Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Dental Journal
On-line version ISSN 0375-1562
Print version ISSN 0011-8516
S. Afr. dent. j. vol.73 n.3 Johannesburg Apr. 2018
CLINICAL COMMUNICATION
Clear cell-variant of epithelial-myoepithelial carcinoma of the parotid gland: the role of immunohistochemistry
ME SamarI; RE ÁvilaII; IB FonsecaIII; WJ AndersonIV; GM FonsecaV
IMaría Elena Samar: M.D., Ph.D. Department of Histology and Embryology, Faculty of Dentistry, National University of Cordoba, Cordoba, Argentina. e-mail: samarcongreso@gmail.com. Role-played and contribution: Principal researcher and writer 25%
IIRodolfo Esteban Ávila: M.D., Ph.D. Department of Cell Biology, Histology and Embryology, Faculty of Medical Sciences, National University of Cordoba, Cordoba, Argentina. e-mail: avilainfo@gmail.com. Role-played and contribution: Principal researcher and writer 25%
IIIIsmael Bernardo Fonseca: M.D., Ph.D. Department of Cell Biology, Histology and Embryology, Faculty of Medical Sciences, National University of Cordoba, Cordoba, Argentina. e-mail: isma_fonseca@hotmail.com. Role-played and contribution: Histopathological diagnosis 20%
IVWilliam J. Anderson: University of Birmingham, Birmingham, United Kingdom. Role-played and contribution: Advisor and writer 15%
VGabriel M. Fonseca: D.D.S., Ph.D. Universidad de La Frontera, Facultad de Odontología, Departamento Odontología Integral Adultos y Centro de Investigación en Odontología Legal y Forense (CIO), Temuco, Chile. e-mail: gabriel.fonseca@ufrontera.cl. Role-played and contribution: Advisor and writer 15%
SUMMARY
Epithelial-myoepithelial carcinoma (EMC) is a rare salivary gland neoplasm with an incidence of less than 1%. It tends to form biphasic ductal structures, consisting of epithelial cells lining the lumen which is surrounded by abluminal myoepithelial cells. Occasionally, EMC may show a predominance of clear myoepithelial cells. In this report involving a 64-year-old woman, we describe a case of EMC of the right parotid gland with a less obvious biphasic pattern, and importantly, a double clear cell component, showing development of clear myoepithelial cells inside and outside of the lumen. Our objective is to demonstrate the importance of immunohistochemistry in identifying this tumour when faced with the differential diagnoses of clear cell tumours.
INTRODUCTION
The salivary glands may give rise to a wide spectrum of benign and malignant neoplasms, including many rare subtypes. Collectively these represent less than 2% of all neoplasias and less than 3% of all head and neck neoplasms.1 In human cancer pathology, this group of tumours demonstrates perhaps the greatest variability when considering the surprising multiplicity of types with different histology, cytology and macroscopic structures. 1,2
Many authors attribute the complexity of these neoplasias to their myoepithelial component. The salivary gland tumours which most frequently contain myoepithelial cells in addition to other cell types include pleomorphic adenoma, adenoid cystic carcinoma and epithelial-myoepithelial carcinoma (EMC).2
EMC is a rare salivary gland tumour, which arises most commonly in the parotid gland. It has an incidence of less than 1%, the peak of which occurs in the seventh decade. It is predominantly therefore, a tumour of adults, and approximately 60% of patients are female.3,4
The histological structure of EMC tends to show multinodular growth, with tubular structures covered with a biphasic population of cells comprising luminal epithelial cells and abluminal myoepithelial cells.3 Occasionally, it shows solid growth with a predominance of clear myoepithelial cells. 4,5
Our objective is to report a case of EMC of the parotid gland with a less obvious biphasic pattern and a two-fold clear cell component, consisting of luminal and abluminal clear myoepithelial cells. In addition, we demonstrate the importance of immunohistochemistry in identifying this tumour and in distinguishing it from other clear cell tumours.
PATIENT HISTORY
A 64-year-old female presented to the hospital of one of the authors (AC), with a four-year history of a right-sided parotid swelling of approximately 3cm in size. Mild facial pain had developed over the 6 months prior to presentation, and was increased on palpation. Initial radiological investigations included ultrasound and computed tomography (CT). The patient subsequently underwent a superficial parotidectomy under general anaesthesia. Pre- and sub-auricular incisions were made to remove the gland, while also identifying and preserving the facial nerve.
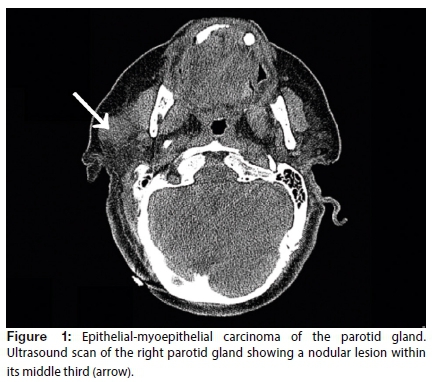
Ultrasound imaging of the right parotid gland revealed a nodular lesion within its middle third. This parenchymal lesion was well demarcated, lobulated and heterogeneous in appearance, and measured 21 x 18 x 16 mm. There was an acoustic shadow posteriorly and the lesion was poorly vascularised peripherally (Figure 1). There was no cervical lymphadenopathy and the other major salivary glands were normal in appearance.
CT revealed a space-occupying lesion in the anterior region of the right parotid gland. This was oval in appearance with well-defined but occasionally irregular borders. Its maximum diameter of 23 mm was visualised following administration of intravenous contrast. The lesion did not extend beyond the gland. The left parotid gland was mildly hypertrophic but with normal morphology. Nonspecific lymphadenopathy was observed bilaterally inferior to the parotid glands and in the submandibular region.
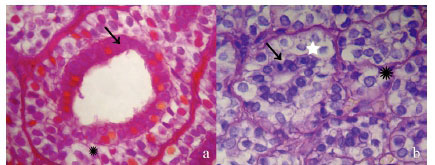
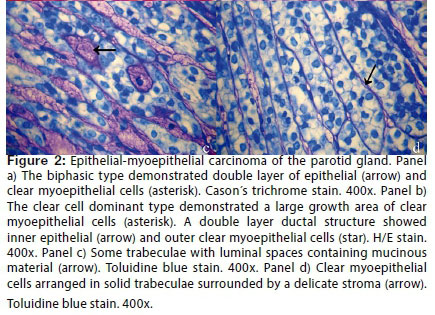
Macroscopic examination of the resected specimen shows a nodule measuring 2.5 cm in diameter. On sectioning it had an elastic consistency and pale colour with cystic areas. Histopathological examination revealed a multinodular epithelial neoplasm surrounded by a thin capsule of fibrous connective tissue, which separated it from the normal parenchyma of the parotid gland. The surgical margins were free of tumour cells. There was a mixed pattern of tumour growth consisting of both biphasic and clear cell areas, with the latter predominating (Figure 2a and Figure 2b). The biphasic pattern consisted of tubules with an internal lining of cuboidal epithelial cells and an external lining of one or more layers of large cells. The internal cuboidal epithelial cells had eosinophilic cytoplasm and round nuclei located centrally or basally, which occupied almost all of the cytoplasm. The large cells were myoepithelial in nature and had clear cytoplasm on H&E and on Cason's trichrome staining, containing Periodic acid-Schiff (PAS)-positive glycogen granules. Their nuclei were vesicular and mildly eccentric. In many areas the predominance of clear cells was well manifested. These cells were arranged in solid trabeculae surrounded by a delicate stroma. Some trabeculae had small spaces filled with PAS-positive and toluidine blue-positive mucin (Figure 2c). Nests and tubules with a double layer of clear cells were also observed which did not contain mucin (Figure 2d).
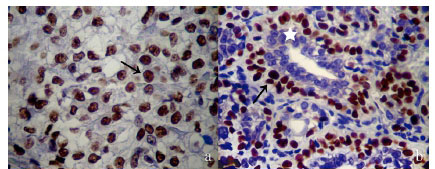
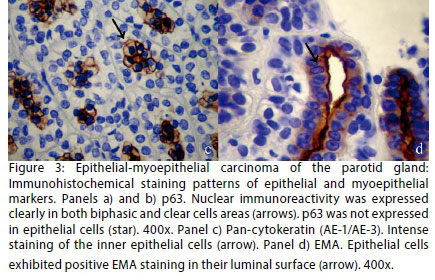
Surrounding the tubular structures and solid areas was a material which was eosinophilic, metachromatic and PAS-positive. There was no nuclear atypia and only occasional mitotic figures (1-2 mitotic figures per 10 high-powered fields). There was no neurovascular invasion. The internal layer of epithelial cell was strongly positive to pan-cytokeratin AE1/AE3 and epithelial membrane antigen (EMA) markers. Immunoreactivity to p63 in the nucleus and alpha-smooth muscle actin (α-SMA) in the cytoplasm was highly specific to the tumour's clear cells, markers which indicate myoepithelial differentiation. These findings corroborate the diagnosis of EMC (Figure 3a, Figure 3b, Figure 3c and Figure 3d).
DISCUSSION
Establishing the EMC of salivary glands as a diagnostic entity in 1991 represents another example of the terminological evolution in the classification of salivary gland neoplasms.4 Given the non-specific clinical and radiological presentation of EMC, the diagnosis of this neoplasm is established through histological and immunohistochemical studies.6 The diagnosis of EMC is based upon the identification of both epithelial and myoepithelial cell types.3
Immunohistochemistry and electron microscopy are used to confirm the diagnosis.3 The external myoepithelial layer expresses p63 and α-SMA. The internal epithelial layer is negative for these markers but positive for AE-1/AE-3 and EMA.
The relative proportions of these two cellular types vary from one tumour to another as well as between different EMC tumours.7 In this case the predominance of clear cells in many areas of the tumour gives the impression of a monocellular neoplasia, although immunohistochemistry confirms the diagnosis of EMC. Despite this, other clear cell-containing tumours (especially those which develop in the salivary glands) must also be considered before arriving at a definitive diagnosis of EMC.
Clear cell tumours of the head and neck are peculiar entities in terms of their classification.8 Understanding the differential diagnosis of these tumours is key to ensure that the subsequent treatment and prognosis are appropriate.9 Included in this differential diagnosis are: clear cell carcinoma, mucoepidermoid carcinoma, acinar cell carcinoma, oncocytoma, myoepithelioma and metastatic clear cell renal carcinoma.5
Clear cell carcinoma is an unusual low-grade neoplasia which develops almost exclusively in the minor salivary glands. The significant glycogen content of its cells confers PAS-positivity. In addition, cells show immunoreactivity to both low and high molecular weight cytokeratins, but are negative for p63 and α-SMA due to a lack of myoepithelial cells.9
Mucoepidermoid carcinoma is a malignant epithelial neoplasm with variable proportions of mucous, epidermoid, intermediate, columnar and clear cells. While the clear cells contain glycogen and are p63-negative, the epidermoid cells and mucous cells which secrete PAS-positive, Alcian blue-positive and metachromatic mucin differentiate this tumour from EMC.9,10
On the other hand, the parotid gland is the site of 83% of acinar cell carcinomas. This malignant epithelial neoplasia has predominantly serous differentiation, and in 6% of cases shows clear cells negative for PAS and p63, distinguishing it from EMC.9
Oncocytoma is a solid benign tumour which is usually well-demarcated. It consists of large epithelial cells (oncocytes) which are polygonal in shape with irregular, pyknotic nuclei and granular, eosinophilic cytoplasm due to the abundance of atypical mitochondria. The tumour cells can be stained with phosphotungstic acid-haematoxylin. Anti-mitochondrial antibodies recognise protein components of human mitochondria. The anti-mitochondrial antibody 113-1 may be used to identify tumours with oncocytes by recognising a 60 KDa glycosylated protein.9
The cells may be either clear or dark, given that the degree of eosinophilia varies between a moderate and high intensity. PAS-positive clear cells demonstrate a marked accumulation of glycogen and margination of the mitochondria. These clear cells are negative for p63 and α-SMA. Differences in cell characteristics may be used to differentiate oncocytomas with clear cells and dark cells from oxyphilic oncocytomas. In exceptional cases both cell types co-exist within the same tumour.11
Clear cell myoepithelioma also has clear cells which demonstrate PAS-positivity related to glycogen content and the same immunohistochemical characteristics as clear cells in EMC. Despite this similarity, myoepithelioma does not tend to develop ductal or tubular structures and in the presence of clear cells, it may also contain foci of plasmacytoid, hyaline or epithelioid cells.9
A study by Thiebault et al. in 199912 highlighted that the differential diagnosis of EMC should also include clear cell renal cell carcinoma. When affecting the parotid gland, metastasis of renal cell carcinoma may be difficult to distinguish from clear cell salivary gland tumours when using routine histological techniques. On the other hand, the use of cytokeratin immunomarkers may aid the diagnosis since positivity would be localised in renal metastasis yet diffuse in epithelial cells of EMC. Other characteristics of renal metastasis are its heterogeneous architecture and the presence of a prominent vascular sinusoidal network with hemosiderin deposits.9
In this case, the absence of renal pathology was corroborated by both clinical examination and complementary radiological studies.
We conclude that the immunohistochemical studies carried out confirm that our case corresponds to an EMC with two distinct tumour cell populations: epithelial and myoepithelial. In addition, the predominance of a clear myoepithelial cell component warranted a differential diagnosis considering other salivary gland primary epithelial tumours with clear cells and metastatic renal neoplasias.
Although EMC is considered a low-grade malignancy, there is a high rate of local recurrence, cervical metastases are seen in 18% of cases, and distant metastases (principally pulmonary, renal and cerebral) and death in 8%.9,13 In light of these data, the concept of EMC being a 'low grade' malignancy should be reconsidered.
Fine-needle aspiration (FNA) is a useful technique in clarifying the nature of these lesions but requires that the pathologist have an extensive knowledge of cervical pathology.14 This technique also risks sampling only one of the two cell populations present in the tumour. Despite this, it remains an excellent initial investigation with minimal side effects.
CONCLUSION
EMC is a rare salivary gland neoplasm with morphological disparities, which can cause significant diagnostic difficulties. As seen in the case presented, the strong positivity of the internal layer of epithelial cell to pan-cytokeratin AE1/AE3 and to EMA markers, as well as the immunoreactivity to markers that indicated myoepithelial differentiation, confirmed EMC. The diagnosis of EMC is usually complex, given the similarity of histological patterns with other neoplasms.
Acknowledgements
This work was supported by Resolution No. 313/2016 taken by the Bureau of Science and Technology of the National University of Cordoba, Argentina (SECyT, UNC).
Conflicts of Interest: The authors declare no conflicts of interest.
ACRONYMS
α-SMA : alpha-smooth muscle actin
CT : computed tomography
EMA : epithelial membrane antigen markers
EMC : epithelial-myoepithelial carcinoma
FNA : fine-needle aspiration
REFERENCES
1. Lawal AO, Adisa AO, Kolude B, Adeyemi BF, Olajide MA. A review of 413 salivary gland tumours in the head and neck region. J Clin Exp Dent 2013; 5: e218-22. [ Links ]
2. Redder CP, Suresh Kandagal VS, Vihute N, Ingaleshwar PS, Shetty SJ, Ahamad S. Myoepithelial cells: Current perspectives in salivary gland tumors. Clin Cancer Investig J 2013; 2: 101-5. [ Links ]
3. Samar ME, Avila RE, Fonseca IB, Ferraris RV. Sebaceous epithelial myoepithelial carcinoma of parotid: histopathological and immunohistochemical analysis. Int J Odontostomat 2010; 4: 189-96. [ Links ]
4. Batsakis JG, el-Naggar AK, Luna MA. Epithelial-myoepithelial carcinoma of salivary glands. Ann Otol Rhinol Laryngol 1992; 101: 540-2. [ Links ]
5. Hussaini HM, Angel CM, Speight PM, Firth NA, Rich AM. A double-clear variant of epithelial-myoepithelial carcinoma of the parotid gland. Head Neck Pathol 2012; 6: 471-5. [ Links ]
6. M'sakni I, Laabidi B, Brougrine F, Sabbegh-Znaïdi N, Benzarti S. Carcinome épithélial-myoépithélial de la cavité nasale. Ann Otolaryngol Chir Cervicofac 2007; 124: 228-31. [ Links ]
7. Pai RR, Sahu K, Kini AU. Clear cell predominant epithelial-myoepithelial carcinoma of the hard palate - role of immunohistochemistry. Indian J Otolaryngol Head Neck Surg 2008; 60: 163-5. [ Links ]
8. Said-Al Naief N, Klein MJ. Clear cell entities of the head and neck: a selective review of clear cell tumors of the salivary glands. Head Neck Pathol 2008; 2: 111-5. [ Links ]
9. Avila RE, Samar ME, Ferraris L, Ferraris RV, Fonseca I, Corball A et al. Non hyalinizing clear cell carcinoma of the parotid: Report of two cases with different grade of differentiation. Int J Morphol 2013; 31: 1056-61. [ Links ]
10. Avila RE, Samar ME, Fonseca I, Olmedo L, Asís OG, Ferraris R. Mucoepidermoid carcinoma of salivary glands: histological and immunohistochemical prognostic factors. In J Morphol 2011; 29: 45562. [ Links ]
11. Saqi A, Giorgadze TA, Ellazar J, Remotti F, Vazquez MF. Clear cell and eosinophilic oncocytomas of salivary gland: cytological variants or parallels? Diagn Cytopathol 2007; 35: 158-83. [ Links ]
12. Thiebault S, Mogras A, Brun I, Hamel S, Colson A, Rozan S et al. Epithelial-myoepithelial carcinoma of the salivary glands: report of a case. Ann Pathol 1999; 19: 30-2. [ Links ]
13. Friedrich RE, Donath K. Epithelial-myoepithelial carcinoma of the parotid gland with multiple metastases. A case report. J Oral Maxillofac Surg. 2000; 58:690-4. [ Links ]
14. Stewart CJ, Walker E, Mowart. FNA cytology of epithelial-myoepithelial carcinoma. Diagn Cytophatol 1999; 21: 363. [ Links ]
 Correspondence:
Correspondence:
Gabriel M. Fonseca
Universidad de La Frontera
Facultad de Odontología, Departamento Odontología Integral Adultos y Centro de Investigación en Odontología Legal y Forense (CIO).
Francisco Salazar 01145, Building L, Temuco. ZIP CODE 4811230, Chile.
Tel.: +56 (45) 2596782.
E-mail address: gabriel.fonseca@ufrontera.cl