Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Dental Journal
On-line version ISSN 0375-1562
Print version ISSN 0011-8516
S. Afr. dent. j. vol.71 n.1 Johannesburg Feb. 2016
RESEARCH
Antifungal susceptibility of Candida albicans isolated from the oral cavities of patients with HIV infection and cancer
FJ OwotadeI; Z GulubeII; S RamlaIII; M PatelIV
IBChD, FWACS, PhD. Department of Oral and Maxillofacial Surgery, College of Health Sciences, Obafemi Awolowo University, Ile-Ife, Nigeria and Faculty of health sciences, University of The witwatersrand
IIBSc, MSc. division of oral microbiology, Department of Oral Biological Sciences, School of Oral Health sciences, Faculty of health sciences, University of the witwatersrand, Johannesburg, South Africa
IIIBDS, MSc Dent. division of oral microbiology, department of oral Biological sciences, School of oral health sciences, Faculty of health sciences, University of the witwatersrand, Johannesburg, South Africa
IVDip Med Tech, BSc, BSc (Hons), MSc, PhD. division of oral microbiology, department of oral Biological sciences, School of oral health sciences, Faculty of health sciences, University of the witwatersrand, Johannesburg, South Africa
ABSTRACT
This study investigated the antifungal susceptibility of Candida albicans isolated from the oral cavities of 205 HIV positive patients, 49 cancer patients and 20 normal healthy individuals. C. albicans were isolated and the antifungal susceptibility was determined. The results were analysed using the clinical break points and epidemiological cut off values. Prevalence of C. albicans carriage in HIV, in cancer patients, and in healthy individuals was 73%, 45% and 43% respectively. Resistance of the fungus to anidulafungin (0.49%), caspofungin (0.97%), posaconazole (3.4%), voriconazole (0.97%), itraconazole (0.97%), fluconazole (1.94%), amphotericin B (0%) was found to be low. For posaconazole the number of resistant strains and the non-wild type (3.4%) were the same. However for the rest of the antifungal drugs, the number of non-wild type was found to be higher than the resistance determined by clinical break points. Multi-azole resistance was also noted in some patients. In conclusion, there is a low rate of antifungal drug resistance among C. albicans isolated from the oral cavities of immunocompromised patients in Johannesburg, South Africa. However, the high number of non-wild type strains suggests that there is a need for an ongoing surveillance.
INTRODUCTION
Candida albicans is a commensal of the human oral cavity, gut and vagina. This yeast causes infections in immunocompromised individuals including HIV and cancer patients. Oral candidiasis is the most common infection in these patients. It is frequently treated with antifungal agents such as amphotericin B, nystatin and fluconazole. In addition, prophylactic antifungals, particularly fluconazole, are prescribed to many patients undergoing cancer therapy especially in advanced cases and in patients with HIV infection (Epstein, 1996, Greenspan, 1994).1,2
Prior to the introduction of HAART, recurrent oral candidia-sis was a problem in HIV positive patients, and still occurs, although to a lesser extent, in the era of HAART therapy. The development of antifungal resistance has been related to the use of antifungal agents to treat recurrent infections in patients with HIV where the appropriate doses, prescribed for the usual duration, become ineffective.3Candida species have exhibited a very high level of variability in the pattern of sensitivity to antifungal agents.4 In Africa, routine antifungal susceptibility testing is not undertaken because oral thrush is treated with empirical antifungal agents. Consequently, the data on the antifungal sensitivity of Candida species from South Africa is still very sparse. A large study on the antifun-gal sensitivity profile of oral isolates was published more than 10 years ago, before fluconazole became widely available.5 More recently, antifungal testing has become standardized globally through the Clinical Laboratory Standards Institute (CLSI) with the objective of detecting sensitivity patterns and discovering the development of resistant strains which is a growing problem.2 It is therefore important to characterize the pattern of antifungal susceptibility of Candida species in the era of moderate availability of antifungal agents. This knowledge serves as a guide for antifungal therapy and can also help to predict the outcomes of therapeutic interventions. The present study investigated the antifungal sensitivity profile of C. albicans isolated from the oral cavities of HIV positive patients, cancer patients on chemotherapy or radiotherapy and normal healthy individuals.
MATERIALS AND METHODS
Study population and identification of C. albicans
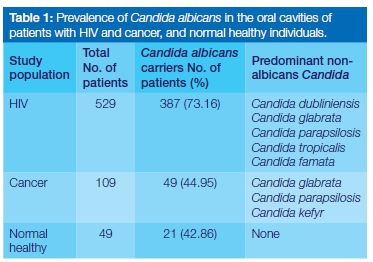
Ethics clearance was obtained from the Committee for Research on Human Subjects (Medical), University of the Witwatersrand, Johannesburg, Gauteng. One hundred and nine patients, diagnosed with mainly head and neck cancers, who were scheduled for either radiation or chemotherapy and were attending clinics at the Department of Oncology, together with 529 HIV positive patients who were attending the HIV clinic in Charlotte Maxeke Johannesburg Academic Hospital, were asked to volunteer for the study. Forty nine normal healthy individuals without any signs of oral candidiasis were also included in the study since Candida is a commensal in oral cavities and candidiasis does sometimes develop. All these patients were also the subjects of three other studies.
The procedure was explained to participants and written consent was obtained. Data such as the presence of active infection, other risk factors and previous exposure to antifungal agents were not available due to incomplete patient records and a lack of verification by the patients. Heterogeneity of the study population and the incomplete patient data are limitations of this study.
An oral rinse with 10ml of sterile distilled water was used to collect samples in sterile sputum jars. 100 μl of the rinse sample was inoculated onto CHROMagar®Candida plates (CHROMagar Microbiology) and incubated at 37°C for 48 hours. CHROMagar is a chromogenic agar that supports the growth of many Candida species and even enables the identification of some species on the basis of the colours of the colonies. For example, C. albicans presents green colonies, C. tropicalis produces steel blue colonies and C. krusei, purple colonies. All the different colour colonies were selected, subcultured on Sabouraud dextrose agar for 48 hours for purity and identified using API 20 C AUX system® (bioMérieux). This is a standard substrate assimilation test that can accurately identify Candida species. These cultures were stored in microbank vials at -70°C for further studies.
Determination of antifungal susceptibility
Financial constraints limited testing for antifungal susceptibility to only 205, 49 and 20 strains of C. albicans which had been isolated from, respectively, HIV patients, cancer patients, and normal healthy individuals. The tests were performed using Sensititre YeastOne® microdilution colorometric microtitre plates. The procedure is based on the Broth Microdilution minimum inhibitory concentration system described by Clinical Laboratory Standards Institute document M27-A2.6 The microtitre plates contained twofold dilutions of anidulafungin, micafungin, caspofungin, 5-flucytosine, posaconazole, voriconazole, itraconazole, fluconazole, amphotericin B. Also contained was AlamarBlue®, a proven cell viability indicator that uses the natural reducing power of living cells to convert resazurin to the fluorescent molecule, resorufin, which produces very bright red fluorescence. Fresh yeast cultures (five randomly selected colonies) were suspended in normal saline and adjusted to a turbidity of 0.5 McFarland standards. 20μl of the inoculum was added to the Sensititre YeastOne® broth and gently vortexed. 100 μl of the inoculated broth was added to each well of the Sensititre YeastOne® panel using a multichannel pipette and incubated at 35°C for 24 hours. Wells with growth were red in colour. The results were read and the minimal inhibitory concentration (MIC) was determined as the lowest concentration that prevented a growth (the first blue well). Control strains of C. parapsilosis ATCC 22019 and C. krusei ATCC 6258 were included as recommended by the CLSI guidelines. The results were interpreted as clinical break point (CBP) and epidemiological cutoff values (ECV). Clinical break point represents clinical isolates that are likely to respond to treatment with a given antimicrobial agent administered using the approved dosing regimen for that agent. They are relevant to clinicians. By contrast, epidemiological cutoff values represent the most sensitive measure of the emergence of strains with decreased susceptibility to a given agent. This means that ECV results will detect development of resistance ahead of clinically relevant resistance, which would present as a treatment failure due to drug resistance. The re-established clinical break points and epidemiological cutoff values of the CLSI and the EUCAST for anidulafungin, micafungin, caspofungin, voriconazole, itraconazole and fluconazole as described by Pfaller and Diekema (2012),4 and for amphotericin B and posaconazole as described by Arendrup et al. (2011)7 and Lass-Flörl et al. (2011)8 were used to characterize the sensitivity profiles of the C. albicans isolates.
RESULTS
Prevalence of C. albicans
The carriage rate of C. albicans was 73.16%, 44.95% and 42.86% in patients with HIV, with cancer, and in normal healthy individuals, respectively (Table 1). Patients with HIV and cancer carried a variety of Candida species other than C. albicans in their oral cavities.
Antifungal susceptibility of Candida albicans
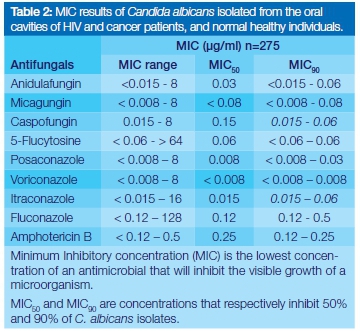
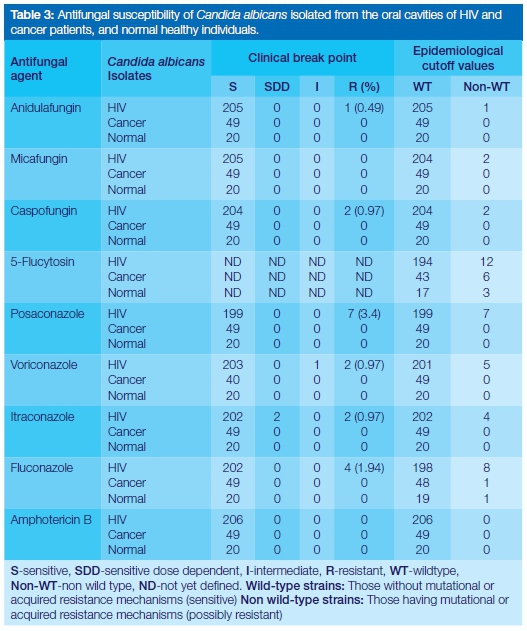
The range of MIC values (MIC obtained by all the strains), MIC50 (MIC obtained by 50% of the strains) and MIC90 (MIC obtained by 90% of the strains) obtained for the test antifungal agents are shown in Table 2. The interpretation of these MIC results were obtained using previously described standards.4,7,8 The characterized sensitivity profile of the C. albicans isolates are given in Table 3.
DISCUSSION
Candida albicans is the most frequently isolated Candida species from the oral cavities of HIV patients (73%) and cancer patients (45%) which is similar to elsewhere.9,10 Although HAART has reduced the carrier rate of Candida species in these patients, the proportions of C. albicans and non-albicans Candida have not dramatically changed.9 Therefore the antifungal agents are still used to target C. albicans and the sensitivity profile of C. albicans has been most commonly described. However, due to the overuse and frequent use of antifungal drugs, resistance to these drugs has increased.11 Although fluconazole was freely available in sub-Saharan Africa, results in this study show that the resistance to this drug is very low i.e. 1.94% (Table 2 and 3) because it is still a prescription drug in South Africa. This resistance has slightly increased since a zero resistance was reported in 2002.5 In Tanzania, where 250 isolates of C. albicans were tested, there was no resistance to fluconazole whereas 4% of isolates were resistant to itraconazole and 0.54% azole resistance has been reported from Tunisia.12,13 More recently, 50% resistance of C. albicans to fluconazole has been reported in southern parts of South Africa.14 Antifungal drug resistance varies in the several African countries. A study in Nigeria in 201115 and recently from Ethiopia16 reported 16% resistance to fluconazole in C. albicans whereas a resistance among isolates from Cameroon was as high as 50%.14
Less than 1% of strains that were tested in this study were resistant to the echinocandins i.e. anidulafungin, micafungin, and caspofungin (Table 3). This is consistent with other reports that ranged from of 1% to 5%.16-18 The reason may be that these drugs have only recently become available in Africa.
Seven isolates (3.4%) of C. albicans were found to be resistant to posaconazole which is unexpected (Table 2). It is the most recently approved triazole with a broad spectrum activity against Candida, Aspergillus, Cryptococcus and many other fungi and is usually reserved for the treatment and prophylaxis of invasive forms of fungal infections. The breakpoints for posaconazole are very close to one another and they are not well described (S < 0.06 and R >0.06). These values are based on pharmacokinetic data epide-miological cut-off values and clinical experience and therefore they are reviewed regularly.7 However, in our study for posaconazole the number of resistant strains and the non-wild type (3.4%) were identical, showing good correlation between the two methods of analysis. This also means that these non-wild type isolates may have acquired or innate mutational resistance mechanism and therefore may or may not show a clinical response to the antifungal therapy. In addition, during surveillance these epidemiological cut-off values are useful in detecting the emergence of potential resistance. This phenomenon has been recorded by Pfaller et al. (2011) whose study recorded that the epidemiological cutoff values for posaconazole increased three times from 2001 to 2009 (1.1 to 3.2%).19
Although a previous South African study reported that 8.4% of C. albicans were resistant to amphotericin B,5 no resistance was detected in the present study. Furthermore, it is known that patients previously exposed to fluconazole for a long time period harbour more fluconazole-resistant C. albicans than patients who are fluconazole-naïve.10,20 Unfortunately it was not possible to collect data on the history of previous antifungal exposure in all the HIV patients.
However, that history was available for the cancer patients and the normal individuals. No antifungal drug resistance was found in the C. albicans isolates from the oral cavities of cancer patients or individuals in the healthy individuals. They had not been exposed to antifungal drugs prior to the respective collection of samples. In addition there was no prophylactic prescription of antifungal agents for patients undergoing either radiation therapy or chemotherapy in the Oncology Departments in this study.
Four fluconazole resistant strains of C. albicans were isolated from HIV positive patients. Two isolates were resistant to all four azole drugs whereas the remaining two isolates were either intermediate or dose dependent on two azole drugs but were resistant to fluconazole. Prophylactic itraconazole is known to induce resistance to fluconazole.21 In addition, the use of ketoconazole and miconazole reduces the susceptibility to fluconazole.22,23 Multiple-resistance seen in our study may have been due to cross resistance among azole drugs.
Strains categorized as resistant according to the clinical break points, are associated with a high likelihood of therapeutic failure. Non-wild type strains have acquired or innate mutational resistance mechanisms and therefore these strains may or may not show a clinical response to the anti-fungal therapy. In our results all the antifungal drugs except for posaconazole showed high epidemiological cut-off values compared with the clinical break points. This means that in our study population drug resistance is developing which will require regular studies or continuous surveillance.
These results have shown that resistance of C. albicans to commonly used antifungal agents is low. However, C. albicans resistant to one azole is likely to be resistant to other azole drugs. Therefore, patients who do not respond to an azole drug should be given drugs other than azole. Furthermore, pathological material should be sent for a microbiological analysis including culturing for Candida. Presence of multiple Candida in these patients suggests a need for further research and data on antifungal susceptibility in these yeasts.
In conclusion, the rate of antifungal drug resistance among C. albicans isolated from the oral cavities of immunocompromised patients is low in Johannesburg, South Africa. Multiple azole resistance was noted in HIV positive patients. Therefore, alternative antifungal drugs should be considered in non-responsive patients. In addition, high ECV suggested the likelihood of development of drug resistance and therefore ongoing surveillance is needed.
Acknowledgements: This work was supported by the Tertiary Education Tax Fund, the Federal Republic of Nigeria.
Competing interest: None declared
ACRONYMS
ECV: Epidemiological Cutoff Values
CBP: Clinical Break Point
CLSI: Clinical Laboratory Standards Institute
EUCAST: European Committee on Antimicrobial Susceptibility Testing
MIC: Minimal Inhibitory Concentration
References
1. Epstein JB, Ransier A, Lunn R, etal. Prophylaxis of candidiasis in patients with leukemia and bone marrow transplants. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1996; 81(3): 291-6. [ Links ]
2. Greenspan D. Treatment of oropharyngeal candidiasis in HIV-positive patients. J Am Acad Dermatol 1994; 31(3 pt 2): S51-S55. [ Links ]
3. Pfaller MA. Antifungal drug resistance: mechanisms, epidemiology, and consequences for treatment. Am J Med 2012; 125 (1 Suppl): S3-S13. [ Links ]
4. Pfaller MA, Diekema DJ. Progress in antifungal susceptibility testing of Candida spp. by use of clinical and laboratory standards institute broth microdilution methods, 2010-2012. J Clin Microbiol 2012; 50 (9): 2846-56. [ Links ]
5. Blignaut E, Messer S, Hollis RJ, Pfaller MA. Antifungal susceptibility of South African oral yeast isolates from HIV/AIDS patients and healthy individuals. Diagn Microbiol Infect Dis 2002; 44 (2): 169-74. [ Links ]
6. Clinical Laboratory Standards Institute M27-A2. Reference method for broth dilution antifungal susceptibility testing of yeasts, Vol 22, No 15. 2002 [ Links ]
7. Arendrup MC, Cuenca-Estrella M, Donnelly JP, et al. EUCAST technical notes on Posaconazole. Clin Microbiol Infect 2011; 17 (11): E16-E17. [ Links ]
8. Lass-Flörl C, Arendrup MC, Rodriguez-Tudela J-L, et al. EU-CAST technical notes on Amphotericin B. Clin Microbiol Infect 2011; 17 (12): E27-E29. [ Links ]
9. Owotade FJ, Patel M, Ralephenya TRMD, Vergotine G. Oral Candida colonization in HIV positive women: associated factors and changes with antiretroviral therapy. J Med Microbiol 2013; 62 (Pt 1): 126-32. [ Links ]
10. Redding SW, Zellars RC, Kirkpatrick WR, et al. Epidemiology of oropharyngeal Candida colonization and infection in patients receiving radiation for head and neck cancer. J Clin Microbiol 1999; 37(12): 3896-900. [ Links ]
11. Perlin, DS. Antifungal drug resistance in developing countries. In: Sosa AJ, Byarugaba DK, Amabile-Cuevas CF, Hsueh PR, Kariuki S, Okeke IN, eds. Springer New York Dordrecht Heidelberg London; 2010, p. 137-56. [ Links ]
12. Hamza OJM, Matee MIN, Moshi MJ, et al. Species distribution and in vitro antifungal susceptibility of oral yeast isolates from Tanzanian HIV-infected patients with primary and recurrent oropharyngeal candidiasis. BMC Microbiol 2008; 8:135. Doi:10.1186/1471-2180-8-135. [ Links ]
13. Eddouzi J, Lohberger A, Vogne C, Manai M, Sanglard D. Identification and antifungal susceptibility of a large collection of yeast strains isolated in Tunisian hospitals. Med Mycol 2013; 51(7): 737-46. [ Links ]
14. Abrantes PMDS, McArthur CP, Africa CWJ. Multi-drug resistant (MDR) oral Candida species isolated from HIV-positive patients in South Africa and Cameroon. Diagn Microbiol Infect Dis 2014; 79 (2): 222-7. [ Links ]
15. Nweze EI, Ogbonnaya UL. Oral Candida isolates among HIV-infected subjects in Nigeria. J Microbiol Immunol Infect 2011; 44(3): 172-7. [ Links ]
16. Mulu A, Kassu A, Anagaw B, et al. Frequent detection of 'azole' resistant Candida species among late presenting AIDS patients in northwest Ethiopia. BMC Infect Dis 2013; 13:82. Doi: 10.1186/1471-2334-13-82. [ Links ]
17. Garcia-Agudo L, Garcia-Martos P, Martos-Canadas J, Aznar-Marin P, Marin-Casanova P, Rodriguez-Iglesias M. Evaluation of the Sensititre Yeast One microdilution method for susceptibility testing of Candida species to anidulafungin, caspofungin, and micafungin. Rev Esp Quimioter 2012; 25(4): 256-60. [ Links ]
18. Badiee P, Alborzi A, Davarpanah MA, Shakiba E. Distributions and antifungal susceptibility of Candida species from mucosal sites in HIV positive patients. Arch Iran Med 2010; 13(4): 282-7. [ Links ]
19. Pfaller MA, Boyken L, Hollis RJ, et al. Wild-type MIC distributions and epidemiological cutoff values for posaconazole and voriconazole and Candida spp. as determined by 24-hour CLSI broth microdilution. J Clin Microbiol 2011; 49 (2): 630-7. [ Links ]
20. Hunter KD, Gibson J, Lockhart P, Pithie A, Bagg J. Fluconazole resistant Candida species in the oral flora of fluconazole exposed HIV-positive patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1998; 85 (5): 558-64. [ Links ]
21. Goldman M, Cloud GA, Smedema M, et al. Does long-term itraconazole prophylaxis result in in vitro azole resistance in mucosal Candida albicans isolated from persons with advanced human immunodeficiency virus infection? The National Institute of Allergy and Infectious Diseases Mycoses Study Group. Antimicrob Agents chemother 2000; 44 (6): 1585-7. [ Links ]
22. Pelletier R, Peter J, Antin C, Gonzalez C, Wood L, Walsh TJ. Emergence of resistance of Candida albicans to clotrimazole in human immunodeficiency virus-infected children: in vitro and clinical correlations. J Clin Microbiol 2000; 38 (4): 1563-8. [ Links ]
23. Rautemaa R, Richardson M, Pfaller M, Perheentupa J, Saxen H. Reduction of fluconazole susceptibility of Candida albicans in APECED patients due to long-term use of ketoconazole and miconazole. Scand J Infect Dis 2008; 40 (11-12): 904-7. [ Links ]
 Correspondence:
Correspondence:
Mrudula Patel
School of oral health sciences, Faculty of health sciences, University of the witwatersrand
Private Bag 3, wits, Johannesburg, 2050, Gauteng
South Africa
Tel: 27 11 717 2110 , Fax: 27 1 1 717 2027/086 553 3020
E-mail: mrudula.patel@wits.ac.za













