Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Dental Journal
On-line version ISSN 0375-1562
Print version ISSN 0011-8516
S. Afr. dent. j. vol.70 n.10 Johannesburg Nov. 2015
RESEARCH
Platelet- Rich Fibrin (PRF) - The effect of storage time on platelet concentration
MT PeckI; D HissII; L StephenI; A SattiIII; A MajeedIII
IDepartment of Oral Medicine and Periodontology, Faculty of Dentistry, University of the Western Cape
IIDepartment of Medical Biosciences, Faculty of Dentistry, University of the Western Cape
IIIDepartment of Prosthodontics, Faculty of Dentistry, University of the Western Cape
ABSTRACT
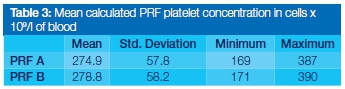
The aim of this study was to determine whether storage time had a significant effect on the platelet concentration of platelet-rich fibrin (PRF). Three blood samples were drawn from each participant into a sterile blood sampling tube. Two of the blood samples were centrifuged to form PRF. The third non-centrifuged sample was used to measure the baseline blood platelet concentration. After PRF had formed, it was removed from the respective test tubes at different time intervals i.e. immediately after centrifugation (Group A) and after 60 min of storage time in the blood collecting tube (Group B). The residual blood from each group was tested for platelet concentration and compared with the baseline reading (as an indirect measure of the platelet concentrate of PRF). The PRF produced in Group A (PRF A) had a mean platelet concentration of 274 ±57.8x109/L, whereas the PRF of Group B (PRF B) was 278 ± 58.2x109/L. A statistically significant difference was seen between the groups (p < 0.001).
CONCLUSIONS: Storage time has a significant effect on the platelet concentration of PRF. Further research is required to determine whether this has any clinical relevance.
INTRODUCTION
Wound healing is a complex process characterised by the repair and reconstitution of lost or damaged tissue. Identification of the pathological and biochemical mechanisms that regulate tissue repair and homeostasis has long been regarded as central to their therapeutic exploitation in the clinical setting. By the mid-1990s, several methods were proposed to enhance wound healing, including the administration of high concentrations of human platelets to affected areas.1-6 It was assumed that platelets optimised wound healing by promoting the secretion of growth factors (GFs) necessary for tissue repair.7-9 The most common platelet concentrate used in these procedures is platelet-rich-plasma (PRP) and by the early part of the 21st century, its use in various surgical procedures was commonplace.1,4,10,11 However, the preparation of PRP often requires the use of specialised equipment, chemicals and animal-derived additives. This increases the risk for complications secondary to allergic reactions to certain animal-derived additives.12-14 As a result, researchers have sought more efficient and safer methods of concentrating platelets for surgical use.1 This led to the production of platelet-rich-fibrin (PRF), a platelet concentrate that neither contained additives nor required the use of specialised equipment during its preparation. First introduced by Choukroun et al (2001), PRF has been studied extensively and is now regarded as a biological scaffold different to PRP.15 However, even though the use of PRF is gaining widespread clinical acceptance, several questions regarding its biological stability remain unanswered.16,17 The aim of this this study was to analyse the effect of storage time on the platelet concentration of this unique biomaterial.
MATERIALS AND METHODS
The study was conducted under the principles outlined by the "World Medical Association Declaration of Helsinki, Ethical Principles for Medical Research Involving Human Subjects" of 2008. Ethical approval was obtained from the Ethics Committee of the University of the Western Cape (Registration number 11/4/29). All participants were fully informed of the research protocol and were required to sign a declaration of informed consent before being allowed to participate in this cohort analytical study. A total of 30 healthy participants (16 females and 14 males) were enrolled into the study. Participants were drawn from the current patient pool as well as staff members based at the Dental Faculty, University of the Western Cape. All participants had three separate blood samples collected by venipuncture. Two of the samples were acquired using tubes containing clot activators, i.e., Vacuette® 10ml serum tubes with Z Serum Clot Activator (Greiner BioOne International AG, Germany), whereas the remaining blood sample was drawn into a 10ml BD Vacutainer® tube that contained dipotassium-EDTA, an anticoagulant (BD Diagnostics, New Jersey USA).
All the blood samples collected in the anticoagulant-containing tubes were used to measure baseline platelet concentrations, whereas the remaining blood samples (collected in the Clot Activator containing tubes) were used to prepare PRF by centrifugation (400g for 12 minutes) in a standard benchtop centrifuge (PLC-03, Hicare International, Taiwan) (Figure 1). Therefore, from each study participant, two samples of PRF were obtained (Figure 2). The two PRF samples were then randomly allocated into either Group A or Group B, using a simple coin toss.

For Group A, the PRF was removed from the tube immediately after preparation (0min), whereas for Group B, the PRF remained in the tube and was only removed after 60 minutes. The PRF produced from each group was designated PRF A and PRF B respectively. Because a direct measurement of platelet concentration of PRF is not yet possible, we calculated the PRF concentration for each group indirectly, by determining the numerical difference between the residual platelet concentration (of the remaining serum after removal of PRF) and the baseline platelet concentration for each specific study participant. In this study, platelet concentration analysis was carried out using an electronic automated cell counter (Advia 2120, Siemens AG, Erglangen, Germany).
Data was collected and entered into a spreadsheet (Microsoft Office 2010 Excel, Microsoft Corporation, Washington). The results were compared and analysed statistically using SPSS® Version 13 for Windows.
RESULTS AND DISCUSSION
A total of 30 participants (16 females and 14 males) were entered into the study. The mean age of the participants was 41.7 years, with male subjects being slightly younger than their female counterparts (41.3 years for males vs 42 years for females). The youngest participant was 24 years old and the oldest, 58 years old.
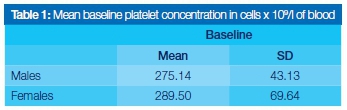
Platelet concentrations obtained from analysis of all 30 participants were within the normal laboratory reference range of 170-400x109L of circulating blood. None of the participants displayed any significant haematological disease. The mean blood platelet concentration was 282.8x109/L (Table 1). There was no significant difference between the genders.
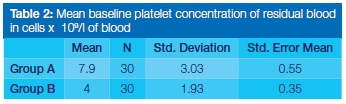
After removal of formed PRF from Group A, the residual serum yielded minimal concentrations of platelets. The mean concentration of remaining platelets was 7.9x109/L (Table 2). For Group B, serum platelet concentrations were also minimal after removal of the prepared PRF with the mean concentration of the platelets being 4.0x109/L (Table 2).
The residual mean platelet concentration of Group A was higher than that determined for Group B with the mean difference in platelet concentrates between the two groups being 3.90 x 109/L. Using a non-parametric Signed Rank Test, the statistical significance of the differences in platelet concentrates between the Groups A and B was analysed. The difference between the two groups was statistically significant (p < 0.001).
The platelet concentration of PRF for both groups was calculated using the difference between baseline and residual platelet concentrations (Table 3). A paired t-test showed a statistically significant difference between the two groups (p < 0.001) (Table 4).
DISCUSSION
The purpose of this study was to determine whether storage time had any significant effect on the platelet concentration of PRF. Because the structural properties of the PRF clot does not allow for a direct measurement of its platelet concentration, we used the residual platelet values left in the blood tube after removal of the PRF clot as an indirect measurement of the platelet concentration in the PRF clot. This is in accordance with a previously published method.18 Higher residual values indicate that less platelets were contained in the removed clot whereas lower residual values would indicate that more platelets were transferred to the clot. The results from this study indicate that there is a significant difference in the mean platelet concentration of PRF when stored for 0 and 60 minutes.
From the platelet counts obtained in both test groups it was clear that a significant proportion of platelets had been removed from the blood after extraction of the PRF clots. In fact, when differences between the test groups and the baseline blood sample were compared, it was evident that 97%-98% of the platelets were concentrated in the PRF clot (Table 3). This observation is similar to results published for previous studies (97%).19 The natural process of fibrin and clot formation that occurs in the blood collecting tubes results in the entrapment of the majority of the available platelets in a fibrin matrix. This acts as a reservoir for a concentration of growth factors (GFs) required in the initial stages of wound healing.20
Although the physiology of PRF has been studied extensively, very few reports adequately document the ability of this platelet concentrate to be stored.21 Previous research has indicated that storing PRF under certain conditions may affect its ability to clinically yield positive results.22 In fact, it is not recommended to store the platelet concentrate in its blood collecting tube since it is assumed that it may disintegrate into an unusable form after about 15 minutes of storage.22 Instead, several authors propose storing the biomaterial in a metal dish or a proprietary designed storage box.17 Data regarding the maximal storage time and ideal storage temperature of PRF are, however, largely lacking.
In this study, we used standard blood collecting tubes with clot activators to store PRF for at least 60 minutes at room temperature. This particular duration of time was chosen based on the average time of typical periodontal surgical procedures at the Faculty of Dentistry, University of the Western Cape. The results of the study indicate that by using the presented protocol for platelet concentrate preparation, it was possible to concentrate more than 97% of the available blood platelets into a readily usable form. This study also showed that by using the blood collecting tubes as a storage medium, there was no detrimental effect on the platelet concentration of PRF. Indeed, storing the concentrate for 60 minutes resulted in a form of PRF that had significantly higher concentrations of platelets compared with non-stored PRF. The reason for this phenomenon is unclear, but may be related to the "clot activators" that line these tubes. Clot activators are often silica based and are used in plastic tubes to mimic the clotting effect of glass based blood tubes.22 As a result, a longer contact time between blood and these activators may enhance fibrin activation and clot formation, with subsequent platelet entrapment.18
Another reason for the variation in platelet concentration seen in this study may be that the recommended centrifuge time is too short to allow for complete clot formation to occur. Therefore, allowing the PRF clot to remain in the tube for a period longer than the recommended time, may result in a more complete physiological reaction taking place.
The ability of PRF to sustain its platelet concentrate over the tested time may have significant clinical implica-tions.8,9,22-24 Rather than having to draw blood during the surgical procedure, it allows for blood to be drawn before the start of the procedure, thereby improving patient comfort and saving operator time. The option to store the PRF in the same tubes in which it was formed negates exposure to other environmental factors that may contaminate the sample. It is also cost-effective, since no specialised equipment or storage facilities are required. This may be significant in resource-poor settings.
PRF has been recognised as a biomaterial that includes living cells.18,19 In order to sustain cell viability over time, an isotonic solution is required for storage. The blood collecting tubes, although not designed to store blood, act as containers for the PRF and the remaining formed elements and serum. Consequently, when PRF is stored in the blood that it was derived from, the remaining serum acts as a natural isotonic solution that sustains cell survival.
The release of growth factors is a significant property of blood platelets.24 Previous studies indicate that several of these factors play an essential role in osteogenesis and periodontal regeneration.20,25 When PRF is used, the release of these growth factors appears to be constant, and over a longer period of time when compared with that seen with PRP.23 In a direct comparison between the two, PRP was shown to have an initial larger release of growth factors after activation. However, these high concentrations were not stable and decreased over time. On the other hand, PRF releases less growth factors initially, but sustains this release for a longer duration.23 A number of authors speculate that this may be due to the fibrin clot that forms a network and acts as a reservoir for the trapped platelets.18,19,26 In the present study, it was shown that the platelet concentration of PRF improved over a period of 60 minutes of storage. We assume that this may be due to prolonged fibrin clot maturation. It is therefore reasonable to speculate that if PRF is stored until optimal fibrin formation is achieved, then higher concentrations of growth factors may be available from the PRF during wound healing. Whether this has any clinical significance requires further investigation.
Temperature may affect the storage potential of PRF and it has been suggested that storing it in near-freezing temperatures is not advisable. In this study, all the samples were stored at room temperature. It was clear that storage under these conditions had no detrimental effect on the platelet concentration of the PRF clot.
Although the present study showed statistically significant differences between the two groups tested, some limitations were also evident. These include the limited number of study participants as well as the inability to directly measure the platelet concentration of PRF. Other factors include the small difference between the platelet concentrations of the groups tested. Although statistically significant, a mean difference of only 3.9 x 109 cells/L may not be clinically relevant and therefore further research is warranted to determine the clinical significance of these findings.
CONCLUSIONS
The viability of storing PRF in its own blood collecting tubes has not yet been reported. This study showed that by using the above method of platelet preparation, it is possible to concentrate more than 97% of the available platelets into a PRF clot. The study also indicated that, unlike in previous reports, clot disintegration does not take place after 15 minutes of storage, but rather remains stable over a period of at least 60 minutes and may in fact increase in platelet concentration during that time. Further investigations are warranted to determine whether this has any clinical implications.
Disclosure policy: The authors declare no conflict of interest regarding the publication of this paper. This paper forms part of the requirements of partial fulfilment towards the degree PhD.
ACRONYMS
GF: Growth factors
PRF: Platelet-rich fibrin
PRP: Platelet -rich plasma
References
1. Carlson,N E,Roach.RB Jr. Platelet-rich plasma: clinical applications in Dentistry. Journal of the American Dental Association 2002; 133 (10); 1383. [ Links ]
2. Jameson,C. Autologous platelet concentrate for the production of platelet gel. Laboratory Medicine 2007; 38 (1); 39-42. [ Links ]
3. Lacci,K M, Dardik, A. Platelet-rich plasma: support for its use in wound healing. The Yale Journal of Biology and Medicine 2010; 83 (1); 1. [ Links ]
4. Marx,RE, Carlson ER, Eichstaedt RM, et al. Platelet-rich plasma: Growth factor enhancement for bone grafts. Journal Oral Maxillofacial Surgery 1998; 85; 638-46. [ Links ]
5. Oz,M., Jeevanandam V, Smith CR, et al. Autologous fibrin glue from intra-operatively collected platelet-rich plasma. Annals of Thoracic Surgery 1992; 53; 530-1. [ Links ]
6. Tözüm,TF,Demiralp,B. Platelet-rich plasma: a promising innovation in Dentistry. Journal Canadian Dental Association 2003.69; (10); 664. [ Links ]
7. Blair,P, Flaumenhaft,R. Platelet [alpha]-granules: Basic biology and clinical correlates. Blood Reviews 2009; 23 (4); 177-89. [ Links ]
8. Hill,AG, Hood,AG, Reeder,GD, et al. Perioperative autologous sequestration II: A differential centrifugation technique for autologous component therapy: Methods and results. Proceedings of the American Academy of Cardiovascular Perfusion 1993; 14; 122-5. [ Links ]
9. Hood,AG, Hill AG, Reeder,GD, et al. Perioperative autologous sequestration III: A new physiologic glue with wound healing properties. Proceedings of the American Academy of Cardiovascular Perfusion 1993; 14; 126-9. [ Links ]
10. Plachkova,AS, NikolidakisD, Mulder,J, et al. Effect of platelet-rich plasma on bone regeneration in Dentistry: a systematic review. Clinical Oral Implant Research 2008; 19; 539-45. [ Links ]
11. Whitman,DH, Berry,RL, Green,DM. Platelet gel: an autologous alternative to fibrin glue with applications in oral and maxillofacial surgery. Journal of Oral and Maxillofacial Surgery 1997; 55 (11); 1294-9. [ Links ]
12. Wei,Y, Tsui,V, Peng,Z, et al. Anaphylaxis from topical bovine thrombin (Thrombostat) during haemodialysis and evaluation of sensitization among a dialysis population. Clinical Experimental Allergy 2003; 33; 1730-4. [ Links ]
13. Clark, J A, Humphries, JE, Crean, S, et al. Topical bovine thrombin: a 21 -year review of topical bovine thrombin spontaneous case safety reports submitted to FDA's Adverse Event Reporting System. Pharmaco-epidemiology and Drug Safety 2010; February; 19 (2); 107-14. [ Links ]
14. Ofosu, FA, Crean,S, Reynolds,MW. A safety review of topical bovine thrombin-induced generation of antibodies to bovine proteins. Clinical Therapeutics 2009; 31 (4); 679-91. [ Links ]
15. Choukroun J, Adda F, Schoeffler C, Vervelle A. Une opportu-nité en paro-implantologie: Le PRF. Implantodontie 2001; 42; 55-62. [ Links ]
16. Wang, HL, Avila, G. Platelet rich plasma: myth or reality. European Journal of Dentistry. 2007; 1; 192-4. [ Links ]
17. Dohan Ehrenfest, DM. How to optimize the preparation of leukocyte- and platelet-rich fibrin (L-PRF, Choukroun's technique) clots and membranes: Introducing the PRF Box. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodon-tology 2010; 101 (6); 275-8. [ Links ]
18. Dohan Ehrenfest, D M, Del Corso, M, Diss, A, et al. Three-dimensional architecture and cell composition of a Choukroun's platelet-rich fibrin clot and membrane. Journal of Periodontology. 2010; 81 (4); 546-55. [ Links ]
19. Dohan,DM, Choukroun,J, Diss,A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part II: platelet-related biologic features. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology. 2006; 101 (3); 45-50. [ Links ]
20. Gassling,V, Douglas,T, Warnke,PH, et al. Platelet-rich fibrin membranes as scaffolds for periosteal tissue engineering. Clinical Oral Implant Research. 2010; 21 (5); 543-9. [ Links ]
21. Dohan Ehrenfest,DM, Del Corso,M, Inchingolo,F, et al. Selecting a relevant in vitro cell model for testing and comparing the effects of a Choukroun's platelet-rich fibrin (PRF) membrane and a platelet-rich plasma (PRP) gel: tricks and traps. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology 2010; 110 (4); 409-11. [ Links ]
22. Choukroun,J, Diss,A, Simonpieri,A, et al. Platelet-rich fibrin (PRF): a second-generation platelet concentrate. Part IV: Clinical effects on tissue healing. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology 2006; 101 (3); 56-60. [ Links ]
23. He,L, Lin,Y, Hu,X, et al. A comparative study of platelet-rich fibrin (PRF) and platelet-rich plasma (PRP) on the effect of proliferation and differentiation of rat osteoblasts in vitro. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology 2009; 108 (5); 707-13. [ Links ]
24. Kang,YH, Jeon,SH, Park,JY, et al. Platelet-rich fibrin (PRF) is a bio-scaffold and reservoir of growth factors for tissue regeneration. Tissue Engineering 2011;17 (3-4); 349-59. [ Links ]
25. Del Corso,M, Sammartino,G, Dohan Ehrenfest,DM. Re: Clinical evaluation of a modified coronally advanced flap alone or in combination with a platelet-rich fibrin membrane for the treatment of adjacent multiple gingival recessions: a 6-month study. Journal of Periodontology 2009; 80 (11); 1694-7; author reply 1697-9. [ Links ]
26. Toffler,M, Toscano,N, Holtzclaw,D, et al. Introducing Choukrouns Platelet Rich Fibrin (PRF) to the reconstructive surgical milieu. Journal of Implant and Advanced Clinical Dentistry 2009; 1; (6): 21-31. [ Links ]
 Correspondence:
Correspondence:
MT Peck
Department of Oral Medicine & Periodontology, Faculty of Dentistry, University of the Western Cape
Private Bag X1, Tygerberg, 7505, South Africa
Tel: 021 9373186. Fax: 086 6187560
E-mail: mpeck@uwc.ac.za













