Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Dental Journal
versão On-line ISSN 0375-1562
versão impressa ISSN 0011-8516
S. Afr. dent. j. vol.70 no.6 Johannesburg Jul. 2015
CASE BOOK
Oral medicine case book 71: Amyloidosis of the tongue
BK BunnI; C SchnetlerII; AW van ZylIII; WFP van HeerdenIV
IBDS, MDent, FC Path(SA)OralPath. Department of Oral Pathology and Oral Biology, School of Dentistry, Faculty of Health Sciences, University of Pretoria
IIBChD. Department of Periodontics and Oral Medicine, School of Dentistry, Faculty of Health Sciences, University of Pretoria
IIIBChD, MChD. Department of Periodontics and Oral Medicine, School of Dentistry, Faculty of Health Sciences, University of Pretoria
IVBChD, MChD, FC Path(SA)Oral Path, PhD, DSc. Department of Oral Pathology and Oral Biology, School of Dentistry, Faculty of Health Sciences, University of Pretoria
CASE REPORT
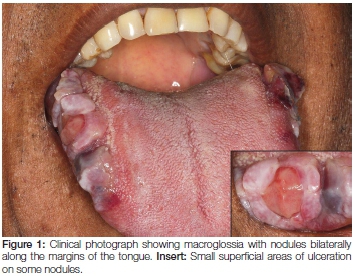
A 57-year-old male patient presented to the Oral Medicine Clinic with a one-month history of painful macroglossia associated with difficulty in speaking and swallowing. The patient complained of recent weight loss and chronic fatigue. Intra-oral examination showed the tongue to be enlarged with conspicuous nodules on the lateral borders (Figure 1). It had a multinodular, firm consistency on palpation. An incisional biopsy from the right lateral area of the tongue was submitted for histology.
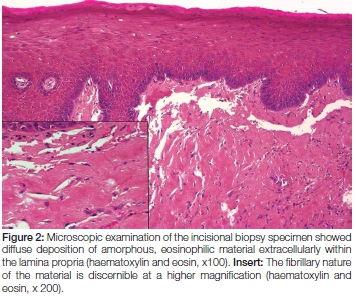
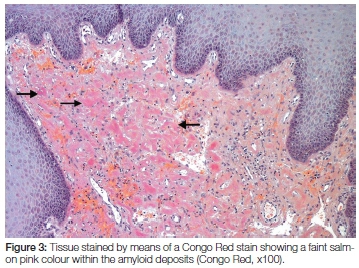
Microscopic examination of the biopsy specimen showed a portion of soft tissue surfaced by focally ulcerated stratified squamous epithelium. The ulcer bed comprised inflamed granulation tissue with a superficial fibrinopurulent surface membrane. Within the lamina propria and extending to the deep aspect of the biopsy specimen was evidence of an amorphous eosinophilic proteinaceous material deposited extracellularly. Scattered between the deposits were occasional small blood vessels and a patchy chronic inflammatory cell infiltrate (Figure 2). The proteinaceous material stained a light salmon pink colour with Congo red staining when visualised by light microscopy (Figure 3). Polarised light showed the deposits to have an apple-green birefringence while an immunofluorescence labelling with Thioflavin-t was also positive (Figure 4). A diagnosis of amyloidosis was therefore made.

Following the histological diagnosis in this case, the patient was referred for systemic work-up which included a bone scan, serum electro- and immunophoresis and urinalysis. A bone marrow trephine biopsy confirmed the presence of sheets of neoplastic plasma cells in keeping with multiple myeloma. The patient is currently receiving chemotherapy although his general condition has deteriorated markedly.
DISCUSSION
Amyloidosis represents deposition of abnormally folded fibrillary proteins. Isolated tongue involvement is considerably rare, with most cases representing systemic involvement secondary to an immune dyscrasia.1 The head and neck region is involved in 12 to 90% of cases of systemic amyloidosis with deposition occurring frequently in the oral cavity, pharynx, larynx, salivary glands and orbital sinuses. The tongue is the most commonly involved intraoral site, however, involvement of the gingiva, palate and lips has also been documented.2,3 The accumulation of these extra-cellular proteins compresses adjacent normal tissues and results in tissue dysfunction and eventual organ failure.4,5
Amyloid is broadly classified as systemic or localised. Systemic amyloidosis affects multiple tissue sites and organs in the body and is aetiologically divided into primary, secondary and hereditary forms. An acquired form as a result of haemodialysis is also included in this group. Primary amyloidosis represents the production of amyloid light chain proteins (AL) in association with plasma cell dyscrasias and multiple myeloma. In such cases increased amounts of immunoglobulin light chains are produced by the lesional plasma cells.6 Amyloidosis is reported in 5 to 15% of patients with multiple myeloma.4 Secondary amyloidosis represents a form of non-immunoglobulin derived protein (AA type), usually produced in increased amounts by the liver in chronic inflammatory disease such as osteomyelitis, tuberculosis, rheumatoid arthritis and Crohn's disease.7 Hereditary amyloidosis is associated with genetic mutations which result in abnormal protein production and systemic deposition.6
Localised amyloidosis is far rarer and represents the deposition of amyloid which is limited to a single organ or tissue. Amyloid of this type is often associated with endocrine neoplasms where it is derived from polypeptide hormones or as a result of aging where transthyretin protein is frequently deposited in the heart.6,7
Head and neck involvement by amyloid deposition may occur in both systemic and localised forms of disease although it is exceptionally rare in the latter form.1 Amyloidosis of the tongue is typically associated with primary systemic amyloidosis in the setting of an immune dyscrasia. The pathogenesis of amyloidosis in this scenario relates to the monoclonal production of immunoglobulin light chains by the lesional plasma cells. Proteolytic enzymes derived from macrophages in these disease processes convert the light chains into fibrillary proteins which are deposited systemically in the tissues.4,6 The excess light chain production results in excretion of Bence-Jones proteins in the urine and may be detected by both electro- and immune - phoresis. The lesional plasma cells proliferate within the bone marrow at the expense of the other haematopoietic elements resulting in anaemia, thrombocytopaenia and leukopenia. Patients will therefore be susceptible to infections, fatigue and have a tendency to bruise easily. The multifocal punched out skeletal lesions further predispose the patient to bone pain and pathological fracture.6,7 Deposition of amyloid in the tongue is slowly progressive and is recognised as one of the important causes of macroglossia. Continued deposition will result in restricted tongue movement and in the dentate patient, pressure of an enlarged tongue against the occlusal surfaces of the adjacent teeth results in the characteristic indentations or crenations as noted in this case. Patients often have a loss of peri-orbital and labial soft tissue elasticity due to the subcutaneous and submucosal deposition of amyloid proteins.6
The clinical differential diagnosis in this patient would include other causes of macroglossia such as lymphangioma, haemangioma, acromegaly, hypothyroidism, syndromic macroglossia and tumour infiltration.8 The characteristic features of tongue rigidity, a waxy firm cut-surface of the biopsy specimen as well as the typical microscopic features as noted in the present case are fairly diagnostic of amyloid deposition.
CONCLUSION
It is absolutely imperative to perform a systemic work-up and investigation in any case of amyloidosis in order to distinguish systemic from localised disease as well as to target the underlying cause in each case. The localised and systemic forms of disease have vastly different prognoses and therapeutic management
References
1. Babburi S, Ramya B, Subramanyam RV, Aparna V, Srivastava G. Amyloidosis of the tongue-report of a rare case. J Clin Diagn Res 2013; 7: 3094-5. [ Links ]
2. Andreadis D, Poulopoulos A, Papadopoulos P, Epivatianos A. Localized tongue amyloidosis in a patient with neurofibromatosis type II. Head Neck Pathol 2011; 5: 302-5. [ Links ]
3. Kerner MM, Wang MB, Angier G, Calcaterra TC, Ward PH. Amyloidosis of the head and neck. A clinicopathologic study of the UCLA experience, 1955-1991. Arch Otolaryngol Head Neck Surg 1995; 121: 778-82. [ Links ]
4. Vasudevan JA, Somanathan T, Patil SA, Kattoor J. Primary systemic amyloidosis of tongue with chondroid metaplasia. J Oral Maxillofac Pathol 2013; 17: 266-8. [ Links ]
5. Raubenheimer EJ, Dauth J, de Coning JP. Multiple myeloma presenting with extensive oral and perioral amyloidosis. Oral Surg Oral Med Oral Pathol 1986; 61: 492-7. [ Links ]
6. Gouvea AF, Ribeiro AC, Leon JE, Carlos R, de Almeida OP, Lopes MA. Head and neck amyloidosis: clinicopathological features and immunohistochemical analysis of 14 cases. J Oral Pathol Med 2012; 41: 178-85. [ Links ]
7. Bucci T, Bucci E, Rullan AM, Bucci P, Nuzzolo P. Localized amyloidosis of the upper gingiva: a case report. J Med Case Rep 2014; 8: 198. [ Links ]
8. Topouzelis N, Iliopoulos C, Kolokitha OE. Macroglossia. Int Dent J 2011; 61: 63-9. [ Links ]
 Correspondence:
Correspondence:
WFP van heerden
Department of Oral Pathology and Oral Biology, University of Pretoria, Pretoria, South Africa
Tel: 012 3192320. Fax: 012 3212225
E-mail: willie.vanheerden@up.ac.za













