Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
African Entomology
On-line version ISSN 2224-8854Print version ISSN 1021-3589
AE vol.33 Pretoria 2025
https://doi.org/10.17159/2254-8854/2025/a20570
RESEARCH ARTICLE
Illegal sand mining alters the species composition of ants, beetles, and spiders in a coastal grassland: a case study in Eastern Cape, South Africa
Inam Yekwayo; Asabonga Mngeni
Department of Biological and Environmental Sciences, Walter Sisulu University, Mthatha, South Africa
ABSTRACT
In the Eastern Cape, natural landscapes that are adjacent to rivers and the sea are threatened by illegal sand mining, which occurs without prior assessment of biodiversity. Sand mining activities lead to the reduction of vegetation cover and plant species richness, which alters microhabitats of epigaeic arthropods. Thus, it is important to assess the effects of sand mining on arthropod diversity. In this study we compared morphospecies richness, abundance, evenness, Shannon-Wiener diversity and composition of functional guilds of ants, beetles and spiders between sand-mined dune and the unmined grassland dune. Arthropods were collected using pitfall traps in the sand-mined and unmined grassland dunes. The sand-mined and unmined grassland dunes supported similar morphospecies richness, evenness and diversity of detritivores, herbivores, omnivores and predators. Sand-mined and unmined grassland dunes had similar abundance of other functional guilds, except for herbivores that were abundant in the unmined grassland. Furthermore, the two types of dunes supported significantly different morphospecies composition of all guilds. Additionally, there were 12 morphospecies that were identified as indicators of the sand-mined dune, and 11 morphospecies that were indicators of the unmined grassland dune, while there was no shared indicator species found between the two dunes. As such, disturbance-tolerant species that prefer open habitats may have replaced specialist arthropods in the sand-mined dune. This study showed that illegal sand mining changes morphospecies composition and reduces the abundance of herbivores, and as such sand mining should be restricted to designated areas as to reduce the impact of mining on arthropods and enhance conservation efforts.
Keywords: abundance; Araneae; arthropods; Coleoptera; conservation; Hymenoptera; species richness
INTRODUCTION
Assemblages of epigaeic arthropods are shaped by a variety of characteristics, such as percentage of canopy cover and bare ground, leaf litter, grass, forbs, and vegetation height (Lingbeek et al. 2017; Lafage et al. 2019). As such, the alteration of habitats often affects epigaeic arthropods due to their limited dispersal abilities (Perry et al. 2021). Willett (2001) reported a decrease in species richness and abundance of spiders following logging and the loss of herbaceous cover. Additionally, greater species richness and abundance of arthropods are often recorded in less disturbed and/ or undisturbed habitats compared to highly disturbed habitats (Lingbeek et al. 2017; Swart et al. 2019). Undisturbed habitats have high species richness of native plants, and according to Botha et al. (2017) and Longcore (2003) a high plant species richness enhances the species richness and diversity of arthropods. For example, both beetles and spiders were found to be more species rich in undisturbed forests than disturbed forests (Uehara-Prada et al. 2009). However, Yekwayo et al. (2017) showed that disturbance does not affect all arthropods because pines reduced abundance of predators only, while species richness and abundance of detritivorous arthropods and ants did not differ between pine plantations and natural forests. Furthermore, in South Africa high species richness of ants and beetles, and high abundance of ants and spiders were recorded in the disturbed savanna compared to the undisturbed savanna (Mavasa et al. 2023). Although some of these studies did not report a decrease in arthropod diversity, disturbed and undisturbed habitats often support different assemblages of arthropods (Uehara-Prada et al. 2009; Lingbeek et al. 2017; Tajthi et al. 2017; Mavasa et al. 2023). This further highlights the sensitivity of arthropods to disturbance, indicating that a disturbance may create microhabitats that are not suitable for some species, while attracting other species. For example, unburned forest plots supported species of ants and spiders that occupy vegetated habitats, while burned forest plots supported species that are associated with open habitats (Vidal-Cordero et al. 2022). In addition, unburned forests supported more web-building spiders than burned plots (Vidal-Cordero et al. 2022), this may have been due to greater availability of web attachment sites.
Sand mining is similar to other disturbances because it is detrimental to the physical structure of the landscape (Rentier and Cammeraat 2022), it may therefore alter microhabitats for arthropods. Legal mining of sand in South Africa contributes towards the country's economy because it provides job opportunities for many people, especially those residing near the mining sites (Naidoo 2008). However, because of the monitory value associated with sand mining, illegal mining has also been reported (Mngeni et al. 2017a). According to Gondo et al. (2019) a total of 11 900 m3 of sand is mined monthly from the Njelele River in Limpopo, South Africa, which indicates that there is a high demand for sand. This high demand for sand together with a high unemployment rate in South Africa (Francis et al. 2020) are the drivers of illegal mining of sand, which occurs in unregistered mining sites (Chevallier 2014; Amponsah-Dacosta and Mathada 2017; Mngeni et al. 2017a). The impact of sand mining is likely to be greater in unregistered mining sites because it occurs without prior environmental assessment (Amponsah-Dacosta and Mathada 2017) and there is no monitoring by authorities. Additionally, when unregistered mining sites close, they leave open pits, and no rehabilitation of the area occurs (Chevallier 2014; Amponsah-Dacosta and Mathada 2017). In addition to the removal of native vegetation and creation of pits, sand mining leads to soil erosion and landslides (Ako et al. 2014; Mngeni et al. 2017b; Gondo et al. 2019). Furthermore, sand mining activities can enhance the invasion of alien plant species. For example, Lantana camara Linnaeus was observed near coastal sand dunes that were being mined in the Eastern Cape, South Africa (Mngeni et al. 2017b). The changes in the natural landscapes that are driven by the extraction of sand also affects animals because their habitats are destroyed or altered (Ako et al. 2014).
An undisturbed dune forest at Richard's Bay in South Africa supported greater diversity of insects compared to postmining rehabilitating dune sites, with the recently rehabilitated site having the lowest diversity (Kumssa et al. 2004). Furthermore, in Uruguay, highly fragmented coastal sand dunes supported low abundance of Allocosa brasiliensis Petrunkevitch (Araneae: Lycosidae) than less fragmented dunes (Jorge et al. 2015). Although Damptey et al. (2023) focused on gravel mining, they recorded low density of spiders in a previously mined area compared to natural forests, restored forests and agroforestry plantations. Furthermore, in a checklist of spiders of coastal dune forest at Richard's Bay, Dippenaar-Schoeman and Wassenaar (2002, 2006) recorded a variety of species of web-building and wandering spiders. However, in South Africa, particularly in the Eastern Cape province, there is limited information on the diversity of epigaiec arthropods on dune vegetation or the response of these taxa to sand mining.
Ants, beetles and spiders are usually sampled in large numbers when using the pitfall trapping method, as such this makes each taxon analysable. These arthropod taxa have diverse functional guilds, leading to specialization in habitat requirements within each taxon (Picker et al. 2019; Dippenaar-Schoeman 2023). The sensitivity of ants and beetles to habitat changes, makes these taxa good bioindicators of disturbances (Chowdhurry et al. 2023). As such, understanding how ants, beetles and spiders respond to a disturbance, such as mining of sand, may provide a broader indication of how the entire ecosystem is being affected. Therefore, this study determined if functional guilds of ants, beetles and spiders respond differently to sand mining. This was through comparing morphospecies richness, abundance, evenness, Shannon-Wiener diversity and composition of each guild between the sand-mined and unmined dune grassland. Given that sand mining is an anthropogenic activity, we expected the sand-mined dune to support low species richness, abundance, evenness and diversity compared to the unmined dune grassland. Furthermore, because of vegetation structural differences between the sand-mined and unmined grassland dune, we expect variations in morphospecies composition of arthropods.
MATERIALS AND METHODS
Study area
The study was conducted at Mbolompo (32°4.612' S, 29°5.060' E), located within the Amathole District Municipality, Eastern Cape, South Africa (Figure 1). The Karoo Supergroup covers the Mbolompo area, which consists of a mosaic of a grassland matrix that is dotted with patches of bush clumps and small forests (Mucina and Rutherford 2006). There is also a belt of coastal sand dune thicket (Mucina and Rutherford 2006). The study site has been exposed to illegal sand mining. The sand-mining area is approximately 268 871 m2 (~27 ha) and is >50 m away from the sea. The study area is exposed to subsistence farming through communal cattle grazing. The Mbolompo area experiences about 1 111.64 mm of summer rainfall, while the winter season is dry (NASA POWER Project 2025). The mean maximum summer temperature of the Mbolompo area is 31°C and the mean annual maximum temperature is 29°C (NASA POWER Project 2025).
We sampled in summer (late November to December 2021) due to warmer conditions in the Eastern Cape (Nkamisa et al. 2022) offering a greater arthropod activity (Mavasa et al. 2022). Using a distance of >200 m between plots within the same category (sand-mined dune or unmined grassland dune), a total of 16 plots were established, eight in each category (Figure 1). We considered these plots to be independent of each other (Samways et al. 2010) as many epigaeic arthropods appear to have limited dispersal abilities (Perry et al. 2021). Establishing all plots in one sand mining site is the limitation of this study. We were unable to avoid this limitation because of the lack of comparable sites within the same area. The altitude of the plots ranged between 12-75 m above sea level.
Sampling design
Each plot was represented by a 5 m × 5 m quadrat. Arthropods were sampled using pitfall traps (6 cm in diameter and 8 cm deep) that were half-filled with 50% ethylene glycol solution, which is not known to attract nor repel arthropods (Gerlach et al. 2009). At each plot six pitfall traps were set out one meter apart in a rectangular shape. To get a more representative sample, we sampled over 28 days, with pitfall traps being emptied on every 7th day after insertion, and this was repeated for a total of four occasions. Data from the six pitfall traps and four sampling times for each plot were pooled to form a single sample. We avoided digging-in effects by leaving pitfall traps uncovered for seven days because Jiménez-Carmona et al. (2019) reported that digging-in effects are more likely to occur within the first 24 hours after setting out traps. The collected specimens were preserved in 70% ethanol. The three studied taxa (ants, beetles, and spiders) were extracted and sorted into morphospecies. Most ants were identified to genus and species levels by the first author and confirmed by Dr Caswell Munyai from the University of KwaZulu-Natal. However, the family level was used for most beetles, and spiders. Various guides were followed when identifying taxa (Dippenaar-Schoeman and Jocqué 1997; White 1998; Davis et al. 2008; Bouchard 2014; Fisher and Bolton 2016; Picker et al. 2019; Dippenaar-Schoeman 2023). Although we acknowledge the limitations of using morphospecies, this is generally acceptable in studies that are focussed on assessing environmental impacts (Derraik et al. 2002; Obrist and Duelli 2010). Classification of arthropods (including ants, beetles and spiders) to morphospecies has been done previously in South Africa (Swart et al. 2019; van Schalkwyk et al. 2021; Geldenhuys et al. 2022). Arthropods were then assigned to functional guilds. Given that some of the specimens (particularly beetles) were identified to family level, for those morphospecies the family was used when assigning guilds. The following guilds were identified, herbivores (ants and beetles), detritivores (beetles), omnivores (ants and beetles), and predators (ants, beetles and spiders) (Fisher and Bolton 2016; Picker et al. 2019; Dippenaar-Schoeman 2023).
Vegetation cover across different plots within each of the two dune types (sand-mined and unmined grassland) appeared similar. As such, at each of the 16 sampling plots a 1 m × 1 m quadrat was used to visually estimate the percentage of vegetation cover. Estimation of vegetation cover was done once during the sampling period. We estimated vegetation cover in the second week of setting out pitfall traps.
Data analyses
Analyses were conducted in R version 4.1.2 (R Core Team) and PRIMER 7 (Anderson et al. 2008). Analyses were conducted separately for each of the four functional guilds (detritivores, herbivores, omnivores and predators). The Shapiro-Wilk test was used to test the data for normality (Rochon et al. 2012). The abundance of detritivores and omnivores, as well as Shannon-Wiener diversity of detritivores and herbivores were normally distributed (p > 0.05). The morphospecies richness of herbivores, omnivores and predators, as well as evenness of omnivores were normally distributed. However, morphospecies richness and evenness of detritivores, as well as abundance and morphospecies evenness of herbivores were nonnormal (p < 0.05). Similarly, abundance, evenness and Shannon-Wiener diversity of predators were nonnormal. Therefore, to compare these measures of diversity between sand-mined and unmined grassland dunes, linear models were used when datasets were normal, while for nonnormal datasets the generalized linear models were used. Spearman's rank correlation was used to determine if the percentage of vegetation cover correlated with morphospecies richness or abundance of each functional guild. The Spearman's rank correlation was used because the dataset for the vegetation cover was not normally distributed. In addition, the generalized linear model with the negative binomial distribution and the MASS package (Venables and Ripley 2002) was used to compare the percentage of vegetation cover between sand-mined dune and unmined grassland dune.
Morphospecies composition of each guild was compared between sand-mined dune and unmined grassland dune using the permutational multivariate analysis of variance (PERMANOVA) in PRIMER. The data were square-root transformed, and the analysis was conducted using the Bray-Curtis similarity measures (Anderson 2001). Principal coordinates analysis in PRIMER (Anderson et al. 2008) was performed to visualize differences in morphospecies composition between the sand-mined dune and the unmined grassland dune. To identify morphospecies that are indicators of the sand-mined dune, unmined grassland dune and shared between these areas, we used the indicator value analysis (De Cáceres and Legendre 2009). The indicator value analysis uses the Multipatt function in the indicpsecies package (De Cáceres and Legendre 2009). The frequent occurrence and abundance of morphospecies in a habitat type were used in determining indicator morphospecies (Samways et al. 2010).
RESULTS
A total of 6 827 individuals of ants (70.8%), beetles (16.8%) and spiders (12.4%) were collected. Out of a total of 244 morphospecies collected, spiders had the highest percentage (45.1%), followed by beetles (42.2) and ants (12.7). There were more individuals sampled in the sand-mined dune (58.8%) than in the unmined grassland dune (41.2%) (Supplementary Tables S1, S2). However, a slightly greater number of morphospecies (166) was collected from the grassland dune than from the sand-mined dune (146 morphospecies) (Supplementary Tables S1, S2). In both dune types, the most abundant and morphospecies rich subfamilies of ants were the Myrmicinae and Formicinae, while the Dorylinae was least abundant, and the Dolichoderinae was represented by one morphospecies (Tapinoma sp.). In the sand-mined dune, families of beetles with the highest abundance were the Cicindelidae, and Scarabaeidae, while families with the highest number of morphospecies were the Scarabaeidae and Staphylinidae (Table S1). In the unmined grassland dune, the most abundant beetle families were the Staphylinidae, Scarabaeidae, Carabidae and Chrysomelidae, while the Staphylinidae, Scarabaeidae and Carabidae had high morphospecies richness (Table S2). Although the Cicindelidae had the highest number of individuals, these were from two morphospecies (Table S1). In the sand-mined dune, the highest number of individuals and morphospecies of spiders were recorded in the Lycosidae, Gnaphosidae, Zodariidae and Theridiidae (Table S1). The families of spiders with the highest abundance in the unmined grassland dune were the Lycosidae, Zodariidae and Theridiidae, whereas the Lycosidae, Zodariidae, Corinnidae and Salticidae were morphospecies rich (Table S2).
Morphospecies richness, evenness, abundance and Shannon-Wiener diversity of detritivores, omnivores and predators were similar between the sand-mined dune and unmined grassland dune (Table 1). Similarly, morphospecies richness, evenness and Shannon-Wiener diversity of herbivores did not differ significantly between the sand-mined dune and unmined grassland dune (Table 1). However, significantly greater abundance of herbivores was recorded in the unmined grassland dune than the sand-mined dune (Table 1, Figure 2). The surrounding grassland had significantly (t = -3.35, p = 0.005) higher percentage of vegetation cover than the sand-mined dune. The percentage of vegetation cover did not correlate with either morphospecies richness of all functional guilds nor abundance of detritivores, omnivores and predators (Table 2). However, the percentage of vegetation cover correlated positively with the abundance of herbivores (Table 2).
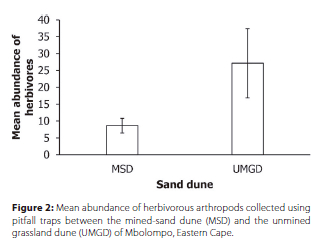
There were significant differences in morphospecies composition of all functional guilds between the sand-mined dune and the unmined grassland dune (Table 1). In addition, the principal coordinates analysis separated plots from each dune type, even though the distribution of plots (particularly omnivores) appeared to overlap between dune types (Figure 3). The indicator value analysis identified 12 morphospecies that were associated with the sand-mined dune and 11 morphospecies that were indicators of the unmined grassland dune (Table 3). The indicator ant morphospecies identified in the sand-mined dune represented the Dolichoderinae (Tapinoma sp.), Formicinae (Lepisiota sp.) and Myrmicinae (Cardiocondyla sp., Crematogaster sp. and Tetramorium sp.) subfamilies (Table 3). However, there were two morphospecies of Tetramorium and Mesoponera caffraria Smith that occurred frequently in the unmined grassland dune (Table 3). There were five morphospecies of beetles that were indicators of the unmined grassland dune, while the sand-mined dune had Lophyra sp. and Rhyssemus sp. as indicators (Table 3). Two morphospecies of the Lycosidae (Foveosa foveolata Purcell and Trabea sp.) and Hahnia sp. were indicators of spiders in the unmined grassland dune (Table 3). However, in the sand-mined dune three morphospecies of the Lycosidae (Pardosa crassipalpis Purcell, Pardosa sp. and Trabea sp.), one morphospecies of the Gnaphosidae (Asemesthes sp.) and Zodariidae (Cydrela sp.) were identified as indicators (Table 3).
DISCUSSION
Our study showed that changes in vegetation cover as a result of sand mining activities do not appear to affect morphospecies richness, evenness and diversity of all functional guilds studied. However, the morphospecies composition of all functional guilds and abundance of herbivores differed between the sand-mined dune and unmined grassland dune. The sand-mined dune had little to no vegetation cover and a greater percentage of bare ground compared with the surrounding unmined grassland dune. As a result, these differences in vegetation cover may have led to differences in microhabitats for arthropods, thus, the sand-mined dune and the surrounding unmined grassland dune supported different morphospecies composition. Although sand mining activities may have provided unsuitable microhabitats for some species of ants, beetles and spiders, the sand-mined dune may have attracted other species. For example, some Tapinoma species build their nests in open habitats that are caused by anthropogenic activities (Sharaf et al. 2017; Seifert et al. 2024). Similarly, disturbed habitats tend to support greater abundance of some Pardosa species (Liu et al. 2024). This could explain the species of Tapinoma and Pardosa that we recorded as indicators of the sand-mined dune.
Hlongwane et al. (2024) recorded greater abundance of Pardosa crassipalpis Purcell in three studied habitat types (sand forest, ecotone and the savanna). However, in our study, P. crassipalpis was sampled in the sand-mined dune only, which may indicate that the generalist nature of this species was influenced by vegetation type.
Although sandy soil is dominant in both mined and unmined dune grassland, the frequent occurrence of some species of ants in the sand-mined dune might be due to both the sandy soil and the presence of artificial ponds from the open pits. This is because moist soils from the artificial ponds may have created a conducive environment for some species of Cardiocondyla, Lepisiota and Tetramorium (Sharaf et al. 2017). Similarly, Lophyra species, one of the indicators morphospecies of the sand-mined dune, are known to be diverse in sandy areas, especially those near water (Mawdsley and Sithole 2008; Dangalle et al. 2012; Picker et al. 2019). Out of the total abundance of beetles, this tiger beetle was the most abundant morphospecies. Furthermore, identification of a Rhyssemus species as an indicator of the sand-mined dune could be because species of this genus prefer sandy areas, including coastal sand dunes (Lim and Bae 2024).
Contrary to our findings, complex habitats support greater species richness and abundance of arthropods than simpler habitats (Gardner et al. 1995). However, when focusing of vegetation presence, according to Sackmann and Farji-Brener (2006) abundance of different arthropod taxa are affected differently by habitat complexity. For example, burnt (simpler) and unburnt (complex) habitats supported similar abundance of beetles, while ant abundance was higher in burnt plots than in the unburnt plots, with the steppe habitat being the most abundant followed by the scrub and then the forest (Sackmann and Farji-Brener 2006). An increase in the percentage of bare ground and simplicity of habitats has been reported to increase abundance (Graham et al. 2004) and species richness (Ossola et al. 2015) of ants compared to complex habitats. According to Gibb and Parr (2010) foraging in ants can be influenced by habitat complexity, because they found that ants detected baits faster in simpler habitats compared to complex habitats. Therefore, the sand-mined dune may not have a decreased morphospecies richness and abundance of ants because of easier foraging. However, in Mexico, ant diversity has been reported to be enhanced by plant species richness and plant cover (Chen et al. 2015). Furthermore, in North Carolina, Menke et al. (2011) recorded low species richness of ants in disturbed open urban areas compared to areas with thick canopy cover.
In Guinea, Yeo et al. (2019) recorded a greater abundance of ant species, such as M. caffraria in open habitats (savanna and meadows) compared to the forest habitat. However, in our study, M. caffraria was an indicator species of the unmined grassland dune, of which in the context of our study, represented closed habitat. Our study and that of Yeo et al. (2019) defined open and closed habitats differently and focused on different vegetation types (savanna, meadows, forest and grassland). However, in both studies M. caffraria was abundant in the habitats dominated by grass. Furthermore, Sharaf et al. (2017) reported that Tetramorium simillimum Smith forages on the ground underneath grasses, likely accounting for the sampling in the unmined grassland dune only.
Grazing by cattle took place in our studied areas, however, it was dominant in the unmined grassland dune because of greater vegetation cover compared to the sand-mined dune. According to Wagner et al. (2021) abundance of dung beetles increases in grazed areas compared to ungrazed areas. This is due to the presence of dung from grazers. This can explain the frequent occurrence of a Garreta species that was an indicator of the unmined grassland dune. Although abundance of other functional guilds did not differ between the sand-mined and unmined grassland dune, there was greater abundance of herbivorous arthropods in the unmined grassland dune compared to the sand-mined dune. In addition, a Chrysomelidae morphospecies was identified as an indicator of the unmined grassland dune, while no herbivorous morphospecies were indicators of the sand-mined dune. High abundance of herbivores in the unmined grassland dune can be attributed to greater vegetation cover because plant species richness and vegetation cover correlate positively with species richness of arthropods (Diehl et al. 2013; Malumbres-Olarte et al. 2013; Rahman et al. 2015; Gómez et al. 2016). In our study, percentage of vegetation cover did not correlate with abundance nor richness of other functional guilds, however, there was a positive correlation between percentage of vegetation cover and abundance of herbivores. Greater vegetation cover improves arthropod diversity because of increased habitat heterogeneity (Malumbres-Olarte et al. 2013; Rahman et al. 2015). Furthermore, web-building spiders occupy habitats based on prey availability and potential sites for constructing webs, both of which are more available in habitats with high vegetation cover (Roberson et al. 2016). Therefore, the sand-mined dune and the unmined grassland dune that we studied supported different assemblages of predators (which were dominated by spiders) because of differences in habitat requirements. However, Lubin et al. (2020) pointed out that even though most spider species require specific habitats, there are some species that are generalists. Furthermore, predatory arthropods are not influenced by plant species richness (Corcos et al. 2021), and we found no correlation of predatory arthropods with vegetation cover in our study. However, differences in morphospecies composition of all functional guilds between the sand-mined and unmined grassland dunes show that even amongst predatory arthropods, there are specific habitat requirements.
Our study provides evidence that sand mining alters morphospecies compositions due to changes in landscape structure by creating open habitats with limited resources. As a result, the foraging activities of arthropods in the sand-mined dune may have increased, leading to greater catch in pitfall traps. Therefore, similarities in morphospecies richness and abundance between sand-mined and unmined grassland dunes could have been due to resource availability, which may have influenced foraging patterns and the catchability of arthropods. Furthermore, differences in morphospecies composition of arthropod guilds between sand-mined dune and the unmined grassland dune may be because of the limited dispersal abilities and microhabitat specialization of the taxa.
ACKNOWLEDGEMENTS
We are grateful to Dr Caswell Munyai for confirming our identification of ants. In addition, we thank Professor Tarombera Mwabvu for his constructive comments on the first draft of the manuscript. Walter Sisulu University is thanked for providing transport during fieldwork.
AUTHORS' CONTRIBUTIONS
IY: conceptualisation, investigation, methodology, data curation, formal analyses and wrote the original draft.
AM: conceptualisation, investigation, methodology, and reviewed and edited the manuscript.
ORCID IDS
Inam Yekwayo: https://orcid.org/0000-0002-9722-6545
Asabonga Mngeni: https://orcid.org/0000-0001-5201-7400
REFERENCES
Ako TA, Onoduku US, Oke SA, Essien BI, Idris FN, Umar AN, Ahmed AA. 2014. Environmental effects of sand and gravel mining on land and soil in Luku, Minna, Niger State, North Central Nigeria. Journal of Geosciences and Geomatics. 2:42-49. http://pubs.sciepub.com/jgg/2/2/1. [ Links ]
Amponsah-Dacosta F, Mathada H. 2017. Study of sand mining and related environmental problems along the Nzhelele River in Limpopo Province of South Africa. In: Wolkersdorfer C, Sartz L, Sillanpää M, Häkkinen A (editors), Mine Water & Circular Economy (Vol II). Lappeenranta: Lappeenranta University of Technology. Pp 1259-1266.
Anderson MJ. 2001. A new method for non-parametric multivariate analysis of variance. Austral Ecology. 26:32-46. https://doi.org/10.1111/j.1442-9993.2001.01070.pp.x.
Anderson MJ, Gorley RN, Clarke R. 2008. PERMANOVA+ for PRIMER: Guide to Software and Statistical Methods. United Kingdom: PRIMER-E Ltd.
Bouchard P. 2014. The Book of Beetles: A Life-Size Guide to Six Hundred of Nature's Gems. Chicago: University of Chicago Press. https://doi.org/10.7208/chicago/9780226082899.001.0001. [ Links ]
Botha M, Siebert SJ, Van Den Berg J. 2017. Grass abundance maintains positive plant-arthropod diversity relationships in maize fields and margins in South Africa. Agricultural and Forest Entomology. 19:154-162. https://doi.org/10.1111/afe.12195. [ Links ]
Chen X, Adams B, Bergeron C, Sabo A, Hooper-Bùi L. 2015. Ant community structure and response to disturbances on coastal dunes of Gulf of Mexico. Journal of Insect Conservation. 19:1-13. https://doi.org/10.1007/s10841-014-9722-9. [ Links ]
Chevallier R. 2014. Illegal sand mining in South Africa. SAIIA: Policy Briefing 116.
Chowdhury S, Dubey VK, Choudhury S, Das A, Jeengar D, Sujatha B, Kumar A, Kumar N, Semwal A, Kumar V. 2023. Insects as bioindicator: A hidden gem for environmental monitoring. Frontiers in Environmental Science. 11:1146052. https://doi.org/10.3389/fenvs.2023.1146052. [ Links ]
Corcos D, Lami F, Nardi D, Boscutti F, Sigura M, Giannone F, Pantini P, Tagliapietra A, Busato F, Sibella R, Marini L. 2021. Cross-taxon congruence between predatory arthropods and plants across Mediterranean agricultural landscapes. Ecological Indicators. 123:107366. https://doi.org/10.1016/j.ecolind.2021.107366. [ Links ]
Damptey FG, Djoudi EA, Birkhofer K. 2023. Effects of post-mining forest restoration and alternative land uses on ground-dwelling arthropods in Ghana. Community Ecology. 24:215-228. https://doi.org/10.1007/s42974-023-00144-8. [ Links ]
Dangalle CD, Pallewatts N, Vogler AP. 2012. Habitat specificity of tiger beetle species (Coleoptera: Cicindelidae) of Sri Lanka. Cicindela. 44:1-32.
Davis ALV, Frolov AV, Scholtz CH. 2008. The African Dung Beetle Genera. Pretoria: Protea Book House. [ Links ]
De Cáceres M, Legendre P. 2009. Associations between species and groups of sites: indices and statistical inference. Ecology. 90:3566-3574. https://doi.org/10.1890/08-1823.1. [ Links ]
Derraik JGB, Closs GP, Dickinson KJM, Sirvid P, Barratt BIP, Patrick BH. 2002. Arthropod morphospecies versus taxonomic species: a case study with Araneae, Coleoptera, and Lepidoptera. Conservation Biology. 16:1015-1023. https://doi.org/10.1046/j.1523-1739.2002.00358.x. [ Links ]
Diehl E, Mader VL, Wolters V, Birkhofer K. 2013. Management intensity and vegetation complexity affect web-building spiders and their prey. Oecologia. 173:579-589. https://doi.org/10.1007/s00442-013-2634-7. [ Links ]
Dippenaar-Schoeman AS. 2023. Field Guide to the Spiders of South Africa. Cape Town: Struik Nature. [ Links ]
Dippenaar-Schoeman AS, Jocqué R. 1997. African Spiders: An Identification Manual. Pretoria: ARC-Plant Protection Research Institute. [ Links ]
Dippenaar-Schoeman AS, Wassenaar TD. 2002. A checklist of the ground-dwelling spiders of coastal dune forests at Richards Bay, South Africa (Arachnida: Araneae). Bulletin of the British Arachnological Society. 12:275-279. [ Links ]
Dippenaar-Schoeman AS, Wassenaar TD. 2006. A checklist of spiders from the herbaceous layer of a coastal dune forest ecosystem at Richards Bay, KwaZulu-Natal, South Africa (Arachnida: Araneae). African Invertebrates. 47:63-70. [ Links ]
Fisher BL, Bolton B. 2016. Ants of Africa and Madagascar: A Guide to Genera. California: University of California Press. https://doi.org/10.1525/9780520962996.
Francis D, Valodia I, Webster E. 2020. Politics, policy and inequality in South Africa under COVID-19. Agrarian South: Journal of Political Economy. 9:342-355. https://doi.org/10.1177/2277976020970036. [ Links ]
Gardner SM, Cabido MR, Valladares GR, Diaz S. 1995. The influence of habitat structure on arthropod diversity in Argentine semi-arid Chaco forest. Journal of Vegetation Science. 6:349-356. https://doi.org/10.2307/3236234. [ Links ]
Geldenhuys M, Gaigher R., Pryke JS, Samways MJ. 2022. Maintaining remnant vegetation along with plant diversification in vineyards is optimal for conserving arthropods in an agricultural mosaic in a biodiversityrich region. Biodiversity and Conservation. 31:3237-3255. https://doi.org/10.1007/s10531-022-02486-7. [ Links ]
Gerlach A, Voigtländer K, Heidger CM. 2009. Influences of the behaviour of epigeic arthropods (Diplopoda, Chilopoda, Carabidae) on the efficiency of pitfall trapping. Soil Organisms. 81:773-790. https://soil-organisms.org/index.php/SO/article/view/221. [ Links ]
Gibb H, Parr CL. 2010. How does habitat complexity affect ant foraging success? A test using functional measures on three continents. Oecologia. 164:1061-1073.https://doi.org/10.1007/s00442-010-1703-4. [ Links ]
Gómez JE, Lohmiller J, Joern A. 2016. Importance ofvegetation structure to the assembly of an aerial web-building spider community in North American open grassland. Journal of Arachnology. 44:28-35. https://doi.org/10.1636/P14-58.1. [ Links ]
Gondo T, Mathada H, Amponsah-Dacosta F. 2019. Regulatory and policy implications of sand mining along shallow waters of Njelele River in South Africa. Jàmbá: Journal of Disaster Risk Studies. 11:a727. https://doi.org/10.4102/jamba.v11i3.727. [ Links ]
Graham JH, Hughie HH, Jones S, Wrinn K, Krzysik AJ, Duda JJ, Freeman DC, Emlen JM, Zak JC, Kovacic DA, et al. 2004. Habitat disturbance and the diversity and abundance of ants (Formicidae) in the Southeastern Fall-Line Sandhills. Journal of Insect Science. 4:30. https://doi.org/10.1093/jis/4.1.30. [ Links ]
Hlongwane ZT, Munyai TC, Majola O, Dippenaar-Schoeman AS, Lagendijk DDG. 2024. Diversity, composition and distribution patterns of spiders (Arachnida: Araneae) in sand forest, South Africa. African Journal of Ecology. 62:e13334. https://doi.org/10.1111/aje.13334. [ Links ]
Jiménez-Carmona F, Carpintero S, Reyes-López JL. 2019. The digging-in effect on ant studies with pitfall traps: influence of type of habitat and sampling time. Entomologia Experimentalis et Applicata. 167:906-914. https://doi.org/10.1111/eea.12834. [ Links ]
Jorge C, Laborda Á, Días MA, Aisenberg A, Simó M. 2015. Habitat preference and effects of coastal fragmentation in the sand-dwelling spider Allocosa brasiliensis (Lycosidae, Allocosinae). Open Journal of Animal Sciences. 5:309-324. http://dx.doi.org/10.4236/ojas.2015.53035. [ Links ]
Kumssa DB, van Aarde RJ, Wassenaar TD. 2004. The regeneration of soil micro-arthropod assemblages in a rehabilitating coastal dune forest at Richards Bay, South Africa. African Journal of Ecology. 42:346-354. https://doi.org/10.1111/j.1365-2028.2004.00537.x. [ Links ]
Lafage D, Djoudi EA, Perrin G, Gallet S, Pétillon J. 2019. Responses of ground-dwelling spider assemblages to changes in vegetation from wet oligotrophic habitats of Western France. Arthropod-Plant Interactions. 13:653-662. https://doi.org/10.1007/s11829-019-09685-0. [ Links ]
Lim C, Bae J. 2024. New distribution record of genus Rhyssemus Mulsant (Coleoptera, Scarabaeidae, Aphodiinae) from South Korea. Journal of Species Research. 13:306-309. https://doi.org.10.12651/JSR.2024.13.3.306. [ Links ]
Lingbeek BJ, Higgins CL, Muir JP, Kattes DH, Schwertner TW. 2017. Arthropod diversity and assemblage structure response to deforestation and desertification in the Sahel of western Senegal. Global Ecology and Conservation. 11:165-176. https://doi.org/10.1016/j.gecco.2017.06.004. [ Links ]
Liu L, Fu D, Luo Y. 2024. Grassland expansions promoted global diversification of the Pardosa wolf spiders during the late Cenozoic (Araneae, Lycosidae). Zoosyst. 100:1287-1296. https://doi.org/10.3897/zse.100.128885. [ Links ]
Longcore T. 2003. Terrestrial arthropods as indicators of ecological restoration success in coastal sage scrub (California, U.S.A.). Restoration Ecology. 11:397-409. https://doi.org/10.1046/j.1526-100X.2003.rec0221.x. [ Links ]
Lubin Y, Ferrante M, Musli I, Lövei GL. 2020. Diversity of ground-active spiders in Negev desert habitats, Israel. Journal ofArid Environments. 183:104252. https://doi.org/10.1016/j.jaridenv.2020.104252. [ Links ]
Malumbres-Olarte J, Vink CJ, Ross JG, Cruickshank RH, Paterson AM. 2013. The role of habitat complexity on spider communities in native alpine grasslands ofNew Zealand. Insect Conservation and Diversity. 6:124-134. https://doi.org/10.1111/j.1752-4598.2012.00195.x. [ Links ]
Mavasa R, Yekwayo I, Mwabvu T, Tsvuura Z. 2023. Response of ants, beetles and spiders to disturbance varies among taxa in a South African savanna biome. African Entomology. 31:a13244. https://doi.org/10.17159/2254-8854/2023/a13244. [ Links ]
Mavasa R, Yekwayo I, Mwabvu T, Tsvuura Z. 2022. Preliminary patterns of seasonal changes in species composition of surface-active arthropods in a South African savanna. Austral Ecology. 47:1222-1231. https://doi.org/10.1111/aec.13213. [ Links ]
Mawdsley JR, Sithole H. 2008. Dry season ecology of riverine tiger beetles in Kruger National Park, South Africa. African Journal of Ecology. 46:126-131 https://doi.org/10.1111/j.1365-2028.2007.00820.x. [ Links ]
Menke SB, Guénard B, Sexton JO, Weiser, MD, Dunn RR, Silverman J. 2011. Urban areas may serve as habitat and corridors for dry-adapted, heat tolerant species; an example from ant. Urban Ecosystems. 14:135-163. https://doi.org/10.1007/s11252-010-0150-7. [ Links ]
Mngeni A, Musampa CM, Nakin MDV. 2017a. The effects of sand mining on rural communities. WIT Transactions on Ecology and the Environment. 210:443-453. https://doi.org/10.2495/SDP160371. [ Links ]
Mngeni A, Betek C, Musampa CM, Nakin MDV. 2017b. The physical and environmental impacts of sand mining. Transactions of the Royal Society of South Africa. 72:1-5. https://doi.org/10.1080/0035919X.2016.1209701. [ Links ]
Mucina L, Rutherford MC. 2006. The Vegetation of South Africa, Lesotho and Swaziland. Pretoria: South African National Biodiversity Institute. [ Links ]
Naidoo D. 2008. Overview of South African sand and aggregate industry. Pretoria: Mineral Economics, Department of Mineral Resources. [ Links ]
NASA POWER Project. 2025. NASA Prediction of Worldwide Energy Resource (POWER) Data Access Viewer. NASA Langley Research Center. https://power.larc.nasa.gov
Nkamisa M, Ndhleve S, Nakin MDV, Mngeni A, Kabiti HM. 2022. Analysis of trends, recurrences, severity and frequency of droughts using standardised precipitation index: Case of OR Tambo District Municipality, Eastern Cape, South Africa. Jàmbá: Journal of Disaster Risk Studies. 14:a1147. https://doi.org/10.4102/jamba.v14i1.1147. [ Links ]
Obrist MK, Duelli P. 2010. Rapid biodiversity assessment of arthropods for monitoring average local species richness and related ecosystem services. Biodiversity and Conservation. 19:2201-2220. https://doi.org/10.1007/s10531-010-9832-y. [ Links ]
Ossola A, Nash MA, Christie FJ, Hahs AK, Livesley SJ. 2015. Urban habitat complexity affects species richness but not environmental filtering of morphologically-diverse ants. PeerJ. 3:e1356. https://doi.org/10.7717/peerj.1356. [ Links ]
Perry KI, Sivakoff FS, Wallin KF, Wenzel JW, Herms DA. 2021. Forest disturbance and arthropods: small-scale canopy and understory disturbances alter movement of mobile arthropods. Ecosphere. 12:e03771. https://doi.org/10.1002/ecs2.3771. [ Links ]
Picker M, Graffiths C, Weaving A. 2019. Field Guide to Insects of South Africa. Cape Town: Penguin Random House South Africa. [ Links ]
R Core Team. 2020. R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing.
Rahman ML, Tarrant S, McCollin D, Ollerton J. 2015. Vegetation cover and grasslands in the vicinity accelerate development of carabid beetle assemblages on restored landfill sites. Zoology and Ecology. 25:347-354. https://doi.org/10.1080/21658005.2015.1068521. [ Links ]
Rentier ES, Cammeraat LH. 2022. The environmental impacts of river sand mining. Science of the Total Environment. 838:155877. https://doi.org/10.1016/j.scitotenv.2022.155877. [ Links ]
Rochon J, Gondan M, Kieser M. 2012. To test or not to test: preliminary assessment of normality when comparing two independent samples. BMC Medical Research Methodology. 12:81. https://doi.org/10.1186/1471-2288-12-81. [ Links ]
Roberson EJ, Chips MJ, Carson WP, Rooney TP. 2016. Deer herbivory reduces web-building spider abundance by simplifying forest vegetation structure. PeerJ. 4:e2538. https://doi.org/10.7717/peerj.2538. [ Links ]
Sackmann P, Farji-Brener A. 2006. Effect of fire on ground beetles and ant assemblages along an environmental gradient in NW Patagonia: does habitat type matter? Écoscience. 13:360-371. https://doi.org/10.2980/i1195-6860-13-3-360.1. [ Links ]
Samways MJ, McGeoch MA, New TR. 2010. Insect Conservation: A Handbook of Approaches and Methods. New York: Oxford University Press. [ Links ]
Seifert B, Kaufmann B, Fraysse L. 2024. A taxonomic revision of the Palaearctic species of the ant genus Tapinoma Mayr 1861 (Hymenoptera: formicidae). Zootaxa. 5435:1-74. https://doi.org/10.11646/zootaxa.5435.1.1. [ Links ]
Sharaf MR, Fisher BL, Collingwood CA, Aldawood AS. 2017. Ant fauna (Hymenoptera: Formicidae) of the Socotra Archipelago (Yemen): zoogeography, distribution and description of a new species. Journal of Natural History. 51:317-378. http://dx.doi.org/10.1080/00222933.2016.1271157. [ Links ]
Swart RC, Pryke JS, Roets F. 2019. The intermediate disturbance hypothesis explains arthropod beta-diversity responses to roads that cut through natural forests. Biological Conservation. 236:243-251. https://doi.org/10.1016/j.biocon.2019.03.045. [ Links ]
Tajthi B, Horváth R, Mizser S,Nagy DD, Tóthmérész B. 2017. Spiderassemblages in floodplain forests along an urbanization gradient. Community Ecology. 18:311-318. https://doi.org/10.1556/168.2017.18.3.10. [ Links ]
Uehara-Prado M., Fernandes JO, Bello AM, Machado G, Santos AJ, Vaz-De-Mello FZ, Freitas AVL. 2009. Selecting terrestrial arthropods as indicators of small-scale disturbance: a first approach in the Brazilian Atlantic forest. Biological Conservation. 142:1220-1228. https://doi.org/10.1016/j.biocon.2009.01.008. [ Links ]
Van Schalkwyk J, Pryke JS, Samways MJ, Gaigher R. 2021. Maintaining high vegetation structural diversity in the landscape promotes arthropod diversity in dynamic production areas. Landscape Ecology. 36:1773-1785. https://doi.org/10.1007/s10980-021-01253-3. [ Links ]
Venables WN, Ripley BD. 2002. Statistics and Computing: Modern Applied Statistics with S. New York: Springer. https://doi.org/10.1007/978-0-387-21706-2. [ Links ]
Vidal-Cordero JM, Arnan X, Rodrigo A, Cerdá X, Boulay R. 2022. Four-year study of arthropod taxonomic and functional responses to a forest wildfire: epigeic ants and spiders are affected differently. Forest Ecology and Management. 520:120379. https://doi.org/10.1016/j.foreco.2022.120379. [ Links ]
Wagner PM, Abagandura GO, Mamo M, Weissling T, Wingeyer A, Bradshaw JD. 2021. Abundance and diversity of dung beetles (Coleoptera: Scarabaeoidea) as affected by grazing management in the Nebraska sandhills ecosystem. Environmental Entomology. 50:222-231. https://doi.org/10.1093/ee/nvaa130. [ Links ]
White RE. 1998. The Beetles of North America. Boston: Houghton Mifflin Harcourt. [ Links ]
Willett TR. 2001. Spiders and other arthropods as indicators in old-growth versus logged redwood stands. Restoration Ecology. 9:410-420. https://doi.org/10.1046/j.1526-100X.2001.94010.x. [ Links ]
Yekwayo I, Pryke JS, Roets F, Samways MJ. 2017. Responses of ground living arthropods to landscape contrast and context in a forest-grassland mosaic. Biodiversity and Conservation. 26:631-651. https://doi.org/10.1007/s10531-016-1262-z. [ Links ]
Yeo K, Kouakou LMM, Touao MK, Gauze E, Ouattara K, Soro AN. 2019. Ants response to mining prospection disturbances across vegetation zones in tropical mountain chains of Mount Nimba, Guinea, West Africa. International Journal of Biological and Chemical Sciences. 13:899-913. https://dx.doi.org/10.4314/ijbcs.v13i2.26.
 Correspondence:
Correspondence:
Inam Yekwayo
Email: iyekwayo@wsu.ac.za
Received: 14 November 2024
Accepted: 13 June 2025
SUPPLEMENTARY DATA:
Supplementary Tables S1 and S2 are available online in a separate pdf













