the pulp specimens which allowed the preparation of several micro specimens from which cells could be grown.
The expressions of the markers CD90 and CD73 were both above 99%, but while testing for expression of CD105, we experienced some difficulty in obtaining a higher than 53% expression. CD105 is a membrane glycoprotein (endoglin), part of the Transforming Growth Factor receptor complex, and is considered as one of the MSC surface markers.15 A number of research reports have shown that its expression varies depending upon MSC source,16 culture time, the state of differentiation17 and culture medium composition.18 Mark et al.18 for example, observed a reduced expression of around 51.7% of the CD105 marker when MSC's were grown in a medium without serum. The cells, however, maintained their multilineage differentiation potential.18
The heterogeneity in CD105 expression may also be due to the presence of differentiated cells in the culture or, as was suggested by Anderson,19 may represent a distinct multipotent MSC subpopulation. Interestingly, MSC's with a low CD105 expression had improved osteogenic differentiation potential.19 This is a property one would expect from stem cells capable of tooth repair.
The presence of differentiated cells such as committed progenitor cells and fibroblasts could also lead to low CD105 expression in a stem cell culture. These cells are extensively proliferative but only in a transient manner until they differentiate terminally into cell types such as odontoblasts.
Although fibroblasts and MSC's have been well studied, the differences are not fully understood as is also the identity of MSC's in relation to fibroblasts. For example, it has been found that conventional stem cell properties such as plastic adherence and the expression of CD44, CD90 and CD105 are not specific for stem cells.2 Furthermore, the differentiation potential into osteoblasts, adipocytes, and chondrocytes, properties that are generally believed to be specific for stem cells and that can be used to discriminate MSC's from fibroblasts,15 have also been shown to be properties of fibroblasts.11
Fibroblasts differ in different parts of the body and also differ from each other in the same region. Many fibroblasts of the pulp, for example, are characterized by being relatively undifferentiated ("immature fibroblasts") and remain in a relatively undifferentiated state.20 These cells have a similar appearance to small, young fibroblasts, but are far more multipotent in terms of the cell types into which they may turn.1 "Mature" fibroblasts with a diminished capacity for transformation, may also exist that can develop into a variety of mature cell types.
Although fibroblasts share mesenchymal phenotypes with stem cells, have differentiation potential and adhere to plastic, they lack the colony-forming potential of MSC's.21 That is a feature which could be used as an additional test to distinguish between stem cells and fibroblasts.
Currently scientists are looking for markers that are more specific for MSC's and Halfon et al.12 have already identified several markers on gene and protein level that have robust differences of expression in MSC's and fibroblasts.
ACRONYMS
MSC's : adult mesenchymal stem cells
PBS : phosphate buffered saline
DMEM : Dulbecco's modified Eagle medium.
FBS : foetal bovine serum
References
1. Ten Cate AR. Oral Histology. Development, Structure and Function. 1994 Mosby, Toronto. [ Links ]
2. Kundrotas G. Surface markers distinguishing mesenchymal stem cells from fibroblasts. Acta Medica Lituanica 2012; 19: 75-9. [ Links ]
3. Nikolic Mesenchymal stem cell properties of dental pulp cells from deciduous teeth. Arch. Biol. Sei. 2011; 63: 933-42. [ Links ]
4. Bluteau G, Luder HU.De Bari C, Mitsiadis TA. Stem cells for tooth engineering. Eur Cell Mater. 2008;16: 1-9. [ Links ]
5. Miura M, Gronthos S, Zhao M, Lu B, Fisher LW . SHED: Stem cells from human exfoliated deciduous teeth. Proceedings of the National Academy of Science USA 100. 2003: 5807 -12. [ Links ]
6. Seo BM, Miura M, Gronthos S, Bertold PM, Batouli S . Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 2004;364: 149-55. [ Links ]
7. Morsczeck C, Götz W, Schierholz J, Zeilhofer F, Kuhn U. Isolation of precursor cells (PCs) from human dental follicle of wisdom teeth. Matrix Biol.2005 ;24:155-65. [ Links ]
8. Sonoyama W, Liu Y, Yamaza T, Tuan RS, Wang S. Characterization of the apical papilla and its residing stem cells from human immature permanent teeth: a pilot study. J Endod. 2008 ;34:166-71. [ Links ]
9. Gronthos S, Mankani M, Brahim J, Gehron Robey P, Shi S. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proceedings of the National Academy of Sciences of the United States of America 2000;97: 13625 - 30. [ Links ]
10. Jo YY, Lee HJ, Kook SY, Choung HW, Park JY. Isolation and characterization of postnatal stem cells from human dental tissues. Tissue Eng. 2007;13: 767-73. [ Links ]
11. Haniffa MA, Collin MP, Buckley CD, Dazzi F. Mesenchymal stem cells: the fibroblasts' new clothes? Haematologica 2009 ;94: 258-63. [ Links ]
12. Halfon S, Abramov N, Grinblat B, Ginis I. Markers distinguishing mesenchymal stem cells from fibroblasts are downregulated with passaging. Stem Cells Dev. 2011; 20: 53-66. [ Links ]
13. Lv FJ, Tuan RS, Cheung KM, Leung VY. Concise review: the surface markers and identity of human mesenchymal stem cells. Stem Cells 2014 ;32:1408-19. [ Links ]
14. van Wyk C.OIivier A, Maritz J. Cultured pulp fibroblasts: are they suitable for in vitro cytotoxicity testing? Journal of Oral Pathology and Medicine 2001; 30: 168-77. [ Links ]
15. Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F . Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006; 8: 315-7. [ Links ]
16. Kern S.Eichler H.Stoeve J.KIuter H, Bieback K. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells 2006; 24: 1294-301. [ Links ]
17. Jin HJ, Park SK, Oh W, Yang YS, Kim SW. Down-regulation of CD105 is associated with multi-lineage differentiation in human umbilical cord blood-derived mesenchymal stem cells. Biochem Biophys Res Commun. 2009 ;381: 676-81. [ Links ]
18. Mark P, Kleinsorge Μ, Gaebel R, Lux CA, Toelk A. Human mesenchymal stem cells display reduced expression of cd105 after culture in serum-free medium. Stem Cells Int. 2013; 69-76. [ Links ]
19. Anderson P.Carrillo-Galvez AB.Garcia-Perez A.Cobo M, Martin F. CD105 (endoglin)-negative murine mesenchymal stromal cells define a new multipotent subpopulation with distinct differentiation and immunomodulatory capacities. PLoS One 2013 ;8: e76979. [ Links ]
20. Hargreaves KM, Berman LH, Rotstein I, ed. Cohen's Pathways of the Pulp. Elsevier 2016. ISBN 9780323096355. [ Links ]
21. Alt E, Yan Y, Gehmert S, Song YH, Altman A. Fibroblasts share mesenchymal phenotypes with stem cells, but lack their differentiation and colony-forming potential. Biol Cell. 2011;103: 197-208. [ Links ]
 Correspondence:
Correspondence:
Dr DS Moodley
Department of Restorative Dentistry, Dental Faculty
University of the Western Cape, Cape Town
Email: dmoodley@uwc.ac.za














