Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Dental Journal
versão On-line ISSN 0375-1562
versão impressa ISSN 0011-8516
S. Afr. dent. j. vol.71 no.3 Johannesburg Abr. 2016
RESEARCH
An in-vitro comparison of microleakage between three calcium silicate cements and amalgam
HC SeedatI; PJ van der VyverII
IBDS (Wits), PG Dip Dent (Endo), MSc Odont (Pret), Department of Odontology, School of Dentistry, University of Pretoria, Gauteng, South Africa
IIBChD (Pret), Dip Odont (Endo), Dip Odont (Aesthetic Dentistry), MSc Odont (Pret). Department of Odontology, School of Dentistry, University of Pretoria, Gauteng, South Africa
ABSTRACT
AIM: The purpose of this in-vitro study was to compare the sealing ability of White ProRoot® MTA, MTA Plus™, BiodentineTM and Permite Amalgam when used as root-end filling materials.
MATERIALS AND METHODS: 120 single rooted, extracted teeth were endodontically treated. The apical 3 mm of each root was resected, and 3 mm deep root-end cavities were prepared.
Specimens were divided into four groups (n=30) and filled with the following materials: ProRoot® MTA, MTA Plus™, Biodentine™, and Permite Amalgam. Specimens were submerged in Indian Ink for 48 hours, and sectioned horizontally in one millimetre increments from the apical end. Dye penetration was measured using a stereomicroscope.
RESULTS: Data for different groups was summarised as percentages. Pairwise comparisons between the calcium silicate materials to amalgam were done at the 0.017 level of significance, using Fisher's exact test. Amalgam showed significantly more leakage than the calcium silicate materials (ProRoot® MTA, MTA PlusTM and Biodenti-neTM) (p<0.001). No significant differences in sealing ability were found among the calcium silicate materials.
CONCLUSION: Amalgam should be regarded as unsuitable for use as a root-end filling material. Calcium silicate cements should be recommended as the material of choice for root-end filling.
INTRODUCTION
Periapical endodontic surgery may be indicated when orthograde retreatment of failed endodontic therapy is unsuccessful, not feasible or contraindicated.1 The sequence of procedures during the surgery is: exposure of the involved apex, root-end resection, root-end cavity preparation and placement of a root-end filling.2 The root-end filling is necessary to provide a hermetic seal, preventing the egress of micro-organisms from the root canal system into the periradicular tissues.3
The ideal root-end filling material should be: non-toxic, non-carcinogenic, non-corrosive, non-staining to periapical tissues, biocompatible with host tissues, able to stimulate the regeneration of the periodontium, insoluble, dimensionally stable, unaffected by moisture, adherent to dentine, radiopaque, easy to use, and have a long shelf life.3-5 Various materials have been suggested and tested in the quest to fulfil all these ideal requirements. Amongst those proposed are: amalgam, gutta-percha, Cavit (3M ESPE, St Paul, Minnesota, USA), glass-ionomer cement, IRM (Dentsply/Maillefer, Ballaigues, Switzerland), Super EBA (Harry J. Bosworth Co., Skokie, Illinois, USA), composite resin, compomer, gold foil, Diaket (3M/ESPE, Seefeld, Germany), polycarboxylate cements, and more recently Mineral Trioxide Aggregate (MTA).3
Dental amalgam was first used as a root-end filling material by Farrar in 1884,6 and has since been the most widely used retrograde filling material, serving as a standard to which other materials are compared.7 The advantages of amalgam are that it is: inexpensive, readily available, easy to manipulate, radiopaque and insoluble in fluids.3,6 The disadvantages include: initial microleakage8, electrochemical corrosion9, induction of inflammation of adjacent periradicular tissues10, amalgam tattoo formation, the need for an undercut in cavity preparation7, zinc toxicity11, delayed expansion12, and concerns over the introduction of mercury into the periradicular tissues.3 In 1991, Friedman identified amalgam as still being the material of choice for retrograde filling.7 However, newer materials have since been developed, challenging this role of amalgam. According to Chong and Pitt Ford (2005), the use of amalgam as a root-end filling should now be confined to history.3 ProRoot® MTA (Dentsply/Maillefer, Ballaigues, Switzerland) was developed for use as a root-end filling material at Loma Linda University by Professor Mahmoud Torabinejad and colleagues in the early 1990's.13,14 The first description of MTA appeared in the scientific literature in 1993. In 1998 the United States Food and Drug Administration (FDA) approved MTA for use in endodontic treatment.13,1516 Mineral Trioxide Aggregate can be described as a hydraulic cement, for it setting is primarily dependant on hydration reactions and once set it is stable underwater.17 This is in contrast to the predominantly acid-base reactions of other dental materials.17 The main constituent of MTA is calcium silicate.18 The expiration of the patent on ProRoot® MTA has resulted in the emergence of several variants, and a generic term has been proposed to classify this category of material.17 The terms 'hydraulic silicate cements' and 'calcium silicate cements' have been proposed, with the latter being more popular.17,18
According to the patent, ProRoot® MTA is a Type 1, ordinary Portland cement.14,19 The constituents of MTA are: 55% tricalcium silicate, 20% dicalcium silicate, 10% tricalcium aluminate, 10% tetracalcium aluminoferrite and bismuth oxide (for radioopacity).20 The cement is prepared by mixing the powder with sterile water using a 3:1 powder to liquid ratio.5 Hydration of ProRoot® MTA results in the formation of calcium hydroxide and a calcium silicate hydrate gel, and this solidifies in approximately 165 minutes.5,20 Grey ProRoot® MTA and white ProRoot® MTA were introduced to the market in 1998 and 2002 respectively.21 White ProRoot® MTA has significantly less (90,8%) iron than grey ProRoot® MTA, and was intended to address the cosmetic concerns of the potential of tooth discolouration associated with grey ProRoot® MTA.13,22
The novel MTA material, MTA PlusTM (Prevest Denpro Limited, Jamu, India) has a finer particle size than ProRoot® MTA.23 The MTA PlusTM kit contains a powder and is supplied with a proprietary salt-free polymer gel and water, either one of which can be used as mixing components.24 The finer particle size improves handling and ease of placement, and the purpose of the gel is to add an anti-washout property to the material,24,25 washout being defined as the tendency of a cement to disintegrate upon early contact with blood and other fluids.26,27 As it is necessary to irrigate the osteotomy site prior to closing a periapical flap to avoid complications,25 one of the drawbacks of MTA is a tendency to washout. The final irrigation and resumption of blood flow to the surgical area may then result in a loss of some of the material placed in the root-end cavity.25 Washout-resistance is an important quality of a root-end filling, as a loss of material would compromise the apical seal.25
An X-ray diffraction analysis of the unhydrated powder of MTA PlusTM demonstrates that its components are tricalcium silicate, dicalcium silicate and bismuth oxide.28 Upon hydration of MTA PlusTM powder with the anti-washout gel or water, calcium silicate hydrate forms.28 The powder: liquid ratio for standard mixing is 3:1, however the amount of gel may be increased to modify the rheological properties (the flow characteristics) and setting time of the cement.24 MTA PlusTM when mixed with water took 65 minutes longer to set than when MTA PlusTM was mixed with the anti-washout gel (180 vs 115 minutes).28
Since January 2011, Biodentine™ a synthetic tricalcium silicate based cement has been commercially available and is marketed as a 'bioactive dentine substitute'.29,30 The production of BiodentineTM is based on 'Active Biosilicate TechnologyTM', which results in the production of pure tricalcium silicate that is free of metallic impurities.31 It has a powder component which comes in a capsule and a liquid packaged in a pipette. The powder is made up of tricalcium silicate (main core material), dicalcium silicate (second core material), iron oxide (colouring agent), zirconium oxide (radiopacifier) and two filler materials, calcium carbonate and calcium oxide.32 The liquid consists of a hydrosoluble polymer (water reducing agent) and calcium chloride (setting accelerator).31 The liquid is mixed with the powder within the capsule in a triturator for thirty seconds at a speed of 4000-4200 rotations per minute.31 The hydration reaction results in the formation of a calcium silicate hydrate gel and calcium hydroxide.33 According to Camilleri, Sorrentino and Damidot (2013), the calcium carbonate acts a nucleation site for the calcium silicate hydrate; and as a result there is a shorter induction period and an initial set within 12 minutes.32 The final setting time of Biodentine™ was found to be 45 minutes.35
Microleakage can be defined as the passage of bacteria, fluids and chemical substances between the root canal filling material and the tooth.36 The various methods used to examine the microleakage of MTA are dye penetration, fluid filtration, bacterial leakage and protein leakage.37
The purpose of this in-vitro study was to compare the sealing abilities of White ProRoot® MTA, MTA Plus™, Biodentine™ and Permite Amalgam when used as root-end filling materials.
MATERIALS AND METHODS
One hundred and twenty single-rooted extracted human teeth were collected from the out-patient dental extraction clinic of the Oral and Dental Hospital, School of Dentistry, Faculty of Health Sciences, University of Pretoria. Every aspect of this research project was conducted in line with the ethical and safety standards for handling human tissues and conducting laboratory research, as prescribed by South African law: the Health Profession Act 56 of 1974 (South African National Health Bill, 2003). The study was approved by the Research Ethics Committee, Faculty of Health Sciences, University of Pretoria, under protocol number 138/2011.
The teeth were stored immediately after extraction in phosphate buffered saline (PBS) (Sigma-Aldrich, Johannesburg, South Africa) at room temperature. The experiments were conducted two weeks later. The prerequisites for the sample selection were similar to those used by Pichardo et al. (2006)38:
1. Root formation should be complete.
2. There should be a single, straight root canal
3. No root canal therapy should have been previously performed on the teeth.
4. The teeth should not have any fractures.
5. There should be no root caries or root resorption.
All the above-mentioned properties were verified radio-graphically except for the absence of fractures (point 4), which was done using a surgical operating microscope (D.F. Vasconcellos, Säo Paulo, Brazil).
The crowns of all teeth were sectioned with a flat-ended, cylindrical diamond bur (Komet, Lemgo, Germany) driven in a high speed hand piece (W&H, Bürmoos, Austria). The bur was held perpendicular to the long axis of the tooth and the cut made to render a standard root measurement of 18mm, as suggested by Pichardo et al. 2006.38
All the root canals were prepared to within 0.5mm of the apical foramen with ProTaper Universal (Dentsply/Maillefer, Ballaigues, Switzerland) rotary instruments using the X-Smart Plus rotary motor (Dentsply/Maillefer, Ballaigues, Switzerland), according to the manufacturer's instructions. A size 15 k-file (Dentsply/Maillefer, Ballaigues, Switzerland) was used to establish patency of the root canals. Glyde Root Canal Conditioner (Dentsply/Maillefer, Ballaigues, Switzerland) was used as a lubricant on the files prior to insertion into the canals. A 6% sodium hypochlorite solution (Nordiska Dental, Angelholm, Sweden) was used for irrigation between rotary instrumentation. The canals were prepared with ProTaper Universal rotary files (Dentsply/ Maillefer, Ballaigues, Switzerland), up to a size F3 instrument. A final irrigation of the canals was performed with 17% EDTA (Topclear, Dental Discounts, Johannesburg, South Africa). The canals were dried with F3 ProTaper Paper Points (Dentsply/Maillefer, Ballaigues, Switzerland). All the prepared root canals were obturated by applying the continuous wave, warm vertical condensation technique using the Calamus Dual Obturation Unit (Dentsply/Maillefer, Ballaigues, Switzerland). The coronal access cavities were sealed with Fuji IX glass-ionomer restorative material (GC Corporation, Tokyo, Japan). The specimens were then stored in a single sterile plastic container filled with PBS solution at room temperature for 48 hours.
The apical 3mms of all the specimens were resected perpendicular to the long axis of the tooth using a straight carbide fissure bur (Komet, Lemgo, Germany) on a high speed hand-piece. Root-end cavities were prepared on all specimens to a depth of 3mm using an ultrasonic ProUltra Surgical Tip (Dentsply/Maillefer, Ballaigues, Switzerland) on an NSK Ultrasonic Scaler Unit (NSK, Nakanishi, Japan). The ultrasonic tip was used in a brushing motion with light pressure, and a cylindrical cavity was created parallel to the long axis of the root.
The entire surface of every specimen, except for the resected apical portion, was coated with two layers of clear nail varnish to seal all other possible portals of communication with the root canal.
The 120 specimens were randomly divided into four groups (n=30) and treated as follows:
Group 1: White ProRoot® MTA (Dentsply/Maillefer, Ballaigues, switzerland) (n=30)
The material was hand-mixed according to the manufacturer's instructions and placed into the root-end cavity preparation, using the MAP system (micro apical placement) (Dentsply/Maillefer, Ballaigues, Switzerland).
Group 2: MTA plus™ (prevest Denpro Ltd., Jamu, India) (n=30)
The material was hand mixed according to the manufacturer's instructions and placed into the root-end cavity preparation, using the MAP system (Dentsply/Maillefer, Ballaigues, Switzerland).
Group 3: Biodentine™ (septodont Ltd., saint Maur des Fausse's, France) (n=30)
The material was mixed in an amalgamator (TPC Advanced Technology, California, USA) according to the manufacturer's instructions and placed into the root-end cavity preparation, using the MAP System (Dentsply/ Maillefer, Ballaigues, Switzerland).
Group 4: Amalgam (Permite, SDI, Victoria, Australia) (n=30)
The material was mixed in an amalgamator (TPC Advanced Technology, California, USA) according to the manufacturer's instructions and placed into the root-end cavity preparation, using a retrograde amalgam carrier (Medesy, Pordenone, Italy).
Each group of specimens was wrapped in gauze that had been moistened with PBS, and stored in separate, sealed, sterile plastic containers. The specimens were then placed in an incubator (Binder, Tuttlingen, Germany), and stored for 24 hours at 37°C to allow the materials to set completely.
The specimen groups were then immersed in Indian Ink dye (Winsor and Newton, London, England), and remained immersed in their individual, sealed, sterile plastic containers for 48 hours at 37°C. The specimens were then removed from the dye reservoir, and excess dye was rinsed off with distilled water for 15 minutes. The apical end of each specimen was sectioned transversely in 1mm increments with a wafering blade in an IsometTM low speed saw (Buehler, Lake Bluff, Illinois, USA) under continuous water irrigation. This rendered three 1mm slices of each root-end which were then packaged in labelled, sealed packets, identifying each sample as being: 1mm, 2mm or 3mm from the apex.
The sections were then mounted on microscopic glass slides, and examined under a stereomicroscope (Carl Zeiss, Jena, Germany) by two independent, blinded and calibrated examiners. The extent of dye penetration was measured to the nearest millimetre based on the presence of visible dye between the root-end filling and dentinal wall interface.
Data was summarized in terms of percentage for the outcome vector (no leak; 1mm leak; 2mm leak; 3mm leak). Furthermore, pairwise comparisons between each of the new generation filling materials and amalgam were done at the 0.017 level of significance using Fisher's exact test. The latter test could also be used at the 0.05 level of significance to assess the four filling materials in a single analysis.
RESULTS
The measurement of the depth of dye penetration between the root-end filling material and the dentinal wall interface was of note.
Group 1: Dye penetration of White ProRoot® MTA specimens
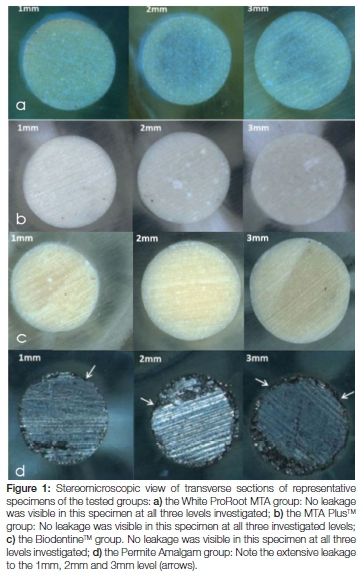
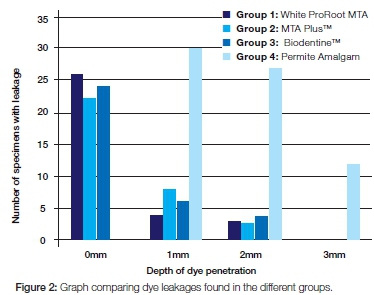
In this group, 26 specimens showed no leakage, four specimens leaked to a depth of 1mm and only three specimens leaked to a depth of 2mm (Figure 1a).
Group 2: Dye penetration of MTA PlusTM specimens
Twenty two of the specimens in this group showed no leakage, eight specimens leaked to a depth of 1mm, and three specimens leaked to a depth of 2mm (Figure 2a).
Group 3: Dye penetration of Biodentine™ specimens
No leakage was observed in 24 specimens of this group, six specimens leaked to a depth of 1mm, and four specimens leaked to a depth of 2mm (Figure 3a).
Group 4: Dye penetration of Permite Amalgam specimens
All 30 of the specimens in this group showed leakage to a depth of 1mm, 27 specimens leaked to a depth of 2mm, and 12 specimens leaked to a depth of 3mm (Figure 4a).
The results of all the specimens from the different groups combined before statistical analysis are illustrated by Figure 2. The specimens from the Permite Amalgam group showed the most leakage while the White ProRoot® MTA group showed the least amount of leakage.
Statistical analysis
The outcomes of the failure vector are reported for each treatment group in Figure 3. Amalgam displayed significantly greater leakage than any of the calcium silicate root-end filling materials (ProRoot® MTA, MTA PlusTM and BiodentineTM).
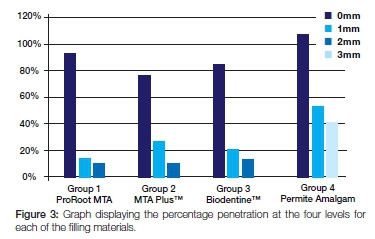
The differences are very clear if the leakage outcome is simplified as present or absent. Then it can be noted that leakage of the amalgam samples was 100% (30/30), and that 20% (6/30) of BiodentineTM samples, 13.3% (4/30) of ProRoot® MTA samples while 27.6% (8/30) of MTA PlusTM samples leaked. It is also important to note that none of the calcium silicate retrograde-filled teeth demonstrated leakage up to 3mm. In contrast, 40% (12/30) of the amalgam-filled teeth showed leakage up to the 3mm level.
Furthermore, no significant differences were found among the calcium silicate cements, namely BiodentineTM vs. ProRoot® MTA (p = 0.776), BiodentineTM vs. MTA PlusTM (p=0.667), and ProRoot® MTA vs. MTA PlusTM (p = 0.350).
DISCUSSION
The purpose of this in-vitro study was to compare the sealing ability of root-end fillings. White ProRoot® MTA, MTA PlusTM, BiodentineTM and Permite Amalgam were compared by measuring the linear dye penetration of Indian ink dye at the interface between the root-end filling and the dentinal wall. The achievement of a hermetic seal by a root-end filling is a critical factor that impacts on the long-term success of endodontic surgery.37
Microleakage is an estimate of the quality of the seal, and can be measured by allowing a tracer to penetrate a filled root-end cavity.39 Commonly used tracers include dyes, radioisotopes, bacteria and bacterial by-products.39 The dye-immersion technique was introduced by Grossman in 1939, and is widely used because it is easy to perform, reproducible, safe, inexpensive, quantifiable and dyes are readily available.39,40 This technique is a passive method that depends on the phenomenon of capillarity, whereby the dye penetrates any space between the root-end filling and the canal wall.39,41 According to Torabinejad et al. (1994), a filling material able to resist the penetration of small molecules such as dye, would have the potential to resist the penetration of larger bacteria and their by-products.42 It is important to use a tracer that mimics the clinical situation.40 Indian ink dye was used in the present study as it is unlikely that bacterial leakage can occur in root canal spaces where this dye is unable to penetrate.43 Chong et al. (1995) compared the penetration of tracers and other assessment methods of the efficacy of the sealing of root-end fillings. The findings of their experiments showed that bacterial penetration and Indian ink dye penetration yielded similar results.44
In the present study, White ProRoot® MTA, MTA Plus™ and BiodentineTM showed significantly better sealing ability when compared with Permite Amalgam. There were no statistically significant differences in sealing ability between the three calcium silicate cements.
Amalgam has been considered as the material of choice for root-end filling for over a century, despite the many disadvantages of its use.7 Most materials that have been developed or considered for root-end filling have been compared with amalgam.48 The use of a cavity varnish in conjunction with amalgam has been shown to improve sealing ability.8,45 Vertucci and Beatty (1986), however, found no significant difference in apical dye penetration, whether cavity varnish was used or not.46 In the present study, no cavity varnish was used in order to obtain an accurate appraisal of the sealing ability of the amalgam alone. An additional factor that improves the sealing ability of amalgam is the formation of corrosion products between the material itself and the dentine wall over a period of time.46 Tronstad et al. (1983) found that corrosion products start to occupy the gap between amalgam and dentine within seven days.8 The generation of corrosion products is however undesirable, especially in the periradicular region.9 In the present study, a high copper non-Gamma 2 Amalgam was used because it undergoes minimal corrosion.47
ProRoot® MTA was found to have a significantly superior sealing ability to amalgam in the present study. The application of MTA in endodontics was first investigated in 199315 by Torab-inejad, Watson and Pitt Ford (1993) who measured the penetration of Rhodamine B dye in longitudinally sectioned teeth by confocal microscopy. The results showed that MTA leaked significantly less than both amalgam and Super EBA (Harry J. Bosworth Co., Skokie, Illinois, USA).48 Methylene Blue dye penetration in longitudinally sectioned samples was not significantly different when the root-end fillings of amalgam, Super EBA, IRM and MTA were tested, either with or without blood contamination.42 Furthermore, MTA was found to have significantly less dye penetration in the presence or absence of blood than amalgam, Super EBA or IRM.42
Mineral Trioxide Aggregate was found to have a superior seal to amalgam and Super EBA by Aqrabawi (2000), when the penetration of 1% methylene blue dye was measured on longitudinally sectioned samples.49 Davis et al. (2003), measured the linear penetration of Indian ink dye in teeth that had been decalcified, rendering them transparent.50 Mineral Trioxide Aggregate, Super EBA and amalgam (Tytin, Kerr Corporation, Michigan, USA) were used by Davis et al (2003) as root-end fillings after the root-end cavities were irrigated with saline, citric acid or doxycycline. It was found that amalgam leaked significantly more than MTA and Super EBA, irrespective of the irrigant used, but there was no statistically significant difference in leakage between MTA and Super EBA.50 Pereira, Cenci and Demarco (2004) evaluated the microleakage allowed by amalgam (GS-80, Southern Dental Industries, Victoria, Australia), MTA, Super EBA and Vitremer (3M ESPE, St.Paul, Minnesota, USA) by measuring linear dye penetration in transversely sectioned apical slices.51 They found that MTA leaked significantly less than Vitremer and Super EBA, and amalgam leaked significantly more than all the other tested materials.51 The present study concurs with other dye penetration studies in the finding that amalgam displays significantly more micro-leakage than ProRoot® MTA.37
BiodentineTM and MTA PlusTM are relatively new calcium silicate materials. The current study is the first to investigate the sealing ability of MTA PlusTM as a root end filling. Kokate and Pawar (2012), compared the efficacy of the marginal seals of BiodentineTM, MTA and glass ionomer cement in root-end fillings, by examining the penetration of 1% Methylene blue in longitudinally sectioned samples with stereomicroscopy.52 It was found that BiodentineTM displayed significantly less leakage than MTA and glassionomer cement, with glass-ionomer cement exhibiting the most microleakage.52 Ravichandra et al. (2014) examined the marginal adaptation of BiodentineTM, MTA and glass-ionomer cement as root-end fillings by measuring the area stained by Rhodamine blue dye in transverse sections using confocal laser scanning microscopy.53
The study revealed that BiodentineTM showed the lowest marginal gaps and the best marginal adaptation, followed by MTA. The largest marginal gaps were found with glassionomer cement. The results of the present study differ from those of Kokate and Parwar (2012)52 and Ravichandra et al. (2014),53 for no significant differences were found between the sealing abilities of BiodentineTM and MTA. A possible reason for the different outcome could be related to the sample size of the previous studies. Both used a sample size of 10 specimens per group while in the present study, 30 specimens were used per group. Other possible differences may be attributed to the fact that different types of dyes were used. Furthermore, Kokate and Pawar (2012) sectioned their specimens longitudinally while in the present study, specimens were sectioned transversely.52 Another possible factor is that Ravichandra et al. (2014) used confocal laser scanning microscopy to assess the specimens while in the present study stereomicroscopy was used.53
The phenomenon of hydroxyapatite formation over MTA, MTA PlusTM and BiodentineTM when immersed in simulated body fluids such as PBS is well documented.24,34 In the present study the specimens were stored in PBS-mois-tened gauze after placement of the retrograde fillings. A suggested hypothesis is that the bioactivity of the calcium silicate cements during storage possibly improved their sealing ability due to chemical bonding of hydroxya-patite crystals to the radicular dentine.24 Amalgam does not bond to dentine, and is reliant for its retention on the preparation of an undercut in the root-end cavity.7
In the clinical scenario, MTA PlusTM and BiodentineTM may have certain advantages over ProRoot® MTA. MTA PlusTM mixed with anti-washout gel would prevent the loss of the material from the root-end cavity when the surgical site is rinsed, or when blood flow resumes. This would probably improve the sealing ability of MTA PlusTM, as more filling material would be retained within the root-end cavity. Due to the addition of the setting accelerator, calcium chloride, Bioden-tineTM reaches an initial set within 12 minutes. This allows the operator to visually observe that the root-end filling material has set prior to closure of the surgical site. This would not be possible with MTA PlusTM or ProRoot® MTA due to the significantly longer setting times of these materials. Further studies are indicated to verify the advantages that MTA PlusTM and BiodentineTM may have over ProRoot® MTA, with regard to sealing ability when used in-vivo.
CONCLUSIONS
Within the limitations of the present study the following can be concluded:
1. Calcium silicate cements showed a significantly better sealing ability than Permite Amalgam when used as root-end filling material (p<0.001).
2. There was no significant differences in the sealing abilities of White ProRoot® MTA, MTA Plus and Biodentine™ (p<0.001).
3. Based on the findings of the present study, amalgam should be regarded as unsuitable for use as a root-end filling material.
4. Based on the findings of the present study, calcium silicate cements should be recommended as the material of choice for root-end filling.
ACRONYMS
FDA: Food and Drug Administration
PBS: phosphate buffered saline
MAP: micro apical placement
MTA: Mineral Trioxide Aggregate
References
1. Chong BS, Rhodes JS. Endodontic surgery. Br Dent J. 2014; 216:281-90. [ Links ]
2. Rubinstein R, Torabinejad M. Contemporary endodontic surgery. J Calif Dent Assoc. 2004; 32:485-92. [ Links ]
3. Chong BS, Pitt Ford TR. Root-end filling materials: rationale and tissue response. Endod Topics. 2005; 11:114-30. [ Links ]
4. Gartner AH, Dorn SO. Advances in endodontic surgery. Dent Clin North Am. 1992; 36:357-78. [ Links ]
5. Parirokh M, Torabinejad M. Mineral trioxide aggregate: a comprehensive literature review-Part I: chemical, physical, and antibacterial properties. J Endod. 2010; 36:16-27. [ Links ]
6. Priyanka SR. A literature review of root-end filling materials. IOSR-JDMS. 2013; 9:20-5. [ Links ]
7. Friedman S. Retrograde approaches in endodontic therapy. Endod Dent Traumatol. 1991; 7:97-107. [ Links ]
8. Tronstad L, Trope M, Doering A, Hasselgren G. Sealing ability of dental amalgams as retrograde fillings in endodontic therapy. J Endod. 1983; 9:551-3. [ Links ]
9. Hohenfeldt PR, Aurelio JA, Gerstein H. Electrochemical corrosion in the failure of apical amalgam. Report of two cases. Oral Surg Oral Med Oral Pathol. 1985; 60:658-60. [ Links ]
10. Baek SH, Plenk H, Kim S. Periapical tissue responses and cementum regeneration with amalgam, SuperEBA, and MTA as root-end filling materials. J Endod. 2005; 31:444-9. [ Links ]
11. Kaga M, Seale NS, Hanawa T, Ferracane JL, Okabe T. Cytotoxicity of amalgams. J Dent Res. 1988; 67:1221-4. [ Links ]
12. Yamada T, Fusayama T. Effect of moisture contamination on high-copper amalgam. J Dent Res. 1981; 60:716-23. [ Links ]
13. Roberts HW, Toth JM, Berzins DW, Charlton DG. Mineral trioxide aggregate material use in endodontic treatment: a review of the literature. Dent Mater. 2008; 24:149-64. [ Links ]
14. Camilleri J, Pitt Ford TR. Mineral trioxide aggregate: a review of the constituents and biological properties of the material. Int Endod J. 2006; 39:747-54. [ Links ]
15. Lee SJ, Monsef M, Torabinejad M. Sealing ability of a mineral trioxide aggregate for repair of lateral root perforations. J Endod. 1993; 19:541-4. [ Links ]
16. Schwartz RS, Mauger M, Clement DJ, Walker WA. Mineral trioxide aggregate: a new material for endodontics. J Am Dent Assoc. 1999; 130:967-75. [ Links ]
17. Darvell BW, Wu RC. "MTA"-an Hydraulic Silicate Cement: review, update and setting reaction. Dent Mater. 2011; 27:407-22. [ Links ]
18. Camilleri J. Hydration characteristics of calcium silicate cements with alternative radiopacifiers used as root-end filling materials. J Endod. 2010; 36:502-8. [ Links ]
19. Torabinejad M, White DJ. Tooth filling material and method of use: US Patent No. 5769638. 1998. [ Links ]
20. Camilleri J. Hydration mechanisms of mineral trioxide aggregate. Int Endod J. 2007; 40:462-70. [ Links ]
21. Bozeman TB, Lemon RR, Eleazer PD. Elemental analysis of crystal precipitate from Gray and White MTA. J Endod. 2006; 32:425-8. [ Links ]
22. Asgary S, Parirokh M, Eghbal MJ, Stowe S, Brink F. A qualitative X-ray analysis of white and grey mineral trioxide aggregate using compositional imaging. J Mater Sci Mater Med. 2006; 17:187-91. [ Links ]
23. Camilleri J, Formosa L, Damidot D. The setting characteristics of MTA Plus in different environmental conditions. Int Endod J. 2013; 46:831-40. [ Links ]
24. Gandolfi MG, Siboni F, Primus CM, Prati C. Ion release, porosity, solubility, and bioactivity of MTA Plus Tricalcium Silicate. J Endod. 2014; 40: 1632-7. [ Links ]
25. Formosa LM, Mallia B, Camilleri J. A quantitative method for determining the antiwashout characteristics of cement-based dental materials including mineral trioxide aggregate. Int Endod J. 2013; 46:179-86. [ Links ]
26. Bortoluzzi EA, Broon NJ, Bramante CM, Garcia RB, de Moraes IG, Bernardineli N. Sealing ability of MTA and radiopaque Portland cement with or without calcium chloride for root-end filling. J Endod. 2006; 32:897-900. [ Links ]
27. Khayat KH. Effects of antiwashout admixtures on fresh concrete properties. ACI Mater J. 1995; 92:164-71. [ Links ]
28. Formosa LM, Mallia B, Camilleri J. Mineral trioxide aggregate with anti-washout gel - properties and microstructure. Dent Mater. 2013; 29:294-306. [ Links ]
29. Pawar AM, Kokate SR, Shah RA. Management of a large periapical lesion using Biodentine as retrograde restoration with eighteen months evident follow up. J Conserv. Dent. 2013; 16:573-5. [ Links ]
30. Rajasekharan S, Martens LC, Cauwels RG, Verbeeck RM. Biodentine material characteristics and clinical applications: a review of the literature. Eur Arch Paediatr Dent. 2014; 15:147-58. [ Links ]
31. Septodont. Biodentine™ Package insert [Online]. Available from: http://www.septodont.ca/sites/default/files/Biodentine%20IFU_0.pdf [Accessed October 2014]. [ Links ]
32. Priyalakshmi S, Ranjan M. Review on Biodentine-A bioactive dentin substitute IOSR-JDMS. 2014; 13:13-7. [ Links ]
33. Camilleri J, Kralj P, Veber M, Sinagra E. Characterization and analyses of acid-extractable and leached trace elements in dental cements. Int Endod J. 2012; 45:737-43. [ Links ]
34. Camilleri J, Sorrentino F, Damidot D. Investigation of the hydration and bioactivity of radiopacified tricalcium silicate cement, Biodentine and MTA Angelus. Dent Mater. 2013; 29:580-93. [ Links ]
35. Grech L, Mallia B, Camilleri J. Investigation of the physical properties of tricalcium silicate cement-based root-end filling materials. Dent Mater. 2013; 29:e20-8. [ Links ]
36. Timpawat S, Vongsavan N, Messer HH. Effect of removal of the smear layer on apical microleakage. J Endod. 2001; 27:351-3. [ Links ]
37. Torabinejad M, Parirokh M. Mineral trioxide aggregate: a comprehensive literature review-part II: leakage and biocompatibility investigations. J Endod. 2010; 36:190-202. [ Links ]
38. Pichardo MR, George SW, Bergeron BE, Jeansonne BG, Rutledge R. Apical leakage of root-end placed SuperEBA, MTA, and Geristore restorations in human teeth previously stored in 10% formalin. J Endod. 2006; 32:956-9. [ Links ]
39. Verissimo DM, do Vale MS. Methodologies for assessment of apical and coronal leakage of endodontic filling materials: a critical review. J Oral Sc. 2006; 48:93-8. [ Links ]
40. Youngson CC, Jones JC, Manogue M, Smith IS. In vitro dentinal penetration by tracers used in microleakage studies. Int Endod J. 1998; 31:90-9. [ Links ]
41. Camps J, Pashley D. Reliability of the dye penetration studies. J Endod. 2003; 29:592-4. [ Links ]
42. Torabinejad M, Higa RK, McKendry DJ, Pitt Ford TR. Dye leakage of four root end filling materials: effects of blood contamination. J Endod. 1994; 20:159-63. [ Links ]
43. Schafer E, Olthoff G. Effect of three different sealers on the sealing ability of both thermafil obturators and cold laterally compacted Gutta-Percha. J Endod. 2002; 28:638-42. [ Links ]
44. Chong BS, Pitt Ford TR, Watson TF, Wilson RF. Sealing ability of potential retrograde root filling materials. Endod Dent Traumatol. 1995; 11:264-9. [ Links ]
45. Abdal AK, Retief DH, Jamison HC. The apical seal via the retrosurgical approach. II. An evaluation of retrofilling materials. Oral Surg Oral Med Oral Pathol. 1982; 54:213-8. [ Links ]
46. Vertucci FJ, Beatty RG. Apical leakage associated with retrofilling techniques: a dye study. J Endod. 1986; 12:331-6. [ Links ]
47. SDI. Pre-dosed amalgam capsules Permite, Lojic+ & GS-80 Instructions For Use [Online]. Available from: http://www.sdi.com.au/images/stories/instructions/instructions_pdf/amalgam _P_L_G/in_amalgam_p_l_g_en.pdf [Accessed October 2014]. [ Links ]
48. Torabinejad M, Watson TF, Pitt Ford TR. Sealing ability of a mineral trioxide aggregate when used as a root end filling material. J Endod. 1993; 19:591-5. [ Links ]
49. Aqrabawi J. Sealing ability of amalgam, super EBA cement, and MTA when used as retrograde filling materials. Br Dent J. 2000; 188:266-8. [ Links ]
50. Davis JL, Jeansonne BG, Davenport WD, Gardiner D. The effect of irrigation with doxycycline or citric acid on leakage and osseous wound healing. J Endod. 2003; 29:31-5. [ Links ]
51. Pereira CL, Cenci MS, Demarco FF. Sealing ability of MTA, Super EBA, Vitremer and amalgam as root-end filling materials. Braz Oral Res. 2004; 18:317-21. [ Links ]
52. Kokate SR, Pawar AM. An in vitro comparative stereomicroscopic evaluation of marginal seal between MTA, glass ionomer cement & Biodentine as root end filling materials using 1% methylene blue as tracer. Endodontology. 2012; 24:36-42. [ Links ]
53. Ravichandra PV, Vemisetty H, Deepthi K, Reddy SJ, Ramakiran D, Krishna MJN, et al. Comparative evaluation of marginal adaptation of Biodentine™ and other commonly used root end filling materials - an in vitro study. JCDR. 2014; 8:243-5. [ Links ]
 Correspondence:
Correspondence:
Hussein C Seedat
Department of Odontology, School of Dentistry
University of Pretoria
Gauteng, South Africa
Cell: 083 564 7860
E-mail: drhseedat@gmail.com













