Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Dental Journal
versão On-line ISSN 0375-1562
versão impressa ISSN 0011-8516
S. Afr. dent. j. vol.70 no.4 Johannesburg Mai. 2015
CASE BOOK
Oral medicine case book 69: Burkitt lymphoma of the oral cavity
Ε MosalleumI; Ν MohamedII; FC BassaIII; WP DreyerIV
IBDS, MCHD, FCPath(SA). Division of Oral Pathology, Faculty of Dentistry, University of the Western Cape; National Health Laboratory Services, Tygerberg Hospital
IIMBChB, MMed(Anat Path). Division of Anatomical Pathology, Stellenbosch University; National Health Laboratory Services, Tygerberg Hospital
IIIMBChB, FCPath(Haem), MMed. Division of Clinical Haematology, Department of Medicine, Faculty of Health Sciences, Stellenbosch University
IVBDS, HDipDent, PhD, FCD(SA)OMP. Division of Oral Medicine and Periodontics, Faculty of Dentistry, University of the Western Cape; Professor Emeritus, Stellenbosch University
CASE REPORT
A 25-year-old female was referred to the Haematology Unit at Tygerberg Hospital for further management of a rapidly expanding and large submandibular mass which on fine needle aspiration was suggestive of lymphoma (Figure 1). Five months earlier she had been diagnosed with pulmonary tuberculosis and was confirmed to be HIV positive with a CD4 count of 17. She was placed on anti-retroviral (ARV) and antituberculous therapy (the ARV therapy included efavirenz, emtricitabine and tenofivir). Her CD4 count, at the time of the current consultation, was 204 and the viral load was suppressed. Lumbar puncture was normal. Significant clinical findings were a large right submandibular mass and right cervical and axillary lymphadenopathy. The submandibular mass was removed and submitted for histological examination.
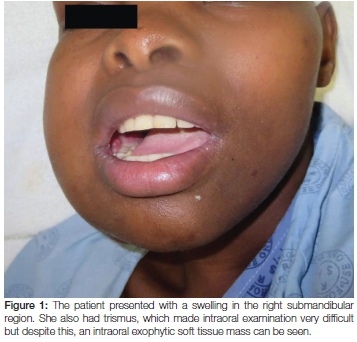
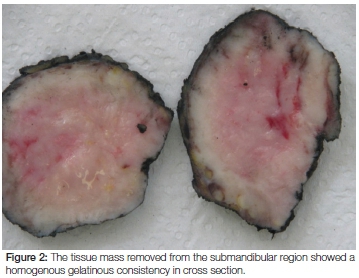
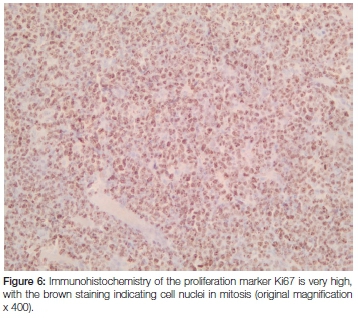
Macroscopic examination revealed a 62 gram, homogenous firm whitish-yellowish mass with areas of myxoid consistency. The size of the mass was 100 x 80 x 50 mm and, on cross section, neither necrosis nor haemorrhage was seen (Figure 2). Microscopic examination showed that the lesion was lymphoid in nature with relatively monomorphic cells with blue cytoplasm and nuclei that contained 3-5 nucleoli. High mitotic and apoptotic activity were seen as well as the presence of prominent scattered tingible body macrophages (ie. macrophages containing phagocytized, apoptotic cells in various states of degradation: "tingible" meaning stainable) (Figures 3 and 4). A panel of immunohistochemical markers was done to type the lymphoma (Figure 5 and 6). The cells were positive for the following markers: CD45 (leukocyte common antigen, confirming the lymphoid nature of the neoplasm); the B-cell antigen, CD20 (confirming that the lymphoma is of B-cell origin); and germinal centre B-cell antigens, CD10 and Bcl6 (confirming that the B-cell lymphoma derives from the lymphocytes in the germinal centre of the lymphoid follicle). The cells showed a 100% proliferative activity as illustrated by the Ki67, which is an immunohistochemical antibody that targets the tumour cells that are dividing or in a mitotic stage.



The morphology of this case is typical of Burkitt lymphoma and, in order to confirm this provisional diagnosis, molecular confirmation of translocation (8;14) involving c-myc gene was requested. The molecular investigations using fluorescent in situ hybridization (FISH) showed the presence of that translocation (8;14), thus confirming a diagnosis of Burkitt lymphoma. Staging CT scan revealed Ann Arbor stage IV disease with extensive lymphadenopathy involving the right submandibular, mediastinal, para-aortic, porta-hepatic and iliac nodes as well as splenic and hepatic involvement. There was no evidence of bony involvement. The patient received intensive combination chemotherapy together with continuation of ARV therapy. At the time of publication of this report, she had received four cycles of chemotherapy and showed an excellent interim response.
DISCUSSION
Lymphomas are neoplasms that affect the lymphoid system. They are divided into two major categories; Hodgkin and non-Hodgkin lymphoma. The non-Hodgkin lymphomas are further sub-classified into B-cell and T-cell lymphomas with B-cell lymphomas showing a high level of morbidity in HIV positive patients. Burkitt lymphoma is a very aggressive high grade non-Hodgkin B-cell lymphoma that is derived from germinal centre lymphoid B cells. This neoplasm is the third most common malignancy in African children, preceded only by Wilms' tumour (a rapidly developing malignant mixed tumour of the kidneys) and brain tumours. Burkitt lymphoma is named after Dennis Parsons Burkitt, a surgeon who first described the tumour in 1958 while working in equatorial Africa.1-3
Burkitt lymphoma is classified into three variants:4-10
- Endemic variant (also referred to as the African variant), most commonly occurring in the jaws of children living in malaria endemic regions of the world (eg. equatorial Africa, Brazil and Papua New Guinea). The endemic form of Burkitt lymphoma accounts for 30-40% of malignancies in children living in equatorial Africa.4 Interestingly, the distribution of the neoplasm in these areas corresponds to the distribution of endemic malaria, hence the term endemic Burkitt lymphoma. It is thought that chronic malaria reduces resistance to infection with the Epstein-Barr virus (EBV), a known oncogenic virus associated with endemic Burkitt lymphoma.4 The common age of presentation is 4 to 7 years and the most common clinical presentation is as a jaw mass (58%), or abdominal mass including involvement of the ileum, kidneys and ovaries. The bone marrow and the central nervous system may also be involved.1,3,4,8
- Sporadic variant (also referred to as the non-African variant), a variant not restricted to areas of endemic malaria and, although the tumour cells have a similar appearance to the endemic form, this tumour is rarely associated with EBV infection and is less commonly found in the jaws. It mainly involves the abdomen (88%), with rare involvement of the head and neck. There is no specific geographic association with this variant of Burkitt lymphoma, however, it is usually seen in European populations.1,3,4,6
- Immunodeficiency associated subtype of Burkitt lymphoma is frequently seen in Aids patients with a CD4 count below 200 cells/pL and can also be found in patients on immunosuppressive drugs following organ transplantation. It is considered to be one of the conditions signalling an underlying HIV infection.4-6 Similar to the sporadic form, the immunodeficiency associated Burkitt lymphoma can be found in extra-nodal sites such as this case presented above. The abdomen is also commonly involved and abdominal pain, vomiting, bowl obstruction and gastrointestinal bleeding constitutes the usual clinical presentation. In 13-17% of patients the brain is involved.6
All forms of Burkitt lymphoma share the same morphology and it is almost impossible to differentiate these three variants on microscopic features alone. The chromosomal rearrangement associated with c-myc oncogene expression is the hallmark of the molecular alterations in Burkitt lymphoma and this leads to the disturbance in germinal centre B-cell proliferation and differentiation. As could be expected, most of the cases seen in South Africa fall in the sporadic or HIV-associated variants.1
Histopathology, genetic features and pathogenesis
The morphology and the immunohistochemistry of Burkitt lymphoma support an origin from the follicular centre B-lymphocytes. The tumour forms sheets of small to intermediate monomorphic lymphocytes with basophilic cytoplasm and central nuclei containing 3-5 nucleoli. High proliferative activity is reflected by the tumour cells and the scattered associated tingible body macrophages give rise to the characteristic picture of a starry sky appearance.4,7 The immunodeficiency phenotype of Burkitt lymphoma is consistent with a B-cell lineage. The cells are reactive for CD20, CD19, CD22, and CD79a, which are antibodies that detect surface antigens of B lymphocytes. They also express germinal centre antibodies, such as can be demonstrated by immuno-staining for Bcl6 and CD10.5
The ability of EBV to transform the B-lymphocytes in Burkitt lymphoma is said to be amplified by malarial parasitaemia.4 This is why the endemic form of this lymphoma is common in the so-called lymphoma belt; an area between 10° south and 10° north of the equator, roughly corresponding to the area of endemic malaria.1 However, additional mutations are required for the development of this lymphoma beside the involvement of EBV. These mutations generally involve chromosome 8 and 14 and, at times, chromosome 2 and 8.4,67
Treatment and prognosis
Chemotherapy is the primary treatment modality for patients with Burkitt lymphoma. The neoplastic cells are very sensitive to cytotoxic therapy and intensive multi-agent chemotherapy regimens are recommended in Burkitt lymphoma patients who are HIV negative. In HIV positive patients, these protocols are modified in order to reduce toxicity particularly in patients who are ARV therapy naive.4,7,11 Involvement of the central nervous system (CNS), bone marrow and HIV infection indicate a less favourable outcome and therefore CNS chemoprophylaxis is advocated.4,7 A recent study in a Korean population showed that the two-year survival rates are lower in patients with bone marrow and CNS involvement, however, involvement of other extra-nodal sites had no effect on the prognosis.8 Therefore, a good understanding of the pathogenesis associated with the different subtypes is essential for the development of new chemotherapeutic therapeutic strategies.7
CONCLUSION
Burkitt lymphoma is a very aggressive fast growing non-Hodgkin lymphoma. The importance of early diagnosis and treatment cannot be overemphasized as treatment success is dependent on the extent of disease. Burkitt lymphoma is one of the human cancers that highlight the role of viruses in the carcinogenic process and, as a consequence, it is one of the lymphomas that develop in persons who are immunodeficient, either as a result of HIV infection or subsequent to organ transplantation. Knowledge about the clinical presentation, usual extra-nodal involvement and the subtypes of this lymphoma is essential for making an early diagnosis and therefore can make a significant difference in the prognosis of the afflicted patient.
Declaration: No conflict of interest declared
References
1. Davidson A, Desai F, Hendricks M, et al. The evolving management of Burkitt's lymphoma at Red Cross Children's Hospital. S Afr Med J 2006; 96: 950-4. [ Links ]
2. Van den Bosch CA, Hills M, Kazembe P, et al. Space-time case-clusters of endemic Burkitt's lymphoma in Malawi. Leukaemia 1993; 7: 1875-8. [ Links ]
3. Orem J, Mbidde EK, Lambert B, de Sanjose S, Weiderpass E. Burkitt's lymphoma in Africa, a review of the epidemiology and etiology. African Health Sci 2007; 7: 166-75. [ Links ]
4. Banthia V, Jen A, Kacker A. Sporadic Burkitt's lymphoma of the head and neck in the pediatric population. Internat J Pediatr Otorhinolaryngol 2003; 67: 59- 65. [ Links ]
5. Rowe M, Kelly GL, Bell AI, Rickinson AB. Burkitt's lymphoma: The Rosetta Stone deciphering Epstein-Barr virus biology. Seminars Cancer Biol 2009; 19: 377-388. [ Links ]
6. Blum KA, Lozanski G, Byrd J C. Adult Burkitt leukemia and lymphoma. Blood 2004; 104: 3009-20. [ Links ]
7. Soldini D, Campo E. Genetic sequencing studies in Burkitt's lymphoma: what can we learn about tumorigenesis? Expert Rev Hematol 2013; 6: 219-21. [ Links ]
8. Jang S-J, Yoon D H, Kim S et al. A unique pattern of extranodal involvement in Korean adults with sporadic Burkitt lymphoma: a single center experience. Ann Hematol 2012; 91:1917-22. [ Links ]
9. Carbone A. Emerging pathways in the development of AIDS related lymphomas. Lancet Oncol 2003; 4: 22-9. [ Links ]
10. Kamulegeya A, Muwazi L, Kasaganki A, Rwenyonyi CM, Kuteesa A. Trends in Burkitt's lymphoma: a three-decade retrospective study from Uganda. Oral Surg 2010; 3: 129-36. [ Links ]
11. Dunleavy K, Wilson W. How I treat HIV associated lymphoma. Blood; 2012;119: 3245-55 [ Links ]
 Correspondence:
Correspondence:
WP Dreyer
PO Box 1285, Sedgefleld, 6573
E-mail wpd@sun.ac.za













